1. Kim WJ, Hwang S, Lee YJ, Kim KH, Park KM, Ahn CS, et al. Clinicopathological features and long-term outcomes of intraductal papillary neoplasms of the intrahepatic bile duct. J Gastrointest Surg. 2016; 20:1368–1375. PMID:
26873016.

2. Nakanuma Y, Sato Y, Harada K, Sasaki M, Xu J, Ikeda H. Pathological classification of intrahepatic cholangiocarcinoma based on a new concept. World J Hepatol. 2010; 2:419–427. PMID:
21191517.

3. Itatsu K, Zen Y, Ohira S, Ishikawa A, Sato Y, Harada K, et al. Immunohistochemical analysis of the progression of flat and papillary preneoplastic lesions in intrahepatic cholangiocarcinogenesis in hepatolithiasis. Liver Int. 2007; 27:1174–1184. PMID:
17919228.

4. Nakanuma Y. A novel approach to biliary tract pathology based on similarities to pancreatic counterparts: is the biliary tract an incomplete pancreas? Pathol Int. 2010; 60:419–429. PMID:
20518896.

5. Lee SS, Kim MH, Lee SK, Jang SJ, Song MH, Kim KP, et al. Clinicopathologic review of 58 patients with biliary papillomatosis. Cancer. 2004; 100:783–793. PMID:
14770435.

6. Rocha FG, Lee H, Katabi N, DeMatteo RP, Fong Y, D'Angelica MI, et al. Intraductal papillary neoplasm of the bile duct: a biliary equivalent to intraductal papillary mucinous neoplasm of the pancreas? Hepatology. 2012; 56:1352–1360. PMID:
22504729.

7. Furukawa T, Klöppel G, Volkan Adsay N, Albores-Saavedra J, Fukushima N, Horii A, et al. Classification of types of intraductal papillary-mucinous neoplasm of the pancreas: a consensus study. Virchows Arch. 2005; 447:794–799. PMID:
16088402.

8. Ohtsuka M, Shimizu H, Kato A, Yoshitomi H, Furukawa K, Tsuyuguchi T, et al. Intraductal papillary neoplasms of the bile duct. Int J Hepatol. 2014; 2014:459091. PMID:
24949206.

9. Yeh TS, Tseng JH, Chiu CT, Liu NJ, Chen TC, Jan YY, et al. Cholangiographic spectrum of intraductal papillary mucinous neoplasm of the bile ducts. Ann Surg. 2006; 244:248–253. PMID:
16858187.

10. Wu SD, Lu CD, Lu CJ, Huang J, Zhou J. Mucin-producing intrahepatic biliary papillomatosis. Surg Today. 2010; 40:845–850. PMID:
20740348.

11. Yang J, Wang W, Yan L. The clinicopathological features of intraductal papillary neoplasms of the bile duct in a Chinese population. Dig Liver Dis. 2012; 44:251–256. PMID:
21930444.

12. Naito Y, Kusano H, Nakashima O, Sadashima E, Hattori S, Taira T, et al. Intraductal neoplasm of the intrahepatic bile duct: clinicopathological study of 24 cases. World J Gastroenterol. 2012; 18:3673–3680. PMID:
22851859.

13. Kloek JJ, van der Gaag NA, Erdogan D, Rauws EA, Busch OR, Gouma DJ, et al. A comparative study of intraductal papillary neoplasia of the biliary tract and pancreas. Hum Pathol. 2011; 42:824–832. PMID:
21292296.

14. Zen Y, Fujii T, Itatsu K, Nakamura K, Minato H, Kasashima S, et al. Biliary papillary tumors share pathological features with intraductal papillary mucinous neoplasm of the pancreas. Hepatology. 2006; 44:1333–1343. PMID:
17058219.

15. Wu SD, Lu CD, Lu CJ, Huang J, Zhou J. Mucin-producing intrahepatic biliary papillomatosis. Surg Today. 2010; 40:845–850. PMID:
20740348.

16. Ohtsuka M, Kimura F, Shimizu H, Yoshidome H, Kato A, Yoshitomi H, et al. Similarities and differences between intraductal papillary tumors of the bile duct with and without macroscopically visible mucin secretion. Am J Surg Pathol. 2011; 35:512–521. PMID:
21412069.

17. Li T, Ji Y, Zhi XT, Wang L, Yang XR, Shi GM, et al. A comparison of hepatic mucinous cystic neoplasms with biliary intraductal papillary neoplasms. Clin Gastroenterol Hepatol. 2009; 7:586–593. PMID:
19245849.

18. Wan XS, Xu YY, Qian JY, Yang XB, Wang AQ, He L, et al. Intraductal papillary neoplasm of the bile duct. World J Gastroenterol. 2013; 19:8595–8604. PMID:
24379576.

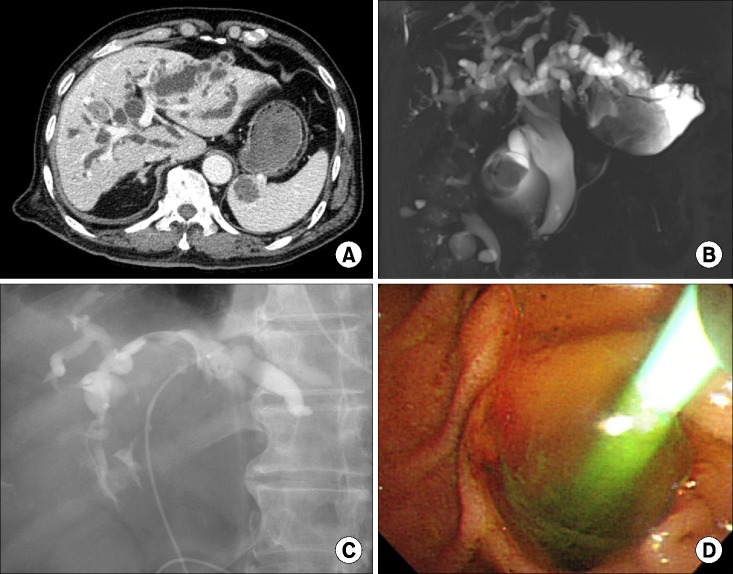
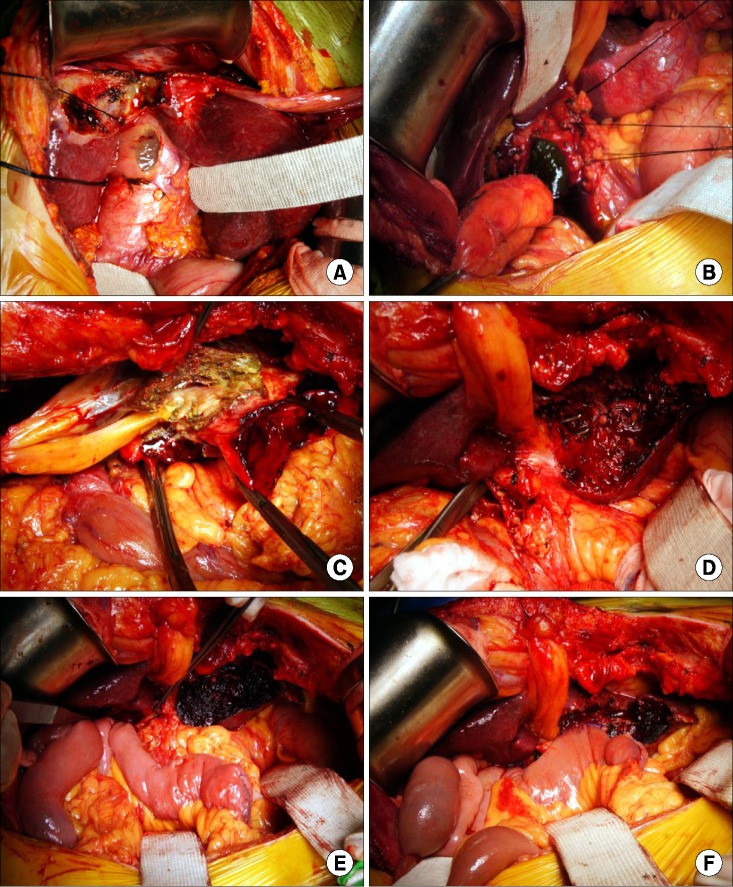
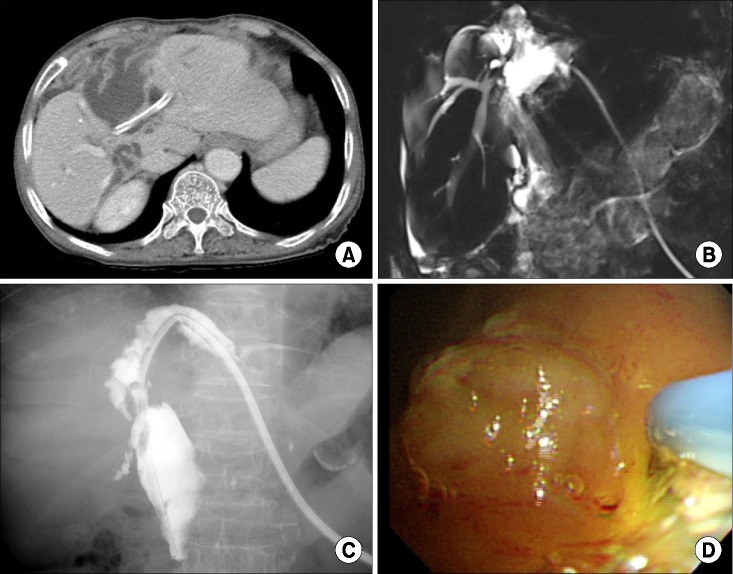
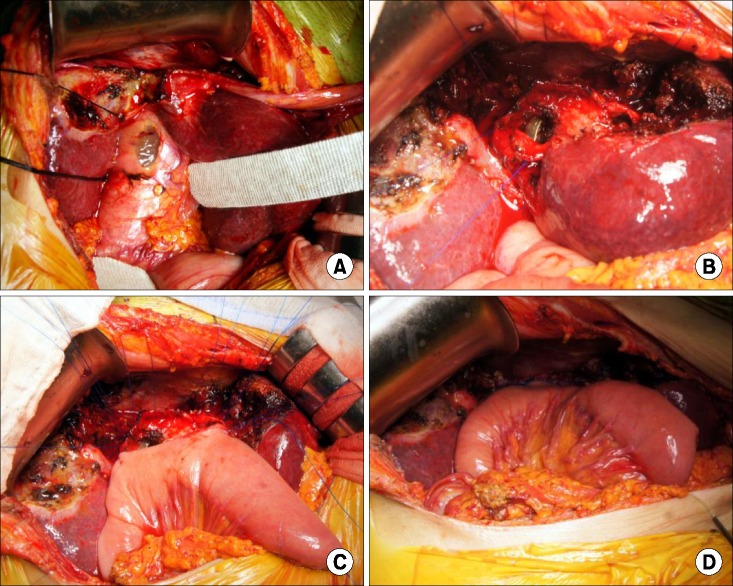




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download