Abstract
Ectopic breast tissue and male breast cancer are both very rare diseases with only a few reports in the literature. Here, we present the first case of ectopic male breast cancer in the perineum. The patient was a 70-year-old man with a palpable mass in the perineum. A wide local excision and inguinal lymph node dissection revealed invasive breast carcinoma of no special type involving the skin and subcutis, and inguinal lymph node metastases. Immunohistochemical staining showed that the tumor cells were strongly positive for estrogen and progesterone receptors and negative for human epidermal growth factor receptor 2. Moreover, no p53 overexpression was observed. Herein, the clinical and pathologic features, as well as a review of ectopic male breast cancer are discussed.
Accessory breast tissue, also known as ectopic breast tissue, is defined as residual breast tissue that persists from normal embryologic development as a result of failed resolution of the ectodermal mammary ridge, which extends from the axilla to the groin. Ectopic breast tissue is prevalent in the axilla and vulva. Pathologic degeneration including breast cancer could occur in accessory breast tissue. The incidence of ectopic breast cancer is 0.2% to 0.6% [1]. The most common site for ectopic breast cancer is the axilla and the most common histologic subtype is invasive ductal carcinoma [2]. Male breast cancer accounts for less than 1% of all breast cancers and ectopic breast cancer in men is extremely rare [3]. To the best of our knowledge, the perineal location of ectopic male breast cancer has not been reported to date. We report a male patient with an ectopic breast cancer in the perineum.
A 70-year-old Asian man with a nonsignificant medical history visited a local clinic with a complaint of a palpable, painless, and nonmovable perineal mass for several months. The overlying skin was slightly erythematous. He denied any recent history of unintended weight loss, fever, nausea, melena, or hematochezia. In February 2016, an excisional biopsy was performed in the local clinic. The pathology results showed metastatic adenocarcinoma with positive margins. Further workup for carcinoma of unknown origin included a thorough physical examination, basic blood tests, esophagogastroduodenoscopy, colonoscopy, contrast-enhanced computed tomography (CT) of the chest, abdomen, and pelvis, and positron emission tomography-CT (PET-CT). On physical examination, painless palpable movable masses were detected in the bilateral inguinal area, which were presumed to be enlarged lymph nodes (LNs). The CT scan showed an enlarged necrotic LN in the left inguinal area with a maximum standardized uptake value of 11.7 on PET-CT, in addition to several small LNs in the bilateral inguinal area (Figure 1).
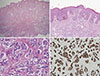
The patient was transferred to Asan Medical Center and the surgical specimen was re-examined. Final histopathologic examination of the excised skin and subcutaneous tissue revealed a tumor with the typical appearance of invasive breast carcinoma of no special specific type without evidence of nonneoplastic breast tissue (Figure 2). The invasive component measured 1.7 cm in the greatest dimension. The tumor involved the dermis, subcutis, and skeletal muscle, and lymphovascular and perineural invasion was also present. The overlying skin showed mild acanthosis. The tumor cells were strongly positive for estrogen receptor (1:100; NCL-L-ER-6F11; Novocastra, Newcastle, UK) and progesterone receptor (1:200; NCL-L-PGR-312; Novocastra), and negative for human epidermal growth factor receptor 2 (1:250; A0485; Dako Glostrub, Denmark). The tumor cells were focally positive for gross cystic disease fluid protein 15 (1:50; MS1170A, NeoMarkers, Fremont, USA), and negative for prostate-specific antigen (1:200; M0750; Dako) staining, which reduced the possibility of metastasis from the prostate. Immunohistochemical staining did not show p53 overexpression (1:1,500; M7001; Dako). The Ki-67 labeling index (1:200; M7240; Dako) was approximately 10%. Since the patient did not have primary breast cancer, we assumed that this tumor had arisen from ectopic breast tissue in the perineum, and the invasive carcinoma had replaced the entire nonneoplastic ectopic breast tissue.
The patient underwent local wide excision in the perineum and dissection of bilateral inguinal LNs at our hospital. Microscopic examination revealed multifocal residual invasive carcinomas measuring up to 0.4 cm in the greatest dimension, and two metastatic left inguinal LNs. Postoperative adjuvant chemotherapy and hormonal therapy were performed. After 8 months and at the time of writing, the patient had showed no evidence of recurrent disease. The study was approved by the Institutional Review Board of Asan Medical Center (IRB No. 2016-1251) and performed in accordance with the principles of the Declaration of Helsinki. The informed consent was waived.
The incidence of accessory breast tissue is 2% to 6% in the general population and is more frequent in women [4]. The most common location of ectopic breast tissue is the axilla, with different frequencies reported for other locations along the milk line, including parasternal, subclavicular, submammary, and vulvar sites [5]. There are two forms of ectopic breast tissue, supernumerary breasts and aberrant breast tissue. Supernumerary breasts, which are characterized by a ductal system communicating with the overlying skin, are usually found along the mammary ridge, an embryologic landmark consisting of an ectodermal thickening extending from the anterior axillary folds to the inguinal region and medial thigh [26]. Aberrant breast tissue consists of fragments of glandular tissue located outside the periphery of orthotopic breasts and lacks an organized secretory system [67].
Ectopic breast tissue may present with characteristics analogous to those of orthotopic tissue in terms of function and pathologic degeneration [6]. However, this remains controversial as several authors have proposed that aberrant breast tissue is more prone to malignant transformation than orthotopic breast tissue, possibly because of stagnation in the ductal lumen, which is a risk factor for the development of carcinoma [789]. The most common site of ectopic breast cancer is the axilla, accounting for 91% of ectopic breast cancers, followed by the vulva [10]. Since the first description by Hartung in 1872, 42 cases of ectopic breast tissue in the vulva have been reported with the first ectopic breast cancer in the vulva reported by Greene in 1935 [11]. The most frequently reported histologic subtype, as in orthotopic breasts, is invasive ductal carcinoma [1].
Male breast cancer is a rare malignancy, accounting for less than 1% of all malignancies in men [3]. Because of its rarity, male breast cancer, unlike female breast cancer, has not been studied extensively, and management guidelines have not yet been established. Survival rates for male breast cancer are equal to or lower than those for female breast cancer [12]. Ectopic breast cancer is extremely rare in men. Case reports on ectopic male breast cancer include invasive ductal carcinoma in the accessory mammary glands of the axilla and chest wall [13]. We assume that the perineal location of the tumor in our case corresponds to the vulva of females. Invasive ductal carcinoma accounts for 90% of male breast cancers, and the rate of positivity for hormone receptors is 75% to 85% [3], which is the same as that of the present case report.
Treatment options for male breast cancer, including ectopic male breast cancer, are the same as those for primary breast cancer in women. However, adjuvant chemotherapy, radiation treatment, and antihormonal therapy protocols are diverse given the rarity of these lesions and the absence of standardized treatment protocols [1415]. In previous reports, women with ectopic breast cancer in the vulva have been treated with wide local excision and inguinal LN dissection, and have been all characterized by LN involvement [15]. Sentinel node mapping in primary vulvar cancer and penile cancer has been evolved; ectopic breast cancer in the perineum could be similar because of the parallel lymphatic drainage in penile carcinoma. In the present case, we did not perform sentinel node mapping because the LNs were clinically palpable. Because of the paucity of ectopic breast cancer and the limited follow-up data, the prognosis of this disease remains unclear. Marshall et al. [7] reported that the poor outcomes in ectopic breast cancer relative to general breast cancer reflected less effective clinical management, and particularly, delayed treatment, rather than the disease process itself.
In conclusion, we report the first case of ectopic breast cancer in the perineum in a male patient. Due to its rarity, diagnosis of ectopic cancers could be delayed. To make a correct diagnosis, we should examine the patient carefully using imaging and pathological testing. Surgical resection with LN sampling is mandated followed by chemotherapy or targeted therapy according to immunohistochemical staining results.
Figures and Tables
Figure 1
Computed tomography (CT) and positron emission tomography-CT (PET-CT) images of the inguinal lymph nodes. (A) An enlarged necrotic lymph node is in the left inguinal area. (B) On PET-CT, the lymph node shows hypermetabolism which was diagnosed as a metastatic lymph node after surgical dissection.

Figure 2
Microscopic findings of the surgical specimen in the perineum. (A) Tumor cells are involving dermis, subcutis, and skeletal muscle (×12.5, H&E stain). (B) The overlying skin shows mild acanthosis, and basal pigmentation is increased (×100, H&E stain). (C) High magnification shows tumor cells arranged in nests with lumen formation in the fibrotic stroma and mild pleomorphism, consistent with invasive breast carcinoma of no special type (×400, H&E stain). (D) Tumor cells are strongly immunoreactive for estrogen receptor (ER), which also suggests the diagnosis of breast carcinoma (×200, ER immunohistochemical staining).
References
1. Nihon-Yanagi Y, Ueda T, Kameda N, Okazumi S. A case of ectopic breast cancer with a literature review. Surg Oncol. 2011; 20:35–42.

2. Hong JH, Oh MJ, Hur JY, Lee JK. Accessory breast tissue presenting as a vulvar mass in an adolescent girl. Arch Gynecol Obstet. 2009; 280:317–320.

3. Giordano SH, Cohen DS, Buzdar AU, Perkins G, Hortobagyi GN. Breast carcinoma in men: a population-based study. Cancer. 2004; 101:51–57.
4. Gutermuth J, Audring H, Voit C, Haas N. Primary carcinoma of ectopic axillary breast tissue. J Eur Acad Dermatol Venereol. 2006; 20:217–221.

5. Wysokinska EM, Keeney G. Breast cancer occurring in the chest wall: rare presentation of ectopic milk line breast cancer. J Clin Oncol. 2014; 32:e35–e36.

6. Francone E, Nathan MJ, Murelli F, Bruno MS, Traverso E, Friedman D. Ectopic breast cancer: case report and review of the literature. Aesthetic Plast Surg. 2013; 37:746–749.

7. Marshall MB, Moynihan JJ, Frost A, Evans SR. Ectopic breast cancer: case report and literature review. Surg Oncol. 1994; 3:295–304.

8. Ghosn SH, Khatri KA, Bhawan J. Bilateral aberrant axillary breast tissue mimicking lipomas: report of a case and review of the literature. J Cutan Pathol. 2007; 34:Suppl 1. 9–13.

9. Cogswell HD, Czerny EW. Carcinoma of aberrant breast of the axilla. Am Surg. 1961; 27:388–390.
10. Pardo M, Silva F, Jiménez P, Karmelic M. Mammary carcinoma in ectopic breast tissue: a case report. Rev Med Chil. 2001; 129:663–665.
11. Greene HJ. Adenocarcinoma of supernumerary breasts of the labia majora in a case of epidermoid carcinoma of the vulva. Am J Obstet Gynecol. 1936; 31:660–663.

12. Gnerlich JL, Deshpande AD, Jeffe DB, Seelam S, Kimbuende E, Margenthaler JA. Poorer survival outcomes for male breast cancer compared with female breast cancer may be attributable to in-stage migration. Ann Surg Oncol. 2011; 18:1837–1844.

13. Yamamura J, Masuda N, Kodama Y, Yasojima H, Mizutani M, Kuriyama K, et al. Male breast cancer originating in an accessory mammary gland in the axilla: a case report. Case Rep Med. 2012; 2012:286210.

14. Markopoulos C, Kouskos E, Kontzoglou K, Gogas G, Kyriakou V, Gogas J. Breast cancer in ectopic breast tissue. Eur J Gynaecol Oncol. 2001; 22:157–159.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download