Abstract
Purpose
The aim of this study is to present the incidence of radiation pneumonitis (RP) reported within 6 months after treatment for breast cancer with or without internal mammary node irradiation (IMNI).
Methods
In the Korean Radiation Oncology Group (KROG) 08-06 phase III randomized trial, patients who were node-positive after surgery were randomly assigned to receive radiotherapy either with or without IMNI. A total of 747 patients were enrolled, and three-dimensional treatment planning with computed tomography simulation was performed for all patients. Of the 747 patients, 722 underwent chest X-rays before and within 6 months after radiotherapy. These 722 patients underwent evaluation, and RP was diagnosed on the basis of chest radiography findings and clinical symptoms. The relationship between the incidence of RP and clinical/dosimetric parameters was analyzed.
Results
RP developed in 35 patients (4.8%), including grade 1 RP in 26 patients (3.6%), grade 2 RP in nine patients (1.2%); there was no incidence of grade 3 or higher RP. Grade 2 RP cases were observed in only the IMNI group. The risk of developing RP was influenced by IMNI treatment; pneumonitis occurred in 6.5% of patients (n=23/356) who underwent IMNI and in 3.3% of patients (n=12/366) who did not (p=0.047). The differences in lung dosimetric parameters (mean lung dose, V10–40) were statistically significant between the two groups.
In 2008, the Korean Radiation Oncology Group (KROG) initiated a prospective phase III randomized trial (KROG 08-06) designed to investigate the effect of internal mammary node irradiation (IMNI) on disease-free survival (DFS) and toxicity in breast cancer patients. Until that time, no consensus existed regarding the use of IMNI in postmastectomy radiotherapy or radiotherapy after breast-conserving surgery (BCS). Inclusion of the internal mammary nodes (IMNs) in breast cancer radiotherapy is mainly dependent on the preferences of the treating radiation oncologists. A Korean patternof-care study showed that approximately 50% of patients received IMNI during postmastectomy radiotherapy [1]. Variable patterns of clinical practice regarding IMNI, which were culture-driven not evidence-based, have been reported [23].
Because IMNI may increase radiation exposure to critical organs, new studies must determine whether the expected benefits of elective IMNI is worth the risk of late toxicity to critical organs, such as the lungs and heart [4]. In the European Organisation for Research and Treatment of Cancer (EORTC) 22922/10925 trial, instances of pulmonary toxicity were significantly higher in the IM-MS (internal mammary and medial supraclavicular chain) treatment group than in the control group [5]. The National Cancer Institute of Canada Clinical Trial Group MA.20 trial demonstrated improved DFS in patients with one to three positive nodes with the addition of regional nodal irradiation, including IMNI. However, this additional irradiation was associated with an increase in the incidence of grade 2 or higher pneumonitis (1.3% vs. 0.2%) [6].
Upon its completion in February 2013, KROG 08-06 had enrolled a total of 747 patients. In this study, we reviewed collected data from the evaluable 722 patients and present the incidence of radiation pneumonitis (RP) reported within 6 months after radiotherapy with or without IMNI.
Eligible patients were pathologically confirmed to have axillary node-positive breast cancer after surgery consisting of either modified radical mastectomy (MRM) or BCS, regardless of histologic type. All patients underwent axillary dissection, in which eight or more lymph nodes were identified. Patients were stratified according to N stage (N1 vs. N2 or N3) and type of surgery (breast conservation vs. mastectomy), and then were randomly assigned to receive radiotherapy either with or without IMNI (Figure 1). Three hundred and one patients were diagnosed with pathologic stage N1 disease, and 416 patients were diagnosed with N2 or N3 disease. Three hundred and sixty-one patients were treated with BCS, and the remaining 356 patients were treated with MRM. Among the patients with BCS, 178 were randomly assigned to the IMNI group, and 183 were randomly assigned to the non-IMNI group. Among the patients with MRM, 173 were randomly assigned to the IMNI group, and 183 patients were assigned to the non-IMNI group. All patients had unilateral invasive breast cancer and were eligible for adjuvant chemotherapy with or without hormonal therapy. Patients who received neoadjuvant systemic therapy or had a previous history of cancer or distant metastasis were excluded.
Between November 2008 And February 2013, We Enrolled 747 Patients From The 12 Participating Institutions In Korea. The Study Protocol Was Approved By The Institutional Review Board (Irb Approval Number: 4-2008-0263), And All Patients Provided Written Informed Consent. Twenty-five Patients (3.3%) Who Had Not Undergone Chest X-ray Within 6 Months Of Radiotherapy Completion Were Excluded From The Analysis, Leaving 722 Analyzable Patients. The Characteristics Of The Patients In This Study Are Presented In Table 1. The Median Patient Age Was 48 Years (Range, 28–77 Years) In Both Groups. The Majority Of The Patients Enrolled (99.6%) Had Eastern Cooperative Oncology Group Performance Scores Of 0 Or 1. The Non-imni Group Included A Significantly Higher Percentage Of Patients With Ductal Carcinoma And A Progesterone Receptor-negative Status Compared With The Imni Group (P=0.003 And P=0.030, Respectively). Other Patient Characteristics Were Not Significantly Different between the two groups.
Radiation was administered once per day at a dose of 1.8–2 Gy, up to a total dose of 45–50.4 Gy; additionally, 381 patients (52.8%) received boost radiotherapy to the primary tumor bed with a median dose of 10 Gy (range, 9–16 Gy) in five fractions. All patients received supraclavicular irradiation, as routinely performed for node-positive disease. Each patient underwent computed tomography (CT)-based simulation, and structures were manually contoured on CT scan slices. This protocol contained no strict guidelines on radiotherapy technique; the techniques were determined at the discretion of the physician, and included the reverse hockey stick, standard tangent, partial wide tangent, and photon/electron combination techniques. A detailed distribution of the patients according to radiotherapy technique is shown in Table 2. In the MRM-IMNI group, the partial wide tangent (n=84, 48.5%) was the most commonly used technique, followed by the reverse hockey stick method (n=51, 29.4%). In the BCS group, the partial wide tangent method (n=101, 58.7%) and the photon/electron combination method (n=71, 41.2%) were used for IMN irradiation. In the non-IMNI group, the most commonly used radiotherapy technique was the standard tangent method for both MRM patients (n=132, 72.1%) and BCS patients (n=182, 100%).
After completion of radiotherapy, follow-up examinations including chest X-rays and physical examinations were obtained within 6 months. Chest X-rays obtained at baseline (before radiotherapy) were compared with those obtained within 6 months after treatment to determine RP levels. RP-related symptoms, such as cough, dyspnea, and the incidence of steroid treatment, were also identified and recorded. RP grade was scored on a scale of 0–5, based on the Radiation Therapy Oncology Group/EORTC toxicity criteria as follows: grade 0=no change over baseline; 1=asymptomatic or mild symptoms (dry cough), slight radiographic appearances; 2=moderate symptomatic fibrosis or pneumonitis (severe cough), low-grade fever, patch radiographic appearances; 3=severe symptomatic fibrosis or pneumonitis, dense radiographic appearance; 4=severe respiratory insufficiency, continuous O2, assisted ventilation; 5=death. To avoid any interobserver variation between the 12 participating institutions, two radiation oncologists on-site visited and reviewed all abnormal chest X-ray findings and assessed them together.
To identify predictive factors associated with RP development, clinical variables and dosimetric parameters were analyzed via univariate analysis using the Pearson chi-square test. Dosimetric parameters such as mean lung dose (MLD), V10, V20, V30, and V40 were included in the analysis, and the correlation with RP was analyzed using the Student t-test. Additionally, the significance of the association between treatment assignment and patient characteristics was assessed using the chi-square test. Statistical analysis was carried out using SPSS version 18.0 (SPSS Inc., Chicago, USA). A p-value ≤0.05 was considered statistically significant.
The incidence of RP was higher in the IMNI group (Table 3). Of the 722 patients, RP developed in 35 patients (4.8%), including 26 patients (3.6%) with grade 1 RP and nine patients (1.2%) with grade 2 RP. No cases of grade 3 or higher RP were found. All grade 2 RP cases developed in the IMNI group. RP occurred in 6.5% (n=23/356) of patients who were treated with IMNI and 3.3% (n=12/366) of those who were treated without IMNI (p=0.047). However, most RP cases were asymptomatic minimal pulmonary radiologic changes defined as grade 1. Of the 26 patients with grade 1 RP, only six patients experienced mild dry cough, which improved spontaneously. In all grades of RP, most radiologic changes developed 2 to 3 months into the follow-up period after radiation treatment.
With respect to radiotherapy techniques, the patterns of RP incidence differed between the two study arms (Table 2). Among the 23 patients who developed RP in the IMNI group, 13 patients (n=13/185, 7.0%) developed RP after undergoing treatment with the partial wide tangent method, and seven patients (n=7/51, 13.7%) developed RP after the reverse hockey stick method. The others (n=3/109, 2.8%) developed RP after the photon/electron combination method. In the non-IMNI group, 12 patients developed RP, including nine patients (n=9/314, 2.9%) after the standard tangent method and three patients (n=3/50, 6.0%) after the reverse hockey stick method. The overall incidence of RP after the partial wide tangent method (13 patients) was similar to that after the reverse hockey stick method (10 patients), but the percentage of RP patients treated with each technique were 7.0% (n= 13/185) and 9.9% (n=10/101), respectively.
All dosimetric parameters were significantly different between treatment groups (Figure 2). The MLD was 17.66±5.33 Gy with IMNI and 13.29±4.37 Gy without IMNI (p<0.001). The V10 and V20 with IMNI were 45.68%±18.19% and 34.94%±12.15%, respectively, and 31.71%±12.87% and 25.49%±10.53, respectively, without IMNI. The V30 and V40 with IMNI were 27.48%±8.86% and 19.40%±6.92%, respectively, and 21.07%±8.81% and 14.65%±6.67%, respectively, without IMNI.
Univariate analysis of dosimetric parameters for predicting the development of RP showed that all dosimetric parameters were significantly different between the RP and non-RP groups (Figure 3A). In the RP group, the MLD was 17.88±5.75 Gy, and the V10, V20, V30, and V40 were 45.31%±15.69%, 36.20%±11.96%, 29.47%±9.73%, and 21.54%±8.67%, respectively. In the non-RP group, the MLD was 15.38±5.30 Gy, and the V10, V20, V30, and V40 were 38.38%±17.25%, 29.93%±12.26%, 24.02%±9.30%, and 16.80%±7.04%, respectively. However, dosimetric parameters were not significantly different between grade 1 and grade 2 RP patients (Figure 3B).
The use of IMNI has been debated, but several reports have provided evidence that it improves survival in patients with breast cancer [78]. A previous retrospective study from our institution reported a long-term DFS advantage following IMNI in postoperative patients [9]. Although more studies are needed to clearly define the role of IMNI in long-term survival and toxicity, the findings of this retrospective study showed that IMNI was obviously effective in patients with N2 disease and patients with inner/central tumors. However, there is no consensus regarding whether IMNs should be treated, because of the possible increase in the risk for late toxicity. As a result of the anatomic location of IMNs, IMNI increases the exposure of critical organs, such as the lungs and heart, to radiation.
In this study, we investigated lung toxicity, which may lead to deterioration of the patient's performance status or quality of life. We specifically examined RP occurring within 6 months of treatment and its association with IMNI. RP is a common type of toxicity caused by radiation exposure to the lung and usually appears within 6 months of the completion of radiotherapy. Clinical symptoms, including cough and low-grade fever, occur following completion of the radiotherapy course, and can also be seen as nonspecific infiltration on chest X-rays. The rate of pneumonitis may be also influenced by systemic therapy [1011]. Various techniques to irradiate the IMN while minimizing normal tissue irradiation have been suggested [12]. We previously reported that the partial wide tangent method is the best technique for patients undergoing BCS because of the IMN coverage involved with this method and because of the reduced dose to the lungs and heart. However, the photon/electron combination method showed better isodose distribution in some patients [13]. The developments in radiotherapy techniques and the availability of three-dimensional (3D) treatment planning have allowed us to more precisely and safely irradiate IMN. In our country, 3D treatment planning with CT simulation has been used since the mid-2000s; therefore, all patients enrolled in this study had undergone CT simulation and 3D treatment planning. In this study, various radiotherapy techniques were allowed at the discretion of the treating radiation oncologists. In the IMNI treatment group, the most commonly used technique was the partial wide tangent method for both MRM and BCS. The reverse hockey stick method was exclusively used for MRM cases, both in the IMNI and non-IMNI groups.
The incidence of symptomatic RP (grade ≥2) was reported as 2.3% after whole breast and supraclavicular lymph node treatment without IMNI, and 3% after breast irradiation using the partial wide tangent technique, which includes the first three IMNs, in single-institution studies [1415]. As a multi-institutional study, we showed that the incidence of RP, when patients were treated without IMNI, was 3.3% (n=12/366), with the BCS group accounting for 2.2% and the MRM group accounting for 4.3%. In addition, the incidence rates of grade 1 and 2 RP after using the partial wide tangent method were 4.9% (n=9/185) and 2.2% (n=4/185), respectively. In the MRM group, the risk of RP can be reduced by using the reverse hockey stick method, in which the anterior chest wall is irradiated with an electron beam using an individualized stepbolus. However, 13.7% of patients experienced RP, which may have resulted from improper administration of the step-bolus. The results of the current study are in line with previous reports on pulmonary toxicity associated with breast radiotherapy [51617]. Although the incidence of RP, including asymptomatic radiologic changes, was significantly increased with IMNI, the clinical impact was minimal. In the EORTC 22922/10925 trial, researchers found no significant difference between the deterioration of the performance status and increased lung toxicity [5]. Thus, we suggest that IMNI can be applied without any significantly increased risk.
The incidence of RP correlates with the irradiated lung volume and radiation dose. A previous study suggested that if the ipsilateral lung irradiation volume is less than 12%, then the risk of pneumonitis is minimal, even with coverage of the supraclavicular area [18]. In general, the MLD and V20 are related to RP, and the ipsilateral V20 can predict the risk of pulmonary toxicity [19]. In the current study, each patient underwent CT-based simulation; therefore, we obtained and analyzed the relationship between the dosimetric parameters and the incidence of RP with or without IMNI. MLD and V20 (17.66±5.33 Gy and 34.94%±12.15%, respectively) in the IMNI group were higher than in the non-IMNI group (13.29±4.37 Gy and 25.49%±10.53%, respectively). Other lung dosimetric parameters such as V10, V30, and V40 also exhibited statistically significant differences between the two groups. A previous single-institution study showed that the incidence of RP was higher in patients with MLD ≥20.5 Gy or a normal tissue complication probability ≥23% [15]. Through this large prospective trial, we confirmed that the incidence of RP, as evaluated using chest X-ray, increased with higher doses of radiation to the lung, which was associated with IMNI. However, clinically meaningful grade 2 RP was not predictable on the basis of dosimetric parameters. Other patient factors that increase the risk of RP can also be considered. It has been reported that RP is more likely to occur when certain chemotherapy drugs are administered along with radiation. However, because all the patients in our trial received chemotherapy, we could not evaluate the effect of chemotherapy.
One drawback of this study is that the chest X-ray follow-up visit could occur at any time within 6 months after RT. Considering that most radiologic changes in this study were found at 2 or 3 months after RT, the heterogeneity of the follow-up time among patients may have caused an underestimation of asymptomatic grade 1 RP. Furthermore, we did not assess the change in performance status between the enrollment and post-RT periods, which may have helped to evaluate the effect of IMNI on quality-of-life. In this study, we focused only on short-term RP incidence; we did not plan to investigate radiation-related cardiac disease, because late cardiac toxicity often appears 10 to 15 years after radiotherapy, meaning long-term follow-up is required [202122]. Nilsson et al. [23] reported that radiation to the supraclavicular lymph nodes and IMNs increased the risk of stroke. The EORTC trial 22922/10925 assessed the impact of elective internal mammary and medical supraclavicular lymph node irradiation on the well-known toxicities of breast cancer radiotherapy, including lung, skin, and heart toxicity [5]. In contrast, they found that increased lung toxicity with IMNI was the only statistically significant factor between the two treatment groups at 3 years.
With newer treatment techniques, such as the breath-hold technique, intensity-modulated RT, particle therapy, and volumetric-modulated arc therapy, IMNI can be delivered at even lower doses to the organ at risk, especially in left-sided breast cancer [242526]. Consequently, the incidence of RP and dose parameters with IMNI estimated in this study can be further decreased by including these newer methods.
In conclusion, results from this large data collection study clearly showed that treatment of IMNs resulted in increased radiation to the lungs and a higher rate of RP, but the incidence and severity of RP was minimal. Therefore, we suggest that IMNI is well tolerated with a very low risk of symptomatic RP; however, future analyses should assess whether this minor impact on morbidity could affect long-term survival outcomes.
Figures and Tables
 | Figure 1KROG-08-06 trial diagram.KROG=Korean Radiation Oncology Group; ALND=axillary lymph node dissection; BCS=breast-conserving surgery; MRM=modified radical mastectomy; IMNI=internal mammary node irradiation.
|
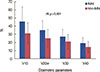 | Figure 2Dosimetric parameters of both internal mammary node irradiation (IMNI) and non-IMNI groups. All dosimetric parameters (V10–40) were significantly different between treatment groups. Data are the mean±SD. |
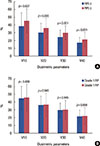 | Figure 3Dosimetric parameters in the patients with radiation pneumonitis. All dosimetric parameters were significantly different between the radiation pneumonitis (RP) and non-RP groups (A), not significantly different between grade 1 and grade 2 RP (B). Data are the mean±SD. |
Table 1
Patients and tumor characteristics

Table 2
Radiotherapy techniques used in each treatment group and incidence of radiation pneumonitis

Notes
References
1. Keum KC, Shim SJ, Lee IJ, Park W, Lee SW, Shin HS, et al. The 1998, 1999 patterns of care study for breast irradiation after mastectomy in Korea. J Korean Soc Ther Radiol Oncol. 2007; 25:7–15.
2. Chargari C, Castadot P, Macdermed D, Vandekerkhove C, Bourgois N, Van Houtte P, et al. Internal mammary lymph node irradiation contributes to heart dose in breast cancer. Med Dosim. 2010; 35:163–168.

3. Taghian A, Jagsi R, Makris A, Goldberg S, Ceilley E, Grignon L, et al. Results of a survey regarding irradiation of internal mammary chain in patients with breast cancer: practice is culture driven rather than evidence based. Int J Radiat Oncol Biol Phys. 2004; 60:706–714.

4. Freedman GM, Fowble BL, Nicolaou N, Sigurdson ER, Torosian MH, Boraas MC, et al. Should internal mammary lymph nodes in breast cancer be a target for the radiation oncologist? Int J Radiat Oncol Biol Phys. 2000; 46:805–814.

5. Matzinger O, Heimsoth I, Poortmans P, Collette L, Struikmans H, Van Den Bogaert W, et al. Toxicity at three years with and without irradiation of the internal mammary and medial supraclavicular lymph node chain in stage I to III breast cancer (EORTC trial 22922/10925). Acta Oncol. 2010; 49:24–34.

6. Whelan TJ, Olivotto I, Ackerman I, Chapman JW, Chua B, Nabid A, et al. NCIC-CTG MA. 20: an intergroup trial of regional nodal irradiation in early breast cancer. J Clin Oncol. 2011; 29:15 Suppl. LBA1003.
7. Overgaard M, Hansen PS, Overgaard J, Rose C, Andersson M, Bach F, et al. Postoperative radiotherapy in high-risk premenopausal women with breast cancer who receive adjuvant chemotherapy: Danish Breast Cancer Cooperative Group 82b Trial. N Engl J Med. 1997; 337:949–955.

8. Chander SS. Postoperative radiotherapy in high-risk postmenopausal breast cancer: authors' reply. Lancet. 1999; 354:865–866.
9. Chang JS, Park W, Kim YB, Lee IJ, Keum KC, Lee CG, et al. Long-term survival outcomes following internal mammary node irradiation in stage II-III breast cancer: results of a large retrospective study with 12-year follow-up. Int J Radiat Oncol Biol Phys. 2013; 86:867–872.

10. Kubo A, Osaki K, Kawanaka T, Furutani S, Ikushima H, Nishitani H. Risk factors for radiation pneumonitis caused by whole breast irradiation following breast-conserving surgery. J Med Invest. 2009; 56:99–110.

11. Lingos TI, Recht A, Vicini F, Abner A, Silver B, Harris JR. Radiation pneumonitis in breast cancer patients treated with conservative surgery and radiation therapy. Int J Radiat Oncol Biol Phys. 1991; 21:355–360.

12. Arthur DW, Arnfield MR, Warwicke LA, Morris MM, Zwicker RD. Internal mammary node coverage: an investigation of presently accepted techniques. Int J Radiat Oncol Biol Phys. 2000; 48:139–146.

13. Jeong K, Shim SJ, You SH, Kim YB, Keum KC, Kim JD, et al. A study of the radiotherapy techniques for the breast including internal mammary lymph nodes. J Korean Soc Ther Radiol Oncol. 2009; 27:35–41.

14. Kim HJ, Jang WI, Kim TJ, Kim JH, Kim SW, Moon SH, et al. Radiation-induced pulmonary toxicity and related risk factors in breast cancer. J Breast Cancer. 2009; 12:67–72.

15. Chung Y, Yoon HI, Kim YB, Ahn SK, Keum KC, Suh CO. Radiation pneumonitis in breast cancer patients who received radiotherapy using the partially wide tangent technique after breast conserving surgery. J Breast Cancer. 2012; 15:337–343.

16. Krengli M, Sacco M, Loi G, Masini L, Ferrante D, Gambaro G, et al. Pulmonary changes after radiotherapy for conservative treatment of breast cancer: a prospective study. Int J Radiat Oncol Biol Phys. 2008; 70:1460–1467.

17. Muren LP, Maurstad G, Hafslund R, Anker G, Dahl O. Cardiac and pulmonary doses and complication probabilities in standard and conformal tangential irradiation in conservative management of breast cancer. Radiother Oncol. 2002; 62:173–183.

18. Das IJ, Cheng EC, Freedman G, Fowble B. Lung and heart dose volume analyses with CT simulator in radiation treatment of breast cancer. Int J Radiat Oncol Biol Phys. 1998; 42:11–19.

19. Lind PA, Wennberg B, Gagliardi G, Rosfors S, Blom-Goldman U, Lideståhl A, et al. ROC curves and evaluation of radiation-induced pulmonary toxicity in breast cancer. Int J Radiat Oncol Biol Phys. 2006; 64:765–770.

20. Prosnitz RG, Chen YH, Marks LB. Cardiac toxicity following thoracic radiation. Semin Oncol. 2005; 32:2 Suppl 3. S71–S80.

21. Prosnitz RG, Hubbs JL, Evans ES, Zhou SM, Yu X, Blazing MA, et al. Prospective assessment of radiotherapy-associated cardiac toxicity in breast cancer patients: analysis of data 3 to 6 years after treatment. Cancer. 2007; 110:1840–1850.

22. Harris EE. Cardiac mortality and morbidity after breast cancer treatment. Cancer Control. 2008; 15:120–129.

23. Nilsson G, Holmberg L, Garmo H, Terent A, Blomqvist C. Radiation to supraclavicular and internal mammary lymph nodes in breast cancer increases the risk of stroke. Br J Cancer. 2009; 100:811–816.

24. Bartlett FR, Colgan RM, Carr K, Donovan EM, McNair HA, Locke I, et al. The UK HeartSpare Study: randomised evaluation of voluntary deep-inspiratory breath-hold in women undergoing breast radiotherapy. Radiother Oncol. 2013; 108:242–247.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download