This article has been
cited by other articles in ScienceCentral.
Abstract
Lymphoepithelioma-like carcinoma (LELC), an undifferentiated carcinoma with intense lymphoplasmacytic infiltrates, is commonly reported in the nasopharynx and occasionally in other organs. Pure type of LELC has previously been reported in the gallbladder. Mixed type could be reportable in comparison with other organs. Here we present a case of an 83-year-old man with mixed LELC and adenocarcinoma in the gallbladder. To the best of our knowledge, this is the first case of mixed LELC and adenocarcinoma in the gallbladder.
Go to :

Keywords: Lymphoepithelioma-like carcinoma, Adenocarcinoma, Gallbladder
INTRODUCTION
Schmincke has described an undifferentiated nasopharyngeal carcinoma showing a diffuse pattern mimicking lymphoma.
1 This has been designated as lymphoepithelial carcinoma (lymphoepithelioma) or undifferentiated carcinoma with lymphocytic stroma. Similar tumors occurring in extrapharyngeal sites have been referred to as lymphoepithelioma-like carcinoma (LELC). LELC has been reported in the lung, salivary gland, thymus, stomach, urinary bladder, uterus, vagina, bile duct, and gallbladder.
2345678 Only two cases of pure type LELC involving the gallbladder have been described in the literature.
78 In mixed type of LELC in the urinary bladder, invasive urothelial carcinoma, squamous cell carcinoma, adenocarcinoma, adenourothelial carcinoma, or
in situ carcinoma of each type has been associated with LELC.
9 Epstein-Barr virus (EBV) infection has been implicated in the oncogenesis of almost all cases of lymphoepithelial carcinoma of the nasopharynx.
10 However, EBV is only detected in LELC of the stomach, esophagus, hepatobiliary tract, lung, thymus, and the salivary gland, but not in LELC of the breast, skin, or urinary bladder.
91112 Herein, we report the first case of mixed type LELC of the gallbladder not associated with EBV.
Go to :

CASE
An 83-year-old Korean man was referred from a local clinic under the suspicion of gallbladder cancer following an abdominal computed tomography (CT) scan. Fluorodeoxyglucose (FDG)-positron emission tomography (PET)-CT revealed a hypermetabolic lesion in the gallbladder (
Fig. 1) suggesting malignancy. The patient had epigastric pain for three days prior to his visit to the clinic. He had no significant medical history. Physical examinations revealed no abnormal findings.
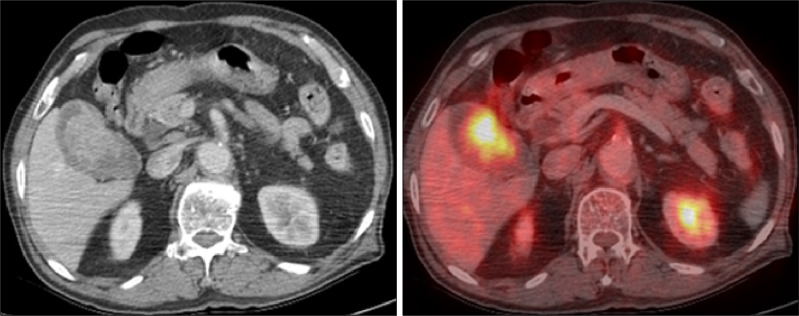 | Fig. 1PET-CT findings showing a hypermetabolic lesion in the gallbladder suggestive of malignancy.
|
Laparoscopic cholecystectomy was performed. No peritoneal or liver metastasis was observed and the cystic duct margin was free of tumor. The resected gallbladder showed diffuse wall thickening and a 3×4 cm-sized mass.
Pathological examination of surgical specimens revealed that the tumor infiltrated diffusely from the mucosa to the subserosa with multiple erosions. The majority (>95% of tumor volume) of the tumor consisted of nests and individual cells of undifferentiated carcinoma admixed with prominent lymphoplasmocytic infiltrates. The epithelial cells had large vesicular oval-shaped nuclei with occasional large prominent nucleoli (
Fig. 2). Immunohistochemical staining results were positive for AE1/AE3 and cytokeratin 7 but negative for cytokeratin 20, carcinoembryonic antigen, or EBV. These findings were consistent with those of EBV-negative LELC. The remaining part of the tumor (<5%) consisted of adenocarcinoma confined to the mucosa. The glandular component was characterized as a well differentiated adenocarcinoma partially overlying the LELC. A transition from adenocarcinoma to LELC was also identified. Lymphoid stroma, especially those surrounding the LELC, was composed of a mixture of CD3-positive T lymphocytes, CD20-positive B lymphocytes, and plasma cells with predominantly CD3-positive T lymphocytes and plasma cells (
Fig. 3).
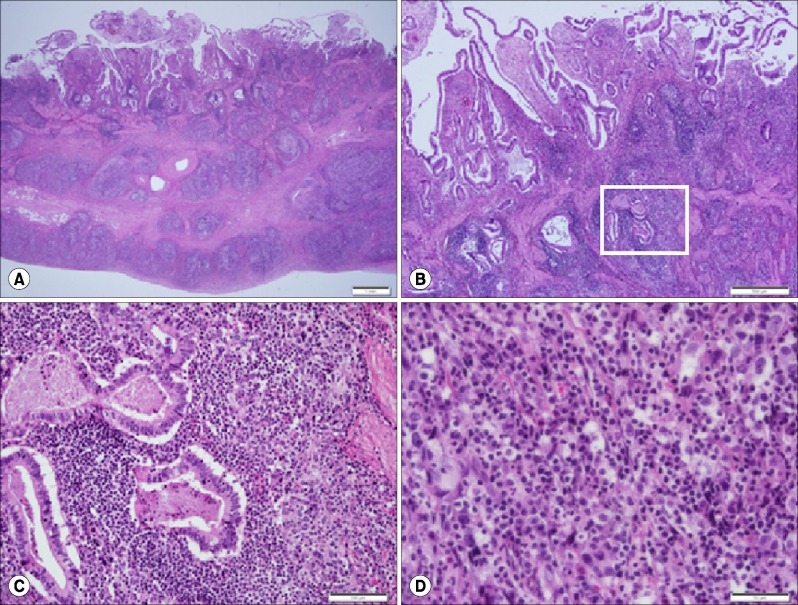 | Fig. 2Microscopic findings. Transmural infiltration of neoplastic cells with lymphoid stroma is identified in the gallbladder wall. Scale bar: 1 mm (A). Well differentiated adenocarcinoma confined to the mucosa is partially overlying lymphoepithelioma-like carcinoma. Scale bar: 500 µm (B). Higher magnification of the boxed area in Figure 2B shows transition from adenocarcinoma to undifferentiated carcinoma. Scale bar: 100 µm (C). Large vesicular oval-shaped nuclei with occasional large prominent nucleoli and lymphocytic infiltration are found. Scale bar: 50 µm (D).
|
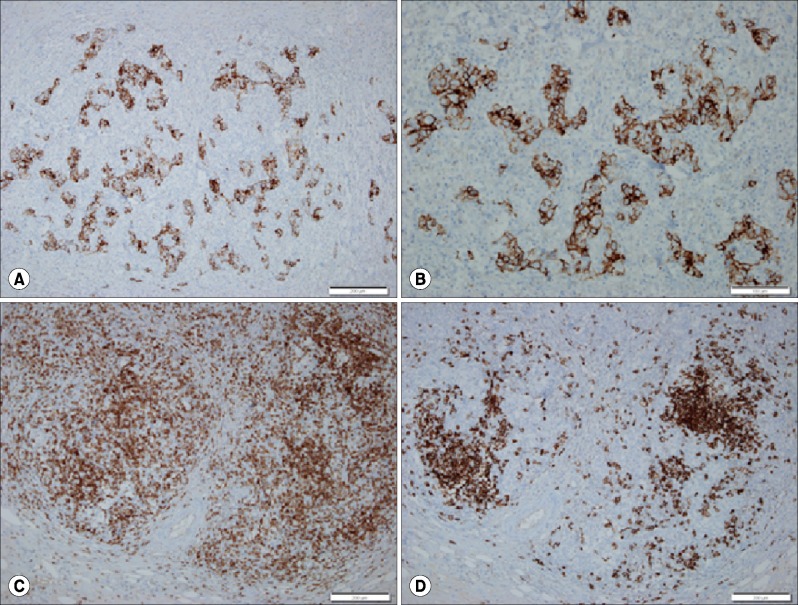 | Fig. 3Immunohistochemical findings. Immunostaining for cytokeratin 7 shows diffuse infiltration of the undifferentiated carcinoma cells in the gallbladder wall. Scale bar: 200 µm (A). Higher magnification shows scattered neoplastic infiltration with lymphoid stroma. Scale bar: 200 µm (B). The same area shows abundant CD3-positive T lymphocytes (C) and CD20-positive B lymphocytes (D) around the tumor cell nests. Scale bar: 200 µm.
|
Based on these findings, a diagnosis of non-EBV-associated mixed type LELC of the gallbladder was made. The patient was discharged without post-operative complications. At present time (4 months after the surgery), the patient has no sign of recurrence.
Go to :

DISCUSSION
Lymphoepithelioma was originally described in the nasopharynx of Asian patients.
1113 It is closely associated with EBV.
1113 There is also a close pathogenetic relationship between LELC and EBV.
11 Cases in individuals of Asian ethnicity have predominated literature reports of biliary LELC.
6 LELC is classified as pure type when 100% of the tumor demonstrates an LELC pattern or mixed type when the tumor is associated with usual urothelial carcinoma, squamous cell carcinoma, or adenocarcinoma.
9 Reported cases of LELC of the urinary bladder have been classified as the pure type in 57% of cases and the mixed type in 43% of cases.
9 According to some studies, pure or predominant LELC has a better prognosis than focal LELC.
111314 However, Tamas et al.
9 have reported that there is no significant difference in prognosis between pure type and mixed type of LELC, although only a few cases of pure type LELC are treated with chemotherapy.
Some cases of LELC are associated with EBV, but some cases are not associated with EBV. There is no obvious histopathological difference between EBV-positive and EBV-negative LELCs in each organ.
67912 LELC can occur at various anatomic sites. Only two cases of pure type LELC of the gallbladder have been reported.
78 EBV is frequently identified in LELC in the lungs, stomach, thymus, and salivary gland, but not in other organs.
6789 The two cases of LELC of the gallbladder
78 and one case of common bile duct LELC
6 reported in the literature are negative for EBV. Therefore, the involvement of EBV genome integration in LELC oncogenesis might depend on the site of origin of LELC.
The overlying mucosa in mixed type LELC can show dysplasia, carcinoma
in situ, or invasive carcinoma.
9 In the present case, dysplasia and adenocarcinoma of the overlying mucosa were observed. The mixed component was minor (<5%) and confined to the mucosa only.
LELC is usually associated with a more favorable prognosis compared to conventional carcinoma.
9 The presence or absence of EBV has no prognostic significance in LELC of various organs.
15 In biliary LELC, differences between the prognoses of EBV-positive and EBV-negative cases have not been established yet due to the limited number of cases reported to date.
In conclusion, we report a case of non-EBV-associated mixed type LELC of the gallbladder. Due to the rarity of this disease, additional case studies are needed to establish clinicopathological findings of LELC of the gallbladder.
Go to :

ACKNOWLEDGEMENTS
This study was supported by Chosun University Hospital research fund 2015.
Go to :

References
1. Schmincke A. Über lymphoepitheliale geschwülste. Beitr pathol Anat. 1921; 68:161–170.
2. Castro CY, Ostrowski ML, Barrios R, Green LK, Popper HH, Powell S, et al. Relationship between Epstein-Barr virus and lymphoepithelioma-like carcinoma of the lung: a clinicopathologic study of 6 cases and review of the literature. Hum Pathol. 2001; 32:863–872. PMID:
11521232.

3. Tsai CC, Chen CL, Hsu HC. Expression of Epstein-Barr virus in carcinomas of major salivary glands: a strong association with lymphoepithelioma-like carcinoma. Hum Pathol. 1996; 27:258–262. PMID:
8600040.

4. Kohrenhagen N, Eck M, Höller S, Dietl J. Lymphoepithelioma-like carcinoma of the uterine cervix: absence of Epstein-Barr virus and high-risk human papilloma virus infection. Arch Gynecol Obstet. 2008; 277:175–178. PMID:
17674013.

5. Dietl J, Horny HP, Kaiserling E. Lymphoepithelioma-like carcinoma of the vagina: a case report with special reference to the immunophenotype of the tumor cells and tumor-infiltrating lymphoreticular cells. Int J Gynecol Pathol. 1994; 13:186–189. PMID:
8005741.
6. Ishida M, Mori T, Shiomi H, Naka S, Tsujikawa T, Andoh A, et al. Non-Epstein-Barr virus associated lymphoepithelioma-like carcinoma of the inferior common bile duct. World J Gastrointest Oncol. 2011; 3:111–115. PMID:
21860687.

7. Todd DL, Ro JY, Gulley ML, Ayala AG. Lymphoepitheliomalike carcinoma of the gallbladder. Int J Surg Pathol. 1996; 4:183–187.

8. Sinha PK, Mangla V, Behari C, Rastogi A, Chattopdhyay TK. Lymphoepithelioma-like carcinoma: an unusual gall bladder tumour. Trop Gastroenterol. 2014; 35:182–183. PMID:
26012324.

9. Tamas EF, Nielsen ME, Schoenberg MP, Epstein JI. Lymphoepithelioma-like carcinoma of the urinary tract: a clinicopathological study of 30 pure and mixed cases. Mod Pathol. 2007; 20:828–834. PMID:
17541442.

10. Gulley ML, Amin MB, Nicholls JM, Banks PM, Ayala AG, Srigley JR, et al. Epstein-Barr virus is detected in undifferentiated nasopharyngeal carcinoma but not in lymphoepithelioma-like carcinoma of the urinary bladder. Hum Pathol. 1995; 26:1207–1214. PMID:
7590694.

11. Amin MB, Ro JY, Lee KM, Ordóñez NG, Dinney CP, Gulley ML, et al. Lymphoepithelioma-like carcinoma of the urinary bladder. Am J Surg Pathol. 1994; 18:466–473. PMID:
8172321.

12. Henderson-Jackson E, Nasir NA, Hakam A, Nasir A, Coppola D. Primary mixed lymphoepithelioma-like carcinoma and intra-hepatic cholangiocarcinoma: a case report and review of literature. Int J Clin Exp Pathol. 2010; 3:736–741. PMID:
20830246.
13. Lopez-Beltrán A, Luque RJ, Vicioso L, Anglada F, Requena MJ, Quintero A, et al. Lymphoepithelioma-like carcinoma of the urinary bladder: a clinicopathologic study of 13 cases. Virchows Arch. 2001; 438:552–557. PMID:
11469686.

14. Holmäng S, Borghede G, Johansson SL. Bladder carcinoma with lymphoepithelioma-like differentiation: a report of 9 cases. J Urol. 1998; 159:779–782. PMID:
9474147.
15. Iezzoni JC, Gaffey MJ, Weiss LM. The role of Epstein-Barr virus in lymphoepithelioma-like carcinomas. Am J Clin Pathol. 1995; 103:308–315. PMID:
7872253.

Go to :







 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download