Abstract
O-linked β-N-acetylglucosamine modification is an important post-translational modification, emerging as a novel regulatory mechanism in various cellular events. Recently, several studies have shown that O-GlcNAcylation plays an essential role in human breast, lung, and colon cancers. With regard to skin cancers, the role of O-GlcNAcylation has yet to be elucidated. To investigate whether O-GlcNAcylation is linked to human skin tumor development, immunohistochemical analysis was performed to investigate the presence of O-GlcNAcylation in various skin tumors. We evaluated the levels of O-GlcNAcylation, O-GlcNAc transferase, and O-GlcNAcase in 29 benign tumors, 12 premalignant tumors, and 26 malignant tumors in skin. Compared to the benign tumors, premalignant and malignant tumors had increased patterns of O-GlcNAcylation. In addition, the O-GlcNAc transferase and O-GlcNAcase levels were higher in premalignant and malignant tumors than in benign tumors. Interestingly, O-GlcNAcase levels were significantly increased in premalignant tumors compared to benign and malignant tumors. These results suggest that O-GlcNAcylation of proteins may play an important role in the development of human skin tumors.
Go to : 
References
1. Torres CR, Hart GW. Topography and polypeptide distribution of terminal N-acetylglucosamine residues on the surfaces of intact lymphocytes. Evidence for O-linked GlcNAc. J Biol Chem. 1984; 259:3308–17.

2. Hart GW, Housley MP, Slawson C. Cycling of O-linked beta-N-acetylglucosamine on nucleocytoplasmic proteins. Nature. 2007; 446:1017–22.
3. Kreppel LK, Blomberg MA, Hart GW. Dynamic glycosylation of nuclear and cytosolic proteins. Cloning and characterization of a unique O-GlcNAc transferase with multiple tetratricopeptide repeats. J Biol Chem. 1997; 272:9308–15.
4. Gao Y, Wells L, Comer FI, Parker GJ, Hart GW. Dynamic O-glycosylation of nuclear and cytosolic proteins: cloning and characterization of a neutral, cytosolic β-N-acetylglu-cosaminidase from human brain. J Biol Chem. 2001; 276:9838–45.
5. Kang JG, Park SY, Ji S, Jang I, Park S, Kim HS, et al. O-GlcNAc protein modification in cancer cells increases in response to glucose deprivation through glycogen degradation. J Biol Chem. 2009; 284:34777–84.

6. Gu Y, Mi W, Ge Y, Liu H, Fan Q, Han C, et al. GlcNAcylation plays an essential role in breast cancer metastasis. Cancer Res. 2010; 70:6344–51.

7. Mi W, Gu Y, Han C, Liu H, Fan Q, Zhang X, et al. O-Glc-NAcylation is a novel regulator of lung and colon cancer malignancy. Biochim Biophys Acta. 2011; 1812:514–9.

8. Snow CM, Senior A, Gerace L. Monoclonal antibodies identify a group of nuclear pore complex glycoproteins. J Cell Biol. 1987; 104:1143–56.

9. Caldwell SA, Jackson SR, Shahriari KS, Lynch TP, Sethi G, Walker S, et al. Nutrient sensor O-GlcNAc transferase regulates breast cancer tumorigenesis through targeting of the oncogenic transcription factor FoxM1. Oncogene. 2010; 29:2831–42.

10. Lynch TP, Ferrer CM, Jackson SR, Shahriari KS, Vosseller K, Reginato MJ. Critical role of O-Linked β-N-acetylglu-cosamine transferase in prostate cancer invasion, angiogenesis, and metastasis. J Biol Chem. 2012; 287:11070–81.

11. Yehezkel G, Cohen L, Kliger A, Manor E, Khalaila I. O-linked β-N-acetylglucosaminylation (O-GlcNAcylation) in primary and metastatic colorectal cancer clones and effect of N-acetyl-β-D-glucosaminidase silencing on cell phenotype and transcriptome. J Biol Chem. 2012; 287:28755–69.

12. Zhu Q, Zhou L, Yang Z, Lai M, Xie H, Wu L, et al. O-Glc-NAcylation plays a role in tumor recurrence of hepatocellular carcinoma following liver transplantation. Med Oncol. 2012; 29:985–93.

13. Ma Z, Vocadlo DJ, Vosseller K. Hyper-O-GlcNAcylation is anti-apoptotic and maintains constitutive NF-κB activity in pancreatic cancer cells. J Biol Chem. 2013; 288:15121–30.

14. Vosseller K, Sakabe K, Wells L, Hart GW. Diverse regulation of protein function by O-GlcNAc: a nuclear and cytoplasmic carbohydrate post-translational modification. Curr Opin Chem Biol. 2002; 6:851–7.

15. Hanover JA, Krause MW, Love DC. Bittersweet memories: linking metabolism to epigenetics through O-GlcNAcylation. Nat Rev Mol Cell Biol. 2012; 13:312–21.
Go to : 
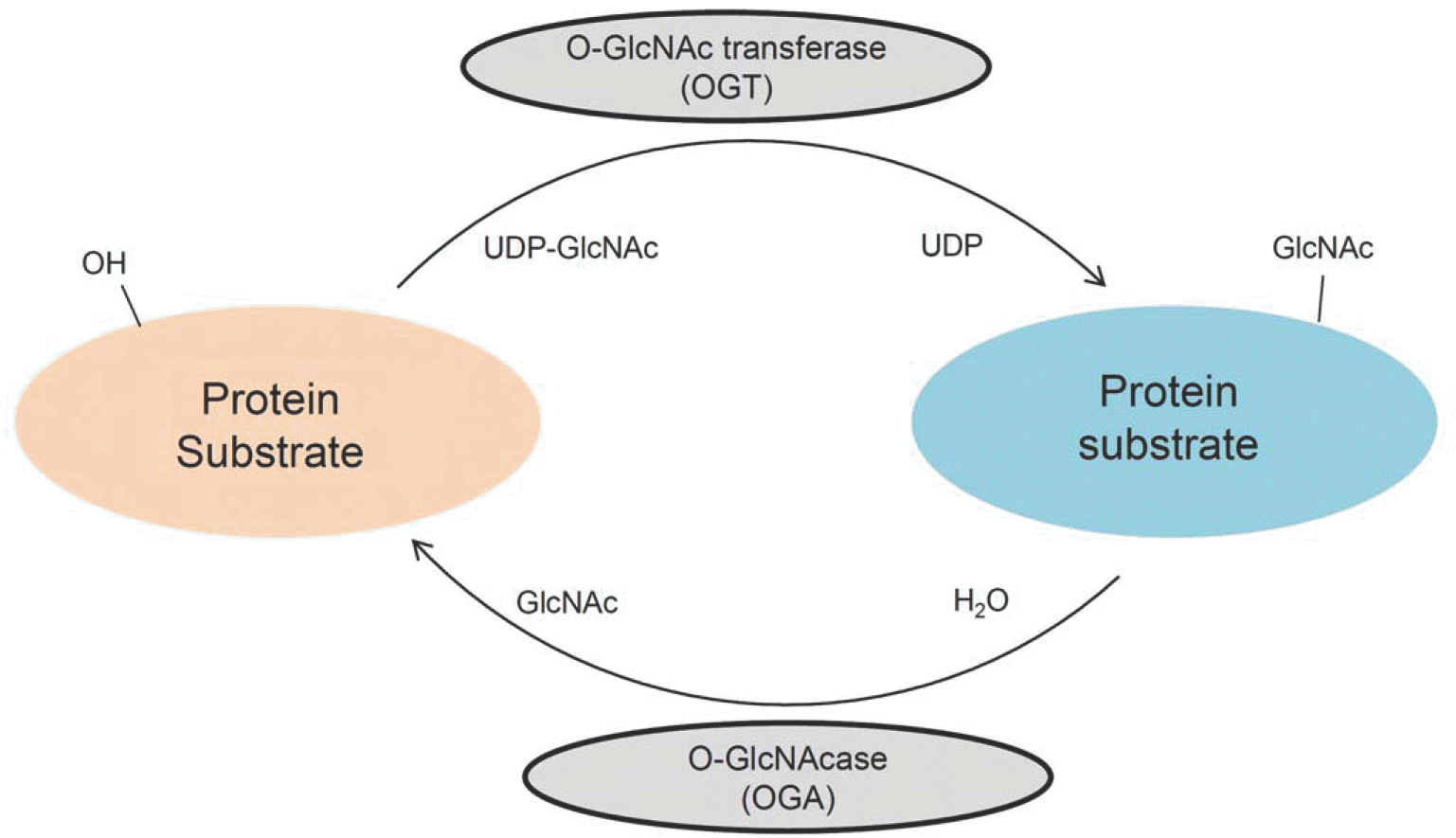 | Fig. 1.The O-GlcNAcylation cycle. O-GlcNAcylation is catalyzed by the enzyme O-GlcNAc transferase(OGT), which transfers GlcNAc from UDP-GlcNAc to the target protein. Conversely, O-GlcNAc is removed from the protein by O-GlcNAcase(OGA). |
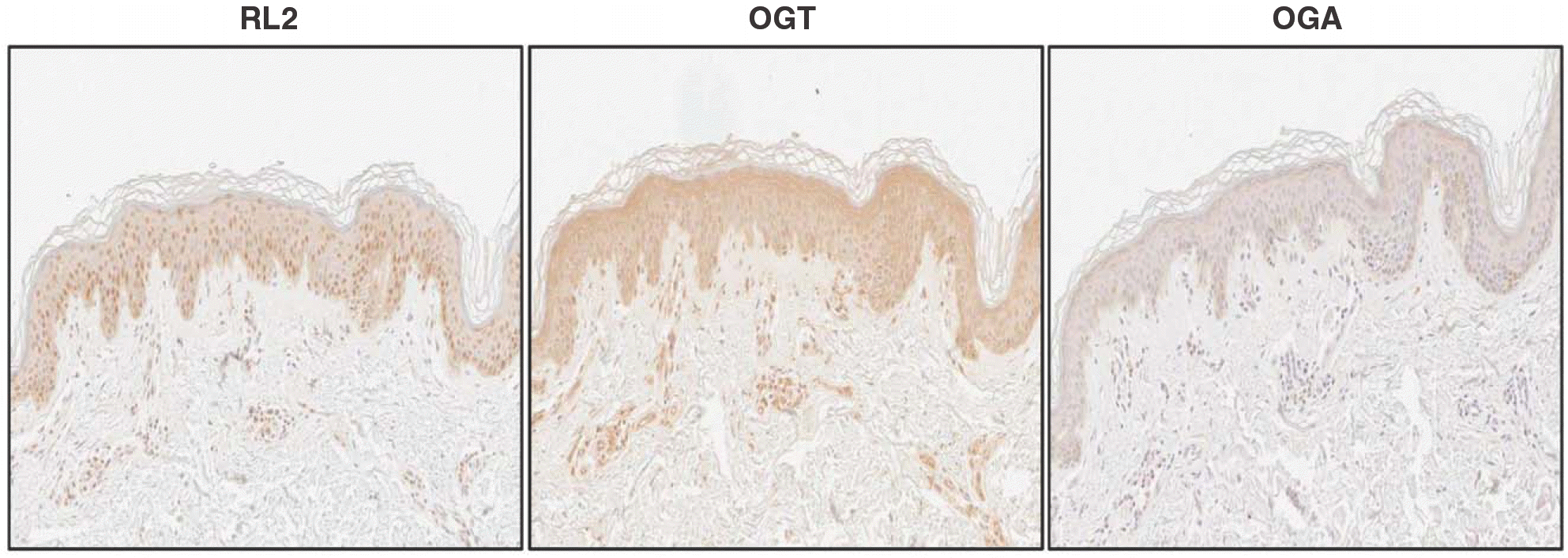 | Fig. 2.Immunohistochemical analysis of O-GlcNAcylation in the epidermis. Normal skin tissues were incubated with anti-O-GlcNAc antibody (RL2), anti-O-GlcNAc transferase (OGT) antibody, and anti-O-GlcNAcase (OGA) antibody. RL2-positive staining is slightly decreased in the upper layer. OGT is observed in all layers, and intense OGA staining is seen in the upper layer(200×). |
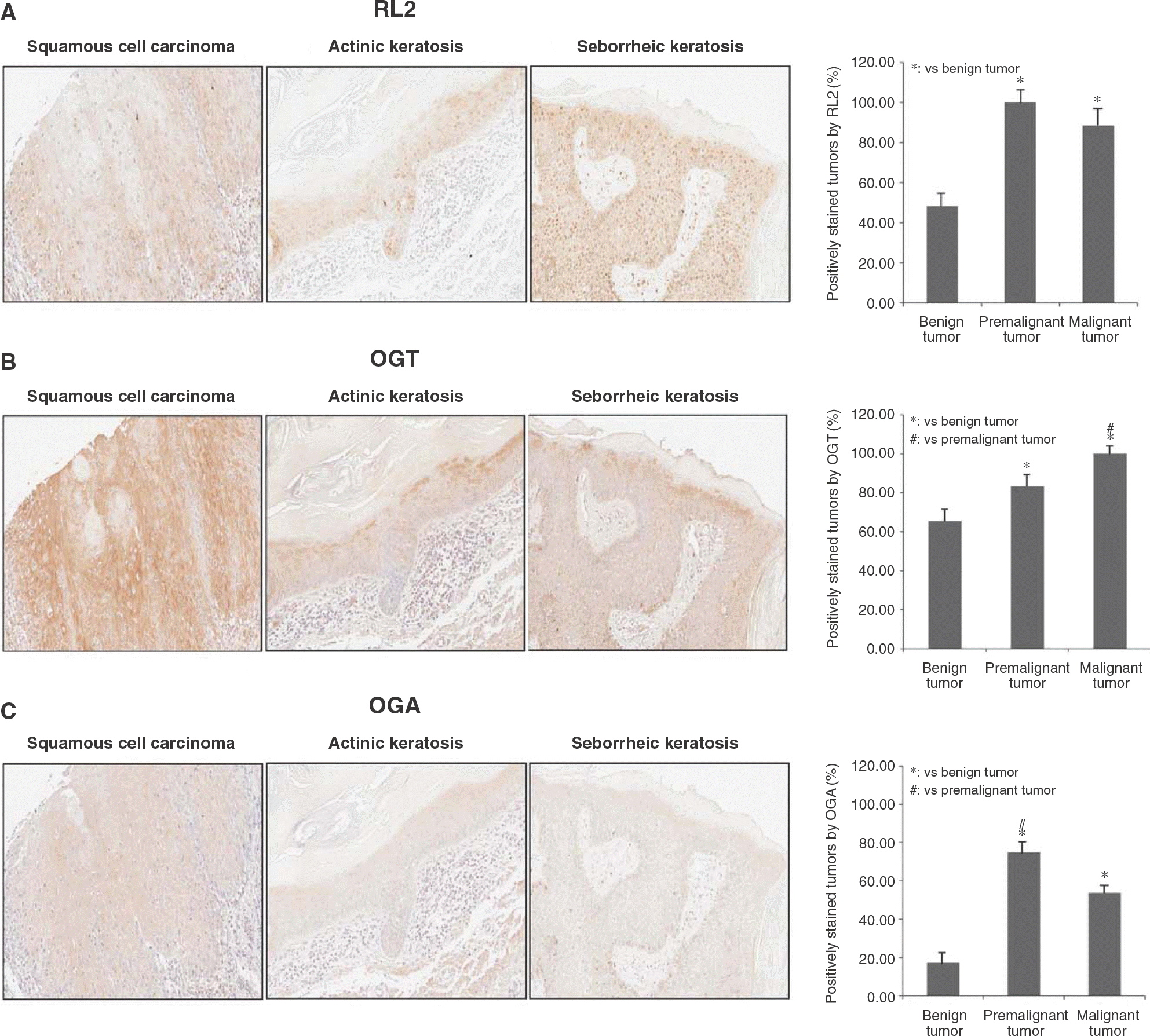 | Fig. 3.Immunohistochemical analysis of O-GlcNAcylation in skin tumors. Skin tumors were categorized as benign, premalignant, or malignant and stained with(A) anti-O-GlcNAc antibody(RL2)(B) anti-O-GlcNAc transferase(OGT) antibody, and(C) anti-O-GlcNAcase (OGA) antibody. The upper panel represents a benign tumor (neurofibroma), the middle panel represents a premalignant tumor (actinic triplicate and data are expressed as the mean±standard deviation(p⁄0.05). keratosis), and the lower panel represents a malignant tumor (squamous cell carcinoma) (200×). Measurements were conducted in triplicate and data are expressed as the mean±standard deviation (p<0.05). |
Table 1.
Summary of immunohistochemistry analysis




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download