Abstract
Backgrounds/Aims
In hepatocellular carcinoma (HCC), bile duct invasion occurs far more rarely than vascular invasion and is not well characterized. In addition, the pathologic finding of bile duct invasion is not considered an independent prognostic factor for HCC following surgery. In this study, we determined the characteristics of HCC with bile duct invasion, and assessed the clinical significance of bile duct invasion.
Methods
We retrospectively reviewed the medical records of 363 patients who underwent hepatic resection for HCC at Seoul National University Hospital (SNUH) from January 2009 to December 2011. Preoperative, operative, and pathological data were collected. The risk factors for recurrence and survival were analyzed. Subsequently, the patients were divided into 2 groups according to disease stage (American Joint Committee on Cancer/International Union Against Cancer 7th edition): early stage (T1 and 2) and advanced stage (T3 and 4) group; and risk factors in the sub-groups were analyzed.
Results
Among 363 patients, 13 showed bile duct invasion on pathology. Patients with bile duct invasion had higher preoperative total bilirubin levels, greater microvascular invasion, and a higher death rate than those without bile duct invasion. In multivariate analysis, bile duct invasion was not an independent prognostic factor for survival for the entire cohort, but, was an independent prognostic factor for early stage.
Hepatocellular carcinoma (HCC), is an aggressive cancer that frequently develops as a result of chronic liver disease or cirrhosis. HCC invasion progresses from the vascular system to the bile duct.1 Because of its complex pathophysiology and treatment, there are several staging systems and treatment strategies for HCC. However, no globally standardized staging system has yet been established. Currently, the most commonly used prognostic factors are vascular invasion, intrahepatic metastasis, tumor size and number, preoperative tumor marker levels, ascites, portal vein tumor thrombosis, and hepatitis B viral (HBV)-DNA status.2 Bile duct invasion is not considered a prognostic factor currently. HCC with bile duct invasion, previously known as icteric HCC, is much rarer than HCC with vascular invasion and is not well characterized.3 Several studies since 1982 have investigated the characteristics of HCC with bile duct invasion and patient prognosis.3 However, the prognosis of these patients remains controversial.3 Furthermore, bile duct invasion has not been included in the HCC staging system.2
Herein, we investigated the characteristics of HCC with bile duct invasion in relation to cases of the disease without such invasion, in order to elucidate the clinical significance of bile duct invasion in terms of survival following surgical resection.
We retrospectively reviewed data from pathologically proven HCC cases of patients who underwent hepatic resection between January 2009 and December 2011 in Seoul National University Hospital (SNUH). Patients with incomplete (pathologic margin positive) resection, missing clinical data and those lost to follow-up were excluded. Thus, 363 patients were enrolled in the study. For comparative prognostic implications, clinical information including age, gender, epidemiologic data (underlying liver disease), biochemical data (α-fetoprotein [AFP], vitamin K agonist-II [PIVKA-II] induced protein by, and total bilirubin level), radiology data (presence and grade of bile duct invasion), preoperative treatments (transarterial chemoembolization [TACE] and, radiofrequency ablation [RFA]), operative technique and findings (tumor size and number as well as bile duct and vascular invasion), pathological data such as tumor stage (TNM staging by the American Joint Committee on Cancer/International Union Against Cancer [AJCC/UICC], 7th edition), and Edmondson grade were collected.
Patient follow-up was performed routinely at 1, 3, and 6 months after surgical resection, and then every 3-4 months as needed. At each follow-up, patients underwent imaging work-up (computed tomography, magnetic resonance imaging, or ultrasonography) and serological tests (tumor markers measurement and liver function tests).
The 2 groups of HCC patients with and without bile duct invasion were compared for disease characteristics and prognosis for recurrence and survival. The risk factors influencing recurrence-free survival and overall survival were analyzed. Patients were sub-categorized into early stage (T1 and T2) and advanced stage (T3 and T4) disease groups, and sub-group analysis of risk factors according to disease stage was performed.
Chi-square and Student's t-tests were used to compare proportions and means of characteristics between patients with or without bile duct invasion. Multivariate Cox modeling was performed using potential risk factors and covariates to identify significant prognostic factors. Forward stepwise selection using likelihood ratios for entry and exit criteria was used to develop the final multivariate Cox proportional-hazards model. Continuous variables were categorized using exploratory data analysis. AFP and PIVKA-II were categorized by the cut-off limits of 200 mg/ml and 400 uAU/ml, respectively. Hazard ratios (HR) were estimated using the Cox proportional-hazard methodology, and estimates were reported as HR (95% confidence interval). Kaplan-Meier product-limit methodology was used to compare survival estimates between matched data sets (with and without bile duct invasion), and an unadjusted comparison of survival was performed using the log-rank test. All statistical analyses were performed using SPSS 18 for Windows (Chicago, IL), and p-values<0.05 were considered statistically significant.
The mean age of the HCC patients was 56±8.03 years, and the male-to-female ratio was approximately 4 to 1. The maximum tumor size was 5.6±3.23 cm in diameter. During the mean follow-up period of 3.5 years (2-58 months), disease recurrence occurred in 260 of 363 patients and 71 patients died.
Table 1 showed the comparative clinical characteristics of patients with and without bile duct invasion. There were no significant differences in age, sex, etiology, preoperative AFP and PIVK-II level, pathologic tumor size, T-stage, histological grade, or recurrence rate between the 2 groups. However, 85% of patients with bile duct invasion developed microvascular invasion, as compared to 43% of patients without bile duct invasion (p=0.04). In addition, the patients with bile duct invasion had higher total bilirubin levels (p=0.04). The 6 patients without bile duct invasion showed a total bilirubin level >4 mg/dl, with relatively huge masses compressing the bile ducts. Elevated levels of bilirubin were attributed to mass effect. All 13 patients with bile duct invasion showed disease recurrence and 6 of these patients (46.2%) died. Among these 13 patients, bile duct invasion was diagnosed preoperatively in 10 patients and detected incidentally in the other 3 patients. In the patient group with bile duct invasion, the levels of bile duct tumor thrombi were as follows: common hepatic duct to the first confluence, 5 cases; the first to second confluence, 3 cases; above the second confluence, 3 cases; and no gross invasion, 2 cases. Of the 5 patients with invasion of the common hepatic duct, 2 patients underwent percutaneous transhepatic biliary drainage, and total 4 patients received TACE before surgery. Two patients (15.4%) had high preoperative bilirubin levels, indicative of icteric HCC.
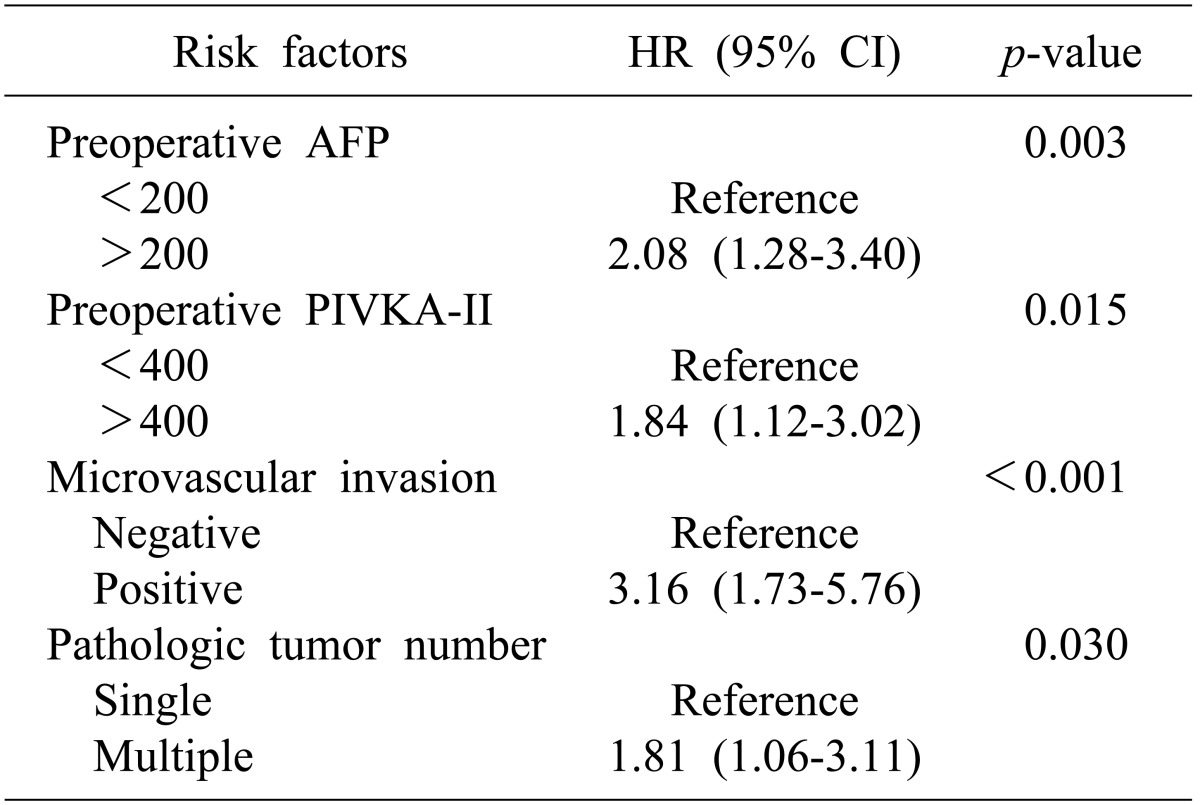
In multivariate analysis, microvascular invasion was identified as a risk factor for recurrence (p=0.01, Table 2). In addition, preoperative AFP (p<0.01), PIVKA-II level (p=0.02), microvascular invasion (p<0.01), and pathologic tumor number (p=0.03) were identified as factors influencing patient survival (Table 3).
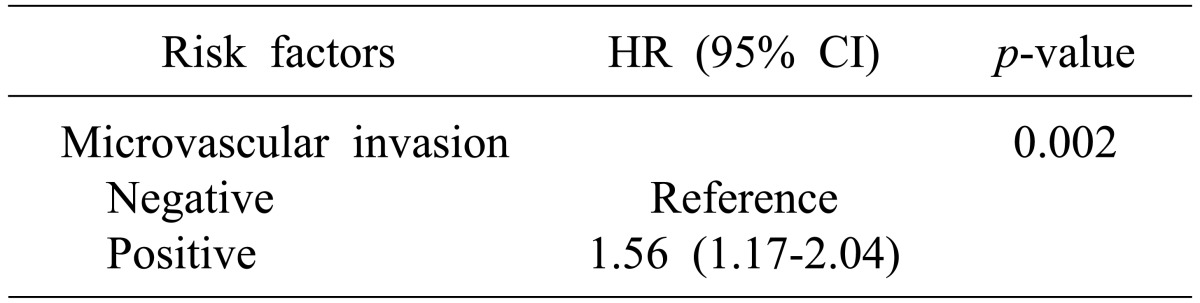
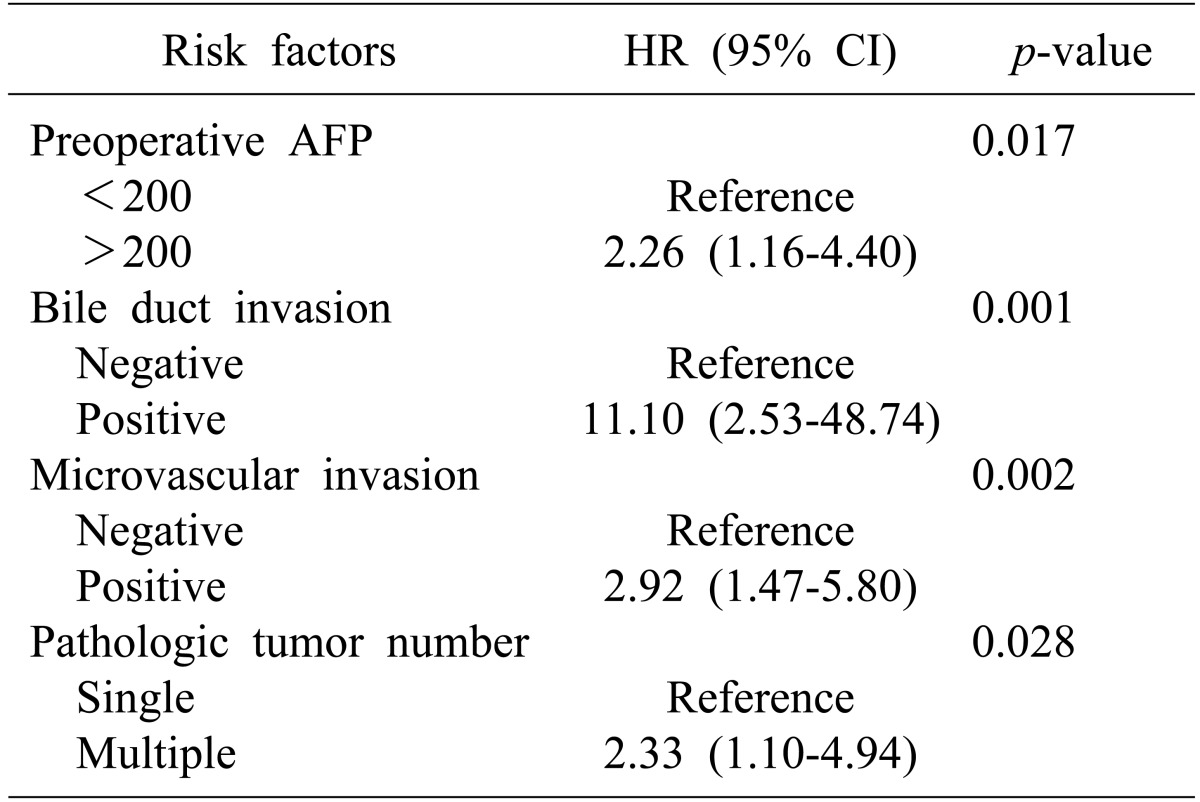
In subgroup analysis according to disease stage, microvascular invasion was identified as a risk factor for recurrence in early stage HCC (p<0.01, Table 4). However, for overall survival, bile duct invasion (p<0.01), together with preoperative AFP (p=0.02), microvascular invasion (p<0.01), and tumor number (p=0.03, Table 5) were significant risk factors. Bile duct invasion was not an independent risk factor for recurrence-free survival and overall survival in the advanced stage group.
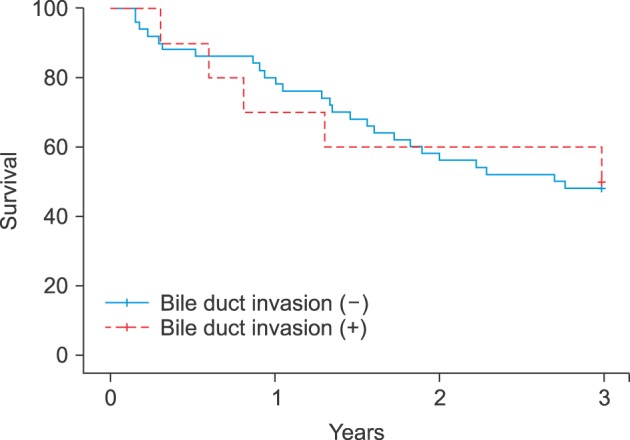
Overall survival was significantly poorer in patients with bile duct invasion than in those without bile duct invasion in the early stage HCC group (100% vs. 96% at 1 year, 65.5% vs. 90.3% at 2 years, and 32.5% vs. 88.7% at 3 years, p<0.01, Fig. 1). However, survival was similar in patients with bile duct invasion and in those without bile duct invasion in the advanced HCC group (Fig. 2).
Several studies since 1982, have investigated the characteristics and prognosis of HCC with bile duct invasion; however, the prognosis of HCC patients with bile duct invasion remains controversial.3456789101112 Lau's study of 530 HCC patients, indicated that patients with bile duct invasion who received proper drainage of obstructive jaundice had a similar prognosis to that of HCC patients without bile duct invasion.9 However, in that study, jaundice was defined as both, due to hepatic insufficiency as well as obstructive jaundice. Satoh et al.5 and Shiomi et al.6 also reported that prognosis of HCC with bile duct invasion is similar to that of patients without bile duct thrombus. Furthermore, recent studies have shown that, a poor prognosis can be overcome by early detection10 or adequate biliary drainage13 and curative resection.1456141516 However, most of these previous studies supported the hypothesis that the prognosis of HCC with bile duct invasion can be worse than HCC without invasion,389111217 which was in agreement with our results.
Until recently, bile duct invasion was not considered a staging factor for HCC in the pathological TNM staging system of AJCC/UICC.2 This is likely due to both, a small number of patients and the lack of a clear characterization of patients with bile duct invasion in the previous studies. We investigated whether bile duct invasion can be used as an independent risk factor for determining patient survival. The results of our analyses indicated that bile duct invasion was not a significant risk factor influencing survival in all HCC patients following hepatic resection, but only for those with early stage disease. Thus, bile duct invasion can be considered an independent factor for HCC staging, especially in early stage disease.
Previous studies have suggested a number of potential reasons for the poor prognosis of HCC patients with bile duct invasion.134791318 The first hypothesis proposes, that the infiltrative nature of HCC with bile duct invasion influences the prognosis.134 However, in the present study, of the 13 patients with bile duct invasion, only 2 showed infiltrative HCC. Therefore, we concluded that the infiltrative nature of the disease was not the reason for the poor prognosis of these patients.
A second hypothesis, suggests that poor prognosis for HCC with bile duct invasion could be due to obstructive jaundice related to hepatic dysfunction.6791318 However, Shiomi et al.6 and Xiangji et al.13 reported that prognosis does not differ between patients with and without bile duct invasion, if proper biliary drainage is performed. All but 1 of the patients with bile duct invasion in our study showed preoperative total bilirubin levels in the normal range. Thus, HCC with bile duct invasion could result in a poor prognosis due to reasons unrelated to obstructive jaundice. In addition, obstructive jaundice can induce infection and inflammation, which could lead to mortality, especially in the early stages of the disease. In the present study, a review of the medical records of HCC patients revealed that 3 of the early stage HCC patients (2 with T-stage 1 and 1 with T-stage 2 disease) showed bile duct invasion. Their preoperative total bilirubin levels were in the normal range (0.4, 1.0, and 1.0 mg/dl). However, 2 of the patients died during the follow-up period (1 patient with T-stage 1 and 2 patients with T-stage 2). The cause of death in the patients was both cancer recurrence and progression. There was no evidence of hepatic infection or inflammation, thereby disproving the hypothesis on the effect of obstructive jaundice on patient survival.
A third hypothesis, suggests that the higher incidence of microvascular invasion in patients with HCC with bile duct invasion may be a reason for the poor prognosis of patients. The findings of our study supported this hypothesis, although our study included only those patients with complete resection. Considering the mechanisms of hepatic tumor growth, it is not surprising that the majority of cases of bile duct invasion of HCC are accompanied by microvascular invasion.1 Therefore, gross bile duct invasion could be considered as vascular invasion in several staging systems for HCC. In a study involving a group of patients with liver cancer classified according to the Japanese staging systems for HCC (3rd edition) based on preoperative image findings, the presence of bile duct invasion upon preoperative imaging increased the T classification by 1 grade.1920 In early stage HCC, if bile duct invasion does not accompany microvascular invasion, it can result in undetected or hidden microvascular invasion. Among 3 early stage HCC patients with bile duct invasion, only 1 patient (T-stage 2) displayed concurrent microvascular invasion. On the other hand, all patients exhibited disease recurrence during follow-up, despite complete hepatic resection, highlighting the aggressive nature of HCC. In this case, bile duct invasion could be an indicator of hidden microvascular invasion rather than an independent prognostic factor.
Finally, if bile duct invasion significantly influences the prognosis, irrespective of microvascular invasion (in cases of patients with bile duct invasion patient and microvascular invasion), bile duct invasion can eventually be the significant factor.
The third hypothesis best explains the results of the current study. However, the limitation of this study was the relatively small patient cohort. Due to the rarity of HCC patients with bile duct invasion, further studies such as multicenter trials are required to confirm our findings.
In conclusion, the results indicated that bile duct invasion accompanied vascular invasion in most cases of HCC included in the study. HCC with bile duct invasion could be an indicator of undetected microvascular invasion. Furthermore, in early stage HCC, bile duct invasion can be used as an independent prognostic factor for survival.
ACKNOWLDEGEMENTS
We thank Dr. Sukwon Suh and Dr. Youngrok Choi for helpful comments on the manuscript.
References
1. Kasai Y, Hatano E, Seo S, Taura K, Yasuchika K, Uemoto S. Hepatocellular carcinoma with bile duct tumor thrombus: surgical outcomes and the prognostic impact of concomitant major vascular invasion. World J Surg. 2015; 39:1485–1493. PMID: 25651961.

2. Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010; 17:1471–1474. PMID: 20180029.

3. Ikenaga N, Chijiiwa K, Otani K, Ohuchida J, Uchiyama S, Kondo K. Clinicopathologic characteristics of hepatocellular carcinoma with bile duct invasion. J Gastrointest Surg. 2009; 13:492–497. PMID: 19011945.

4. Zeng H, Xu LB, Wen JM, Zhang R, Zhu MS, Shi XD, et al. Hepatocellular carcinoma with bile duct tumor thrombus: a clinicopathological analysis of factors predictive of recurrence and outcome after surgery. Medicine (Baltimore). 2015; 94:e364. PMID: 25569656.
5. Satoh S, Ikai I, Honda G, Okabe H, Takeyama O, Yamamoto Y, et al. Clinicopathologic evaluation of hepatocellular carcinoma with bile duct thrombi. Surgery. 2000; 128:779–783. PMID: 11056440.

6. Shiomi M, Kamiya J, Nagino M, Uesaka K, Sano T, Hayakawa N, et al. Hepatocellular carcinoma with biliary tumor thrombi: aggressive operative approach after appropriate preoperative management. Surgery. 2001; 129:692–698. PMID: 11391367.

7. Kojiro M, Kawabata K, Kawano Y, Shirai F, Takemoto N, Nakashima T. Hepatocellular carcinoma presenting as intrabile duct tumor growth: a clinicopathologic study of 24 cases. Cancer. 1982; 49:2144–2147. PMID: 6280834.

8. Yeh CN, Jan YY, Lee WC, Chen MF. Hepatic resection for hepatocellular carcinoma with obstructive jaundice due to biliary tumor thrombi. World J Surg. 2004; 28:471–475. PMID: 15085398.

9. Lau W, Leung K, Leung TW, Liew CT, Chan MS, Yu SC, et al. A logical approach to hepatocellular carcinoma presenting with jaundice. Ann Surg. 1997; 225:281–285. PMID: 9060584.

10. Qin LX, Ma ZC, Wu ZQ, Fan J, Zhou XD, Sun HC, et al. Diagnosis and surgical treatments of hepatocellular carcinoma with tumor thrombosis in bile duct: experience of 34 patients. World J Gastroenterol. 2004; 10:1397–1401. PMID: 15133842.

11. Shao W, Sui C, Liu Z, Yang J, Zhou Y. Surgical outcome of hepatocellular carcinoma patients with biliary tumor thrombi. World J Surg Oncol. 2011; 9:2. PMID: 21214943.

12. Noda T, Nagano H, Tomimaru Y, Murakami M, Wada H, Kobayashi S, et al. Prognosis of hepatocellular carcinoma with biliary tumor thrombi after liver surgery. Surgery. 2011; 149:371–377. PMID: 20869094.

13. Xiangji L, Weifeng T, Bin Y, Chen L, Xiaoqing J, Baihe Z, et al. Surgery of hepatocellular carcinoma complicated with cancer thrombi in bile duct: efficacy for criteria for different therapy modalities. Langenbecks Arch Surg. 2009; 394:1033–1039. PMID: 19421771.

14. Moon DB, Hwang S, Wang HJ, Yun SS, Kim KS, Lee YJ, et al. Surgical outcomes of hepatocellular carcinoma with bile duct tumor thrombus: a Korean multicenter study. World J Surg. 2013; 37:443–451. PMID: 23188531.

15. Esaki M, Shimada K, Sano T, Sakamoto Y, Kosuge T, Ojima H. Surgical results for hepatocellular carcinoma with bile duct invasion: a clinicopathologic comparison between macroscopic and microscopic tumor thrombus. J Surg Oncol. 2005; 90:226–232. PMID: 15906365.

16. Peng SY, Wang JW, Liu YB, Cai XJ, Deng GL, Xu B, et al. Surgical intervention for obstructive jaundice due to biliary tumor thrombus in hepatocellular carcinoma. World J Surg. 2004; 28:43–46. PMID: 14639491.

17. Kim JM, Kwon CH, Joh JW, Sinn DH, Park JB, Lee JH, et al. Incidental microscopic bile duct tumor thrombi in hepatocellular carcinoma after curative hepatectomy: a matched study. Medicine (Baltimore). 2015; 94:e450. PMID: 25674733.
18. Ueda M, Takeuchi T, Takayasu T, Takahashi K, Okamoto S, Tanaka A, et al. Classification and surgical treatment of hepatocellular carcinoma (HCC) with bile duct thrombi. Hepatogastroenterology. 1994; 41:349–354. PMID: 7959570.
19. Ueno S, Tanabe G, Nuruki K, Hamanoue M, Komorizono Y, Oketani M, et al. Prognostic performance of the new classification of primary liver cancer of Japan (4th edition) for patients with hepatocellular carcinoma: a validation analysis. Hepatol Res. 2002; 24:395–403. PMID: 12479938.

20. Korean Liver Cancer Study Group (KLCSG). National Cancer Center, Korea (NCC). 2014 Korean Liver Cancer Study Group-National Cancer Center Korea practice guideline for the management of hepatocellular carcinoma. Korean J Radiol. 2015; 16:465–522. PMID: 25995680.
Fig. 1
Correlation between overall survival and bile duct invasion in early stage HCC patients (T-stage 1 and 2).
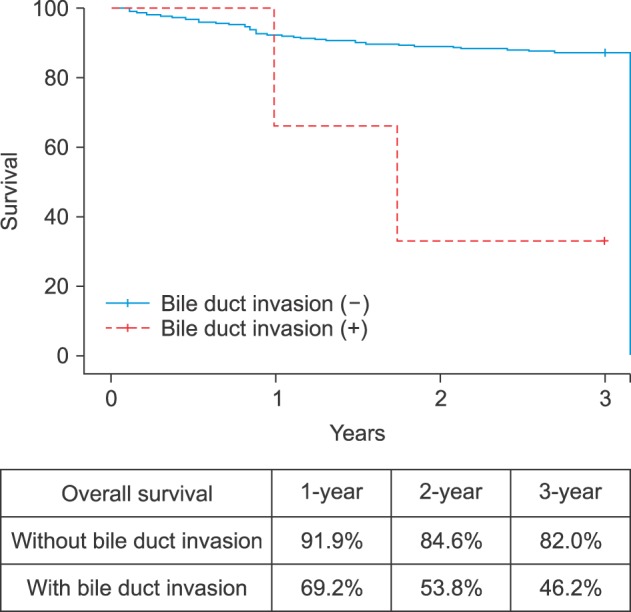
Fig. 2
Correlation between overall survival and bile duct invasion in advanced stage HCC patients (T-stage 3 and 4).




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download