DISCUSSION
Unlike pancreatic adenocarcinoma, the cystic neoplasms of pancreas are known to show relatively good prognosis even though it is diagnosed with carcinoma. In addition, it is difficult to establish a uniform rule for clinical course, differentiation and treatment because most tumors with the exception of serous cystic tumors show a variety of pathologic findings such as pre-cancer and cancer in benign tumors.
1
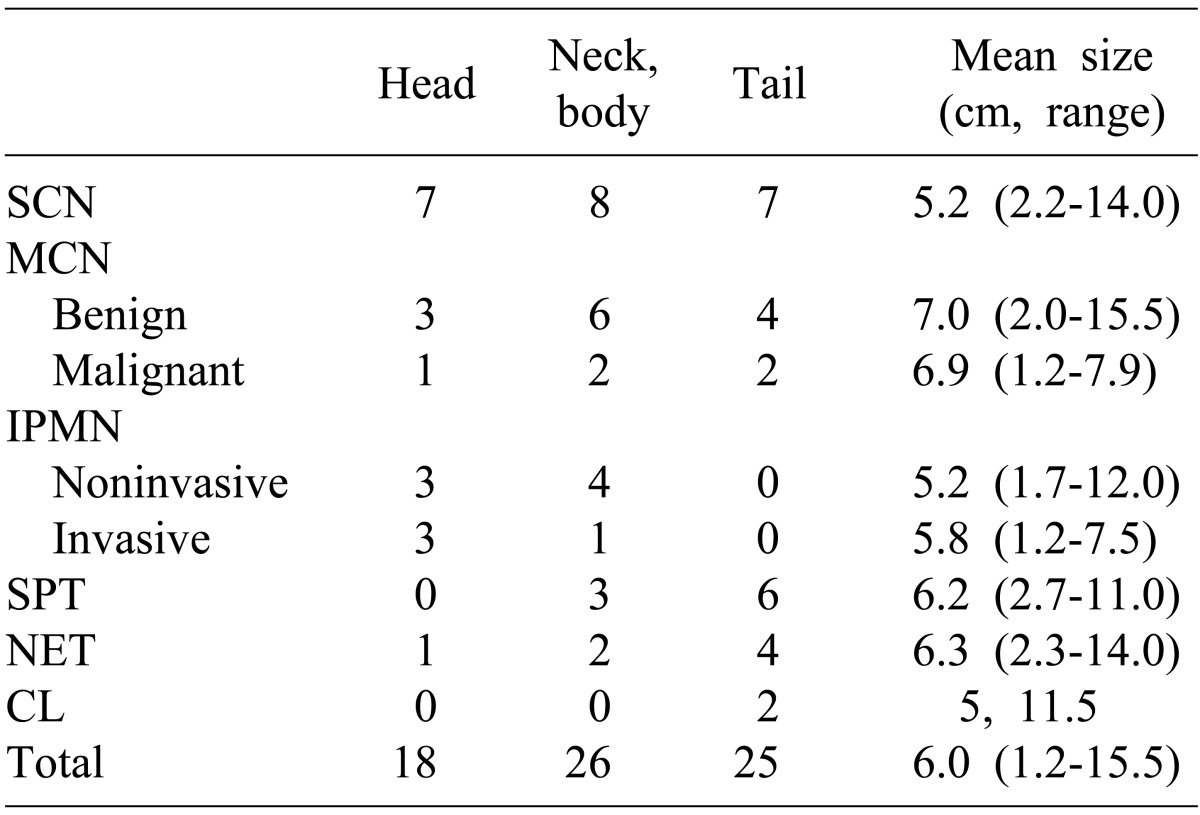
Serous cystic neoplasm is most common among cystic neoplasms of pancreas, accounting for 40% of frequency and being discovered accidentally. Approximately 20 cases of malignant cancer were reported around the world, but most tumors are considered to be benign in clinical studies.
2-
4 A large number of reports on the surgical results of serous cystic tumors claimed that the incidence of malignant cancer is extremely rare, but such problems are rather the inability of differential diagnosis from different precancerous lesions.
2 Galanis et al.
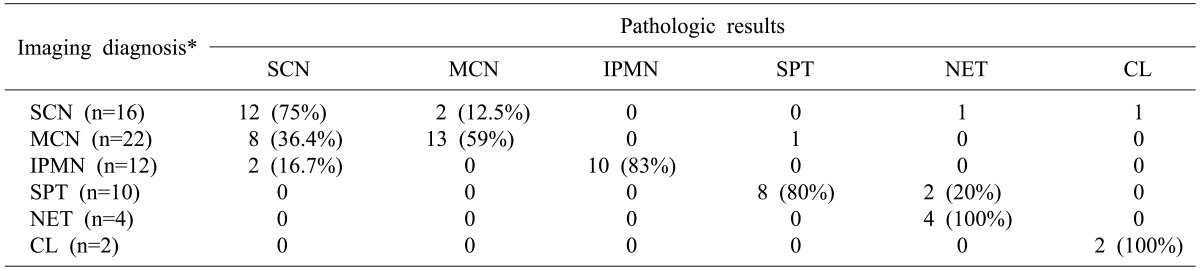
3 found 2 patients (1.3%) with the malignant tumors after 158 cases of serous cystic tumor resection who showed very good prognosis after resection due to the focal invasions. The authors found no malignant tumors after surgery for 22 patients with serous cystic neoplasms. Most patients undertook surgeries because differential diagnosis was not apparent with other premalignant diseases such as mucinous cystic neoplasms. Seven patients who were found with cystic neoplasms of pancreas by chance during health screening, all underwent surgery because they failed to completely rule out the possibilities of other pre-cancerous lesions. Twelve (75%) out of 16 patients were diagnosed with serous cystic tumor as a top priority in preoperative radiologic examinations and had the same pathologic biopsy results. The others were diagnosed with mucinous cystic neoplsms (2), with neuroendocrine tumors (1), and with cystic lymphangioma (1), respectively. Thus, surgical resections may be recommended as far as possible if it was difficult to distinguish from serous cystic tumors in the preoperative diagnosis. It was reported that the radical resection accounting for the location of tumor is safer and has less chances of complications rather than enucleation of cystic neoplasm.
4 All patients who were confirmed with serous cystic tumors received radical resections at our hospital.
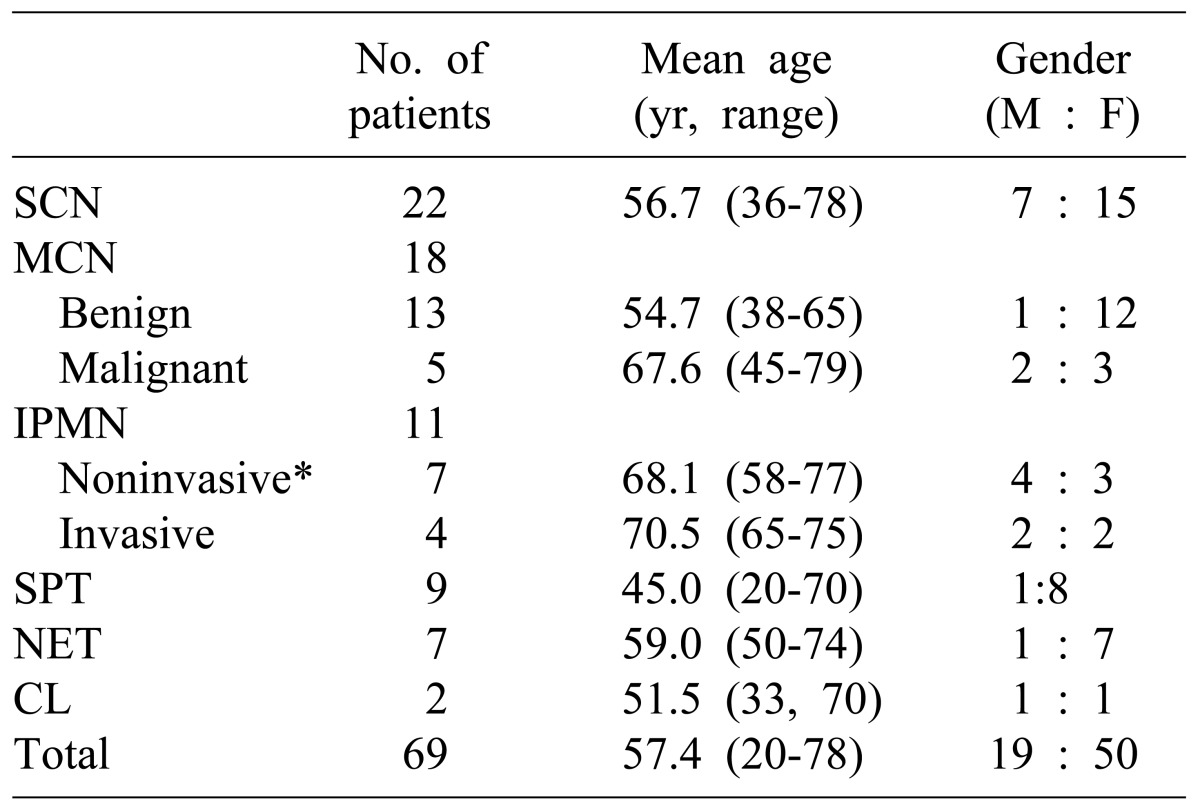
Mucinous cystic neoplasm is considered to be a precancerous lesion accounting for 30-40% of the entire cystic neoplasm. It commonly indicated malignant transformations only in some of the tumors and has a variety of distributions for benign tumor, borderline tumor, carcinoma in situ and invasive carcinoma.
5,
6 Thus, a complete tumor resection should be done in accordance with the malignant cancer; appropriate surgical treatment is required because it is known to have a better prognosis as compared with pancreatic adenocarcinoma even if it was diagnosed with a malignant cancer. The patient's ages and tumor sizes have been proposed as preoperative predictors of mucinous cystic neoplasms with malignancy.
5,
7 In our study, the average age (67.6 years) of patients diagnosed with malignant tumors was significantly higher than that of patients (54.7 years) with benign mucinous cystic neoplasms (
p=0.013). The size of benign and malignant tumors was not significantly different. Out of total 114 patients who underwent surgery for cystic diseases of the pancreas, 11 cases have been observed with infiltrations or adhesions with surrounding tissues in the preoperative radiologic examination, and thus, are suspected of malignant mucinous cystic neoplasms. Of these, only two were diagnosed with malignancy in the final pathologic diagnosis; six were diagnosed with pancreatitis and pancreatic pseudocyst, and two, with solid pseudopapillary tumor. Thus, it turned out to be significantly difficult to judge the presence of malignant behaviors in mucinous cystic neoplasms or to distinguish it from other diseases. Out of 69 patients who underwent surgery for cystic neoplasms of pancreas, 29 patients (42.0%) had mucinous cystic neoplasms or intraductal papillary mucinous tumors, both of which are the mucin secreting cystic neoplasms of pancreas considered as pre-cancerous lesions. Twenty two patients were diagnosed with mucinous cystic neoplasms as priority during preoperative radiologic examinations, but only 13 patients (59%) were diagnosed as the same in the pathologic findings. 8 patients (36.4%) were diagnosed with serous cystic neoplasms and one case was diagnosed with solid pseudopapillary tumor, it was not easy to distinguish between serous cystic neoplasms and mucinous cystic neoplasms with diagnostic imaging tests alone.
Inadequate biopsy was often presented as the recurrence factor after surgical resections of mucinous cystadenocarcinoma.
7,
8 Even though the resection tissues were diagnosed as benign or borderline tumors in the past, if recurrences occurred later, it turned out to be insufficient diagnosis for malignant cells of the past biopsy.
8 It was considered important that the patient's prognosis and tumor recurrence took detailed pathologic examinations and keeping it in mind that mucinous cystic neoplasms could have a variety of aspects of tissue cells. The authors diagnosed only benign and malignant tumors but did not strictly investigate the borderline tumors. However, considering that tumor recurrences were not found in patients diagnosed with benign tumors, the prognosis of borderline tumors included herein are expected to be very good. Therefore, the importance of early diagnosis and aggressive surgery are being emphasized once again before proceeding to malignant tumors.
Intraductal papillary mucinous tumor is a disease that gradually increases in numbers among cystic neoplasms of pancreas. It is often difficult to pathologically differentiate from mucinous cystic neoplasms, which is similar in that opinions vary from benign tumors to carcinoma in situ or invasive cancer. Especially when the main pancreatic duct types are known, and that surgery is needed in most cases pursuant to pancreatic adenocarcinoma because malignant cancers are more commonly found in this type.
9,
10 The likelihood of malignancy is higher and requires more attention, for cases such as obstructive jaundice, main pancreatic duct type and hybrid type, where the main pancreatic duct significantly expands more than 10-15 mm, intramural nodules are larger than 10 mm, duct wall irregularities, or tumors are larger than 3 cm.
11,
12 Jang et al.
13 reported that careful diagnosis and treatment protocols were required because the incidences of malignant tumors are more than 25% if the tumor size is larger than 2 mm, and there exists the intramural nodule even if it is the branch duct type. In our study, patients diagnosed with invasive intraductal papillary mucinous tumors did not show statistically significant differences from benign tumors of patients' ages and tumor sizes. In the preoperative radiologic diagnosis, 12 cases were diagnosed with intraductal papillary mucinous tumors as top priority, 4 of which were main pancreatic duct types; 2 hybrid types; and 6 branch duct types. Of the four patients who were diagnosed with invasive tumors, 3 patients had the main pancreatic duct type. The 3 patients who were diagnosed with main pancreatic duct type were also diagnosed with invasive tumors in preoperative imaging tests before surgery because of characteristic findings such as the main pancreatic duct dilatation, irregularities and intramural nodules. The location of the tumor in the pancreatic duct seems to be a relatively important factor to predict the malignant potential of the intraductal papillary mucinous tumor, but further studies will be needed for a larger number of patients.
Cystic neoplasm of the pancreas has limitations in diagnosis and differential diagnosis only by the abdominal computed tomography (CT), thus, the fine needle aspirations of the cyst fluid, endoscopic ultrasound and magnetic resonance imaging (MRI) are performed in the course of preoperative diagnosis.
14 Especially, it is reported to increase the diagnostic accuracy as to analyze the cystic fluid amylase, lipase and the tumor factors such as CEA, CA19-9 and CA125.
15,
16 The differentiation of potentially malignant cystic neoplasm before surgery plays an important role to define the treatment plan.
17 It is not recommended to excise a part of the cyst wall in the operating room and try frozen section examination
1 because it may degrade the diagnostic accuracy and cause disseminations of cyst fluid and potential malignant cells. Fine needle aspiration through the endoscopic ultrasound is also an invasive test and may cause the risk of tumor seeding or complications such as bleeding and perforation. However, we did not experience particular complications from preoperative fine needle aspirations and troubles during the surgery. Since the introduction of test equipments, 6 patients were checked by fine needle aspirations and the mucinous cystic neoplasm was predicted when the CEA level of the cystic fluid is over 400 ng/ml or CA19-9 is greater than 40 U/ml. The number of patients was limited, but all four patients whose CEA levels were higher than 400 ng/ml were diagnosed as mucinous cystic neoplasms and showed the specificity of 100%. Thus, it is suggested that to perform both endoscopic ultrasounds and fine needle aspirations for all patients in the future to increase the accuracy of the diagnosis. Researches would be needed for a larger number of patients, but by considering the experiences to date, it is often judged that the serous cystic neoplasm is the primary suspect when the CEA and CA19-9 of the fluids are in the normal range. However, there are still limitations for accurately diagnosing mucinous cystic neoplasms even if these diagnoses are used. We experienced that two patients who had relatively high CEA and CA19-9 of the fluid were diagnosed with pancreatic pseudocyst and cystic lymphangioma, respectively. It is recommended on one hand to use a combination of various diagnostic methods for the preoperative diagnosis in order to reduce unnecessary surgery for patients with observable progress, and on the other hand, to prevent the delays in surgery.
17
Despite some controversies, most studies recommended surgical resection in principle for the management of cystic neoplasms of pancreas. The biggest reason is said to be the difficulty of accurate diagnosis on the preoperative tumor pathological form and the difficulty to identify the presence of malignancy.
18,
19 It is also said that the development of surgical techniques has significantly lowered the mortality and complications of pancreatic resections. In recent years, the increasing application of laparoscopic pancreatic surgery has made surgical treatments more common. Nevertheless, it is important that relatively good prognosis is noted if tumors are operated early and completely removed despite the diagnosis of malignancy. It is also known that mucinous cystic tumor or intraductal papillary mucinous tumor which includes borderline tumors or carcinoma in situ has high, long-term survival rates if it is being completely resected. Even the malignant cancer was reported to have long-term survival rates around 50-76% after radical resections.
19,
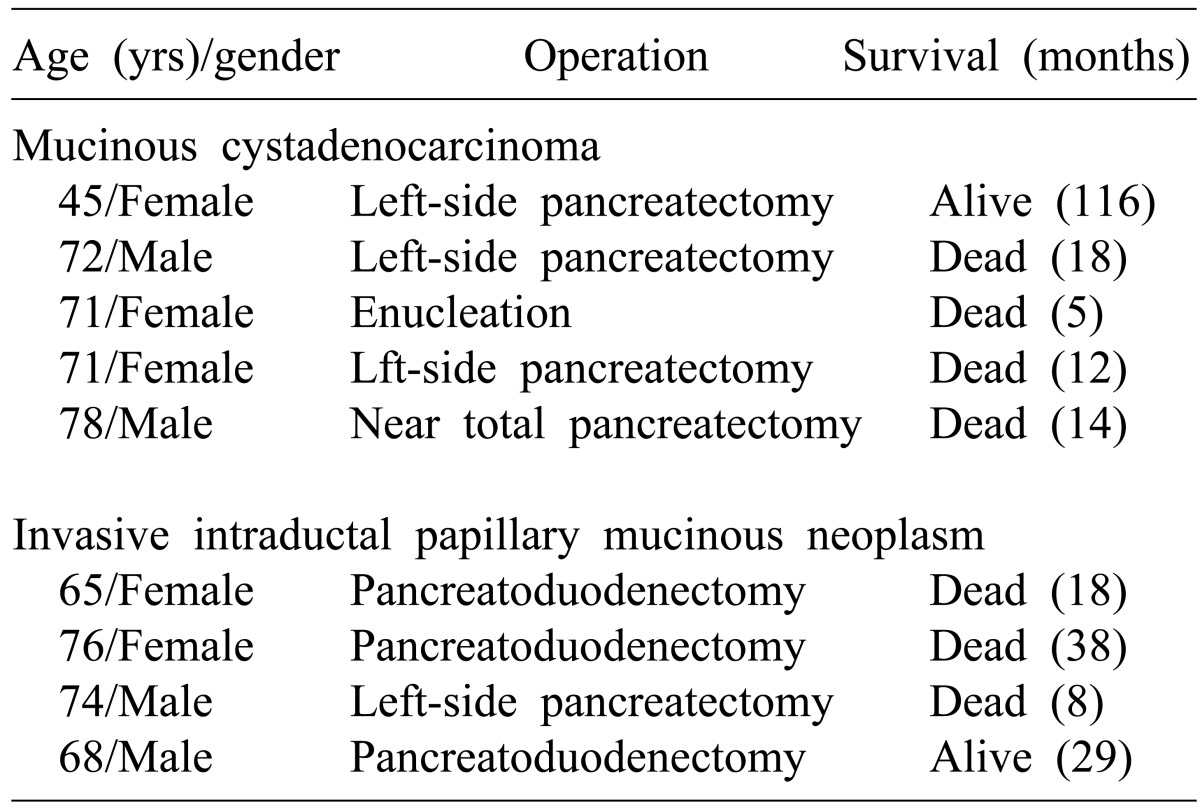
20 In case of the authors, malignant cancers were found in about 30% of the 29 patients with mucin secreting cystic tumors, including 5 patients (27.8%) of mucinous cystic tumors and 4 patients (36.4%) of intraductal papillary mucinous tumors. Long-term survival is not available due to the small number of patients, but they showed relatively poor prognosis after surgical resections. The reason is mostly because that most patients with infiltrative status had delays in diagnosing. Among the five patients with mucinous cystadenocarcinoma, four patients had left adrenal gland resections and or bowel resections due to the invasion. The patient who died of early recurrences 5 months after surgery was the case who only had enucleation as the mucinous cystic tumor was not suspected in the preoperative diagnosis. Four patients with invasive intraductal papillary mucinous tumors were characterized by distributions mainly in head of the pancreas (n=3), thus, they had pancreatoduodenectomy and one had left-side subtotal pancreatectomy. One of the patients with double primary cancer was diagnosed with bladder cancer at the same time. Mucinous cystadenocarcinoma showed the median survival of 14 months (5-116 months); invasive intraductal papillary mucinous neoplasm (invasive IPMN), 24 months (8-38 months), respectively. Thus, more thorough follow-ups and observations are required and aggressive surgical resections should be recommended if the cystic neoplasms of pancreas are found and cancer risk factors have been suspected. Lee et al.
19 argued that the possibility of malignancy should be considered in cases such as the presence of solid parts, expansion of main pancreatic duct, expansion of common bile duct, and enlargement of lymph node in imaging studies. When male patients older than 70 years had occurred jaundice and weight loss, they needed careful evaluations, even if the sizes were relatively smaller than 3 cm.
Laparoscopic resections of pancreas seemed to be commonly applied recently. We experienced four cases, and all of which were the left pancreatic resection. Postoperative complications included one intra-abdominal bleeding, and one pseudocyst formation from pancreatic fluid leakage. We have limited experience, but increasing reports indicate that benefits of laparoscopic surgeries are certain, and that surgical complications are not that different as compared with conventional open surgeries.
21,
22 Therefore, large scale prospective studies would be needed for the future applications of laparoscopic surgeries to patients with malignant pancreatic body and tail tumors as well as benign tumors.
Go to :





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download