Abstract
Purpose
This study assessed the axillary lymph node (ALN)-to-primary tumor maximum standard uptake value (SUVmax) ratio (ALN/T SUV ratio) in invasive ductal breast cancer (IDC) on preoperative 18F-fluorodeoxyglucose positron emission tomography/computed tomography (FDG PET/CT) to determine the effectiveness in predicting recurrence-free survival (RFS).
Methods
One hundred nineteen IDC patients (mean age, 50.5±10.5 years) with pathologically proven ALN involvement without distant metastasis and preoperative FDG PET/CT were enrolled in the study. SUVmax values of the ALN and primary tumor were obtained on FDG PET/CT, and ALN/T SUV ratio was calculated. Several factors were evaluated for their effectiveness in predicting RFS. These included several parameters on FDG PET/CT as well as several clinicopathological parameters: pathologic tumor/node stage; nuclear and histological grade; hormonal state; status with respect to human epidermal growth factor receptor 2, mindbomb E3 ubiquitin protein ligase 1 (MIB-1), and p53; primary tumor size; and ALN size.
Results
Among 119 patients with breast cancer, 17 patients (14.3%) experienced relapse during follow-up (mean follow-up, 28.4 months). The ALN/T SUV ratio of the group with disease recurrence was higher than that of the group without recurrence (0.97±1.60 and 0.45±0.40, respectively, p=0.005). Univariate analysis showed that the primary tumor SUVmax, ALN SUVmax, ALN/T SUV ratio, ALN status, nuclear and histological grade, estrogen receptor (ER) status, and MIB-1 status were predictors for RFS. Among these variables, ALN/T SUV ratio with hazard ratio of 4.20 (95% confidence interval [CI], 1.74-10.13) and ER status with hazard ratio of 4.33 (95% CI, 1.06-17.71) were predictors for RFS according to multivariate analysis (p=0.002 and p=0.042, respectively).
Breast cancer is the most common malignancy among women worldwide, and the incidence has increased tremendously in the past several decades. Newly developed imaging techniques and adjuvant systemic therapy have improved breast cancer survival; however, nearly 30% of breast cancer patients still die due to disease progression [1]. Therefore, it is important to identify cases which are likely to experience relapse and decide on optimal therapy.
Proven prognostic factors in breast cancer include the following: pathologic tumor size; histological grade; axillary lymph node (ALN) involvement; hormonal status including estrogen receptor (ER) and progesterone receptor (PR) status; and status with respect to human epidermal growth factor receptor 2 (HER2), mindbomb E3 ubiquitin protein ligase 1 (MIB-1), and p53. ALN involvement is currently the most important predictor of breast cancer recurrence [23]. Current advances in 18F-fluorodeoxyglucose positron emission tomography/computed tomography (FDG PET/CT) have facilitated the prediction of recurrence in breast cancers. Several studies have reported that 18F-FDG uptake by primary tumor on FDG PET/CT is of prognostic value [45678].
Recent studies have evaluated the utility of the lymph node-to-primary tumor standard uptake value (SUV) ratio as an index of the biological interaction between the primary tumor and the lymph node [91011]. Results from these studies showed that the lymph node-to-primary tumor SUV ratio was useful for initial staging or predicting relapse in lung cancer. Park et al. [12] recently revealed that ALN-to-primary tumor SUV ratio (ALN/T SUV ratio) measured by preoperative FDG PET/CT is a significant factor for predicting ALN involvement in patients with invasive ductal breast cancer (IDC). Until now, no prior studies have investigated the clinical utility of the ALN/T SUV ratio for predicting relapse in IDC. The present study was undertaken to evaluate ALN/T SUV ratio measured by preoperative FDG PET/CT as a predictor for relapse in IDC with metastatic ALN.
A total of 153 female IDC patients with ALN metastasis were enrolled at our institution between November 2010 and December 2012, and their medical charts were retrospectively reviewed. Our Institutional Review Board approved the retrospective chart review (2014-04-027-002). The inclusion criteria were verification of primary IDC and ALN metastasis by histological evaluation, preoperative FDG PET/CT for clinical staging, and tumors >1 cm in pathologic size based on full-width at half maximum (FWHM) of PET [131415]. Patients who had bilateral breast cancers and those who had received neoadjuvant chemotherapy or radiotherapy before surgery were excluded from the study. A total of 119 IDC patients with mean age of 50.5±10.5 years (range, 30-76 years) were included in the present study.
All patients had undergone breast-conserving surgery or modified radical mastectomy with sentinel lymph node biopsy and/or ALN dissection. Systemic chemotherapy postsurgery included a taxane-based regimen consisting of doxorubicin and cyclophosphamide followed by docetaxel. Radiotherapy was conducted postsurgery. Hormonal therapy or trastuzumab was provided to the patients with hormonal receptor positive or HER2-positive primary tumors. Patients visited the hospital every 6 months for a minimum of 3 years after initial treatment and returned annually thereafter. Physical examinations and follow-up examinations including mammo-graphy, breast ultrasonography, and whole-body bone scan were routinely performed. FDG PET/CT and breast magnetic resonance imaging were performed 1 and 3 years after the initial treatment. In patients with suspected relapse, additional analysis by conventional imaging techniques followed. Survival time was computed from the date of first pathological diagnosis until recurrence or the date of the last follow-up. Recurrence-free survival (RFS) was defined as survival without any of the following events: invasive recurrence in the ipsilateral breast or locoregionally; distant recurrence; or death from breast cancer, nonbreast cancer causes, or unknown causes as mentioned in the previous study [16]. Breast cancers were determined to be recurrent or metastatic based on the evaluation of the physicians responsible for the patients as reported in the medical charts. All of the data were obtained from medical records.
FDG PET/CT images were obtained using a Siemens Biograph mCT/128 PET/CT scanner (Siemens Medical Solutions, Knoxville, USA). Before administration of 3-5 MBq/kg of 18F-FDG, patients fasted for at least 6 hours to ensure a serum glucose level of less than 150 mg/dL. Sixty minutes after administration of 18F-FDG, CT scans were performed in the craniocaudal direction, and the parameters were set to 120 kVp and 50 mAs using dose reduction software (CARE Dose; Siemens Medical Solutions). FDG PET/CT was acquired with a 2-minute emission acquisition per bed position from the mid-thigh to the skull base. CT images were used as a transmission map for attenuation correction, and PET images were reconstructed to 200×200 image matrices using an ordered subset expectation maximum iterative reconstruction algorithm (two iterations, 21 subsets).
To measure the SUVmax of the ALN and the primary tumor on preoperative FDG PET/CT, a semiautomatically delineated spherical volume of interest (≥1 cm in diameter) was drawn over the primary breast tumor and axillary area on transaxial fusion images using a custom software program (syngo.via; Siemens Medical Solutions). Margins of volume of interest were manually readjusted by visual inspection to avoid overlapping of surrounding 18F-FDG avid structures, especially in ALN with low 18F-FDG uptake. Afterward, ALN/T SUV ratios were calculated (Figure 1). For calculating ALN size, the longest ALN diameters were measured on CT images of preoperative FDG PET/CT.
The immunohistochemical features of breast cancer were confirmed by an experienced breast pathologist, following the procedure and interpretation reported in a previous study [17]. Briefly, pathologic tumor and ALN staging was interpreted according to the guidelines recommended by the American Joint Committee on Cancer [18]. Furthermore, histological or nuclear grade was determined in accordance with a modified Bloom-Richardson Grading system in which grade 3 was defined as positive [19]. An Allred score of >2 was used to define ER or PR positivity as described previously [20]. Positivity with respect to p53 was defined as the presence of tumor cells showing moderate or high scores (2+ or 3+) of at least 5% staining. MIB-1 positivity was defined as staining of ≥20% of invasive tumor cells, and HER2 positivity as presence of at least 10% of tumor cells showing high intensity staining with a receptor antibody or low-to-moderate intensity with gene amplification (>2.1) using fluorescent in situ hybridization [2122]. For primary tumor size, the longest primary tumor diameter was measured from permanent tissue after the operation.
Numeric data were expressed as the mean±standard deviation. Optimal cutoff values for primary tumor SUVmax, ALN SUVmax, and ALN/T SUV ratio were determined by using the receiver operating characteristic (ROC) curve for the prediction of relapse. Survival analysis was conducted using the Kaplan-Meier method. Univariate and multivariate analyses were performed using the Cox proportional hazards model. Statistical significance was defined as p<0.05. All statistical analyses were performed using MedCalc software version 11.4.4 (MedCalc Software, Mariakerke, Belgium).
The characteristics of the 119 study patients are shown in Table 1. The interval between preoperative FDG PET/CT and surgery was 6.7±9.4 days and the mean follow-up duration was 28.4±9.0 months. Ten of the 119 breast cancer patients underwent ALN biopsy before FDG PET/CT. Among the 119 patients, 118 (99.2%) received adjuvant chemotherapy and 101 (84.9%) received radiation therapy after operation. There were 82 patients (68.9%) in stage II and 37 (31.1%) in stage III of breast cancer. During follow-up, 102 patients (85.7%) stayed disease-free and 17 (14.3%) experienced disease recurrence. Among the 17 patients in whom the disease recurred, six had locoregional recurrent lesions and the other 11 had distant metastatic lesions. The average RFS of the 17 IDC patients with disease recurrence was 16.3±8.6 months.
IDC patients with relapse had significantly higher primary tumor SUVmax and ALN SUVmax than those without relapse. The primary tumor SUVmax of the group with disease recurrence was 11.3±6.7 and that of the group without recurrence was 7.9±4.6 (95% confidence interval [CI] of difference, -6.00 to -0.83; p=0.010). The ALN SUVmax of the two groups was 6.9±6.5 and 3.4±4.2, respectively (95% CI, -5.86 to -1.15; p=0.004). The ALN/T SUV ratio of the former was 0.97±1.60 and that of the latter was 0.45±0.40 (95% CI, -0.88 to -0.16; p=0.005). Primary tumor size and ALN size in IDC patients with relapse had no significant differences from those of IDC patients without relapse (p=0.698 and p=0.068, respectively).
Using an ROC curve to predict relapse, the optimal cutoff values for the ALN and primary tumor SUVmax were 2.2 (sensitivity, 88.2%; specificity, 56.9%; area under the curve [AUC], 0.738; standard error [SE], 0.0575) and 11.1 (sensitivity, 52.9%; specificity, 81.4%; AUC, 0.660; SE, 0.0815), respectively. The ALN/T SUV ratio for predicting relapse was 0.43 (sensitivity, 70.6%; specificity, 69.6%; AUC, 0.657; SE, 0.0777). Comparison between the AUCs derived from the ROC curves showed that there were no significant differences in performance among primary tumor SUVmax, ALN SUVmax, and ALN/T SUV (all p>0.05). Survival analysis by using Kaplan-Meier analysis showed that primary tumor SUVmax (>11.1), ALN SUVmax (>2.2), and ALN/T SUV ratio (>0.43) were significant predisposing factors for RFS in IDC (Figure 2).
Univariate analysis also showed that primary tumor SUVmax, ALN SUVmax, and ALN/T SUV ratio were significant predisposing factors for RFS in IDC (Table 2). Clinicopathological findings including ALN status (pN1 vs. pN2, pN3), nuclear and histological grade (I, II vs. III), ER (positive vs. negative), and MIB-1 (positive vs. negative) were also significant predisposing factor for RFS in IDC. Multivariate analysis showed that ALN/T SUV ratio (hazard ratio, 4.20; 95% CI, 1.74-10.13; p=0.002), and ER status (hazard ratio, 4.33; 95% CI, 1.06-17.71; p=0.042) were significant independent factors for predicting RFS.
To the best of our knowledge, this is the first study to evaluate the value of ALN/T SUV ratio in prediction of relapse in IDC patients with ALN metastasis. The results revealed that ALN/T SUV ratio together with ER status were significant independent factors for predicting RFS.
Okereke et al. [23] recently reported that the SUVmax of primary tumors was higher than that of metastatic lesions in lung cancer. Nguyen et al. [24] showed a significant correlation between malignant lymph node and primary tumor with respect to SUVmax (r=0.6451, p<0.0001), % glucose transporter 1 (Glut-1) expression (r=0.8341, p<0.0001) and Glut-1 staining intensity (r=0.827, p<0.0001) in lung cancer. Furthermore, he suggested that lymph node-to-primary tumor SUV ratio of 0.58 was a useful value to predict the presence of metastatic lymph nodes. Based on these results, several studies evaluated whether lymph node-to-primary tumor SUV ratio was a useful factor for initial staging or prediction of relapse in lung cancer [91011]. Moreover, a recent study by Park et al. [12] revealed that ALN/T SUV ratio had significantly higher AUC than ALN SUVmax for discriminating ALN metastasis in breast cancer (0.776% vs. 0.705%, p=0.005). Therefore, we speculated that lymph node and primary tumor SUV ratio could be linearly related, and studied the clinical value of ALN/T SUV ratio for predicting disease-free survival in IDC with metastatic ALN. This study revealed that ALN/T SUV ratio was a significant independent factor for predicting RFS by multivariate analysis. Actually, breast cancer with relapse had higher ALN/T SUV ratio than breast cancer without relapse. Our data indicated that the incidence of relapse in IDC patients with ALN/T SUV ratio >0.43 was 27.3% (12/44), whereas that in IDC patients with ALN/T SUV ratio ≤0.43 was 6.7% (5/75).
In the present study, ALN SUVmax was a significant factor for predicting relapse by univariate analysis but not by multivariate analysis. Previously, Inoue et al. [7] showed that both primary tumor SUVmax and a combination of primary tumor SUVmax and ALN avidity were significant factors for predicting relapse by multivariate analysis. However, Song et al. [25] reported that ALN SUVmax was the only significant independent factor for predicting relapse and that the optimal cutoff for ALN SUVmax was 2.8. The use of nonnormalized SUV in different studies may be the cause for such discrepancies. Generally, several factors such as tumor type, blood glucose level, time interval between tracer injection and scanning time, type of machines, and acquisition time per bed can result in SUV variability. Unlike previous studies, our study included an additional 18F-FDG value, namely the ALN/T SUV ratio, to predict RFS in IDC with metastatic ALN. ALN/T SUV ratio can reflect the biologic interactions between the primary tumor and ALN as reported previously [10]. Furthermore, a recent study by Park et al. [12] revealed that ALN/T SUV ratio is a more accurate value for discriminating ALN involvement than ALN SUVmax value.
Generally, FDG PET/CT is based on the initial metabolism of the primary tumor. In this study, we found that primary tumor SUVmax was a significant factor for predicting relapse in IDC by univariate analysis. The incidence of relapse in IDC patients with primary tumor SUVmax >11.1 was 32.1% (9/28), but that in patients with primary tumor SUVmax ≤11.1 was lower, at 8.8% (8/91). This finding was consistent with previous studies [26]. Ohara et al. [26] predicted that patients with triple-negative breast cancer with primary tumor SUVmax >8.6 would have short RFS and would require additional adjunct therapy. However, multivariate analysis showed that primary tumor SUVmax was not a significant independent factor for predicting relapse as reported previously [25].
ER was a significant independent factor for predisposition to relapse in multivariate analysis [202627]. According to Ohara et al. [26], ER negativity together with high primary tumor SUVmax offered poor prognosis for relapse in breast cancer. Generally, hormonal therapy is provided after breast cancer surgery in accordance with the hormonal status. Therefore, the possibility of relapse may be reduced in the ER positive group with hormonal therapy, compared to the ER negative group without hormonal therapy. Other clinicopathological features of primary tumors including pathologic tumor/node stage, nuclear or histologic grade, PR status, HER2 status, MIB-1 and p53 status, and primary tumor size were not significant independent variables for predicting RFS. This is consistent with previous reports [152628]. The size of the ALN was also not a significant factor for predicting relapse in this study.
In the present study, there was no significant difference in performance among the parameters on FDG PET/CT including ALN/T SUV ratio, primary tumor SUVmax, and ALN SUVmax. However, the ALN/T SUV ratio on FDG PET/CT was found to be the only significant independent factor for predicting relapse in IDC with metastatic ALN. There were several limitations to the present study. We retrospectively reviewed the medical charts of IDC patients with ALN metastasis. Additionally, the follow-up period was relatively short (28.4 months). Thus, the results may not extend to generalized breast cancers. Nonetheless, IDC is the most common breast cancer subtype. Further prospective studies with a longer follow-up period are needed to confirm our results. The 18F-FDG uptake of ALN metastasis could be underestimated due to the partial volume effect. Although the 18F-FDG uptake of small sized ALN may be underestimated, only two of 60 breast cancer patients with ALN SUVmax ≤2.2 relapsed during follow-up. Axillary 18F-FDG uptake can be overestimated due to ALN biopsy prior to FDG PET/CT because of inflammatory changes. Actually, 10 patients among 119 breast cancer patients underwent ALN biopsy before FDG PET/CT in the present study. Thus, subgroup analyses were performed on 109 breast cancer patients who underwent FDG PET/CT before ALN biopsy. The ALN/T SUV ratio of several parameters on FDG PET/CT was still a single independent factor for predicting relapse by multivariate analysis (hazard ratio, 19.48; 95% CI, 1.23-307.73; p=0.037). Further prospective studies without ALN biopsy before FDG PET/CT are needed to confirm our results.
In conclusion, this study is the first report on the use of ALN/T SUV ratio to predict relapse in breast cancer. Our study demonstrated that ALN/T SUV ratio together with ER status was an independent factor for predicting relapse in IDC patients with metastatic ALN. ALN/T SUV ratio could be used to identify candidates in need of closer follow-up or treatment intensification.
Figures and Tables
Figure 1
Image findings of a 47-year-old woman with primary breast cancer and axillary lymph node metastasis. Preoperative 18F-fluorodeoxyglucose positron emission tomography/computed tomography (FDG PET/CT) demonstrating a right breast cancer with intense increased FDG uptake (maximum standard uptake value [SUVmax], 3.2) in the upper outer quadrant area of the right breast (A-C, arrows). Axillary lymph node metastasis with faintly increased FDG uptake (SUVmax, 1.5) was seen in the right axillary area (D, E, arrowheads). The SUVmax ratio of the axillary lymph node to primary tumor was 0.47 on FDG PET/CT.
MIP=maximum intensity projection.
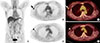
Figure 2
Survival analysis by using Kaplan-Meier method for invasive ductal breast cancer with metastatic axillary lymph node (ALN). A significant statistical difference was observed between high primary tumor maximum standard uptake value (SUVmax) and low primary tumor SUVmax (A), between high ALN SUVmax and low ALN SUVmax (B), and between high ALN-to-primary tumor SUV ratio (ALN/T SUV) ratio and low ALN/T SUV ratio (C).
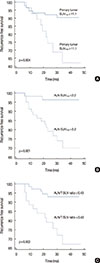
Table 1
Tumor characteristics of invasive ductal breast cancer with axillary lymph node metastasis

Table 2
Tumor factor associated with recurrence in invasive ductal breast cancer with axillary lymph node metastasis

HR=hazard ratio; CI=confidence interval; SUVmax=maximum standard uptake value; ALN=axillary lymph node; ALN/T=axillary lymph node to primary tumor; ER=estrogen receptor; PR=progesterone receptor; HER2=human epidermal growth factor receptor 2; MIB-1=mindbomb E3 ubiquitin protein ligase 1.
*Continuous value.
References
2. Kim JY, Ryu MR, Choi BO, Park WC, Oh SJ, Won JM, et al. The prognostic significance of the lymph node ratio in axillary lymph node positive breast cancer. J Breast Cancer. 2011; 14:204–212.

3. Lale Atahan I, Yildiz F, Ozyigit G, Sari S, Gurkaynak M, Selek U, et al. Percent positive axillary lymph node metastasis predicts survival in patients with non-metastatic breast cancer. Acta Oncol. 2008; 47:232–238.

4. Cochet A, Dygai-Cochet I, Riedinger JM, Humbert O, Berriolo-Riedinger A, Toubeau M, et al. 18F-FDG PET/CT provides powerful prognostic stratification in the primary staging of large breast cancer when compared with conventional explorations. Eur J Nucl Med Mol Imaging. 2014; 41:428–437.

5. Oshida M, Uno K, Suzuki M, Nagashima T, Hashimoto H, Yagata H, et al. Predicting the prognoses of breast carcinoma patients with positron emission tomography using 2-deoxy-2-fluoro[18F]-D-glucose. Cancer. 1998; 82:2227–2234.

6. Alberini JL, Lerebours F, Wartski M, Fourme E, Le Stanc E, Gontier E, et al. 18F-fluorodeoxyglucose positron emission tomography/computed tomography (FDG-PET/CT) imaging in the staging and prognosis of inflammatory breast cancer. Cancer. 2009; 115:5038–5047.

7. Inoue T, Yutani K, Taguchi T, Tamaki Y, Shiba E, Noguchi S. Preoperative evaluation of prognosis in breast cancer patients by [(18)F]2-deoxy-2-fluoro-D-glucose-positron emission tomography. J Cancer Res Clin Oncol. 2004; 130:273–278.

8. Ueda S, Tsuda H, Asakawa H, Shigekawa T, Fukatsu K, Kondo N, et al. Clinicopathological and prognostic relevance of uptake level using 18F-fluorodeoxyglucose positron emission tomography/computed tomography fusion imaging (18F-FDG PET/CT) in primary breast cancer. Jpn J Clin Oncol. 2008; 38:250–258.

9. Cerfolio RJ, Bryant AS. Ratio of the maximum standardized uptake value on FDG-PET of the mediastinal (N2) lymph nodes to the primary tumor may be a universal predictor of nodal malignancy in patients with nonsmall-cell lung cancer. Ann Thorac Surg. 2007; 83:1826–1829.

10. Kaira K, Endo M, Asakura K, Tsuya A, Nakamura Y, Naito T, et al. Ratio of standardized uptake value on PET helps predict response and outcome after chemotherapy in advanced non-small cell lung cancer. Ann Nucl Med. 2010; 24:697–705.

11. Iskender I, Kadioglu SZ, Kosar A, Atasalihi A, Kir A. Is there any maximum standardized uptake value variation among positron emission tomography scanners for mediastinal staging in non-small cell lung cancer? Interact Cardiovasc Thorac Surg. 2011; 12:965–969.

12. Park J, Byun BH, Noh WC, Lee SS, Kim HA, Kim EK, et al. Lymph node to primary tumor SUV ratio by 18F-FDG PET/CT and the prediction of axillary lymph node metastases in breast cancer. Clin Nucl Med. 2014; 39:e249–e253.

13. Kaida H, Toh U, Hayakawa M, Hattori S, Fujii T, Kurata S, et al. The relationship between 18F-FDG metabolic volumetric parameters and clinicopathological factors of breast cancer. Nucl Med Commun. 2013; 34:562–570.

14. Boellaard R, O'Doherty MJ, Weber WA, Mottaghy FM, Lonsdale MN, Stroobants SG, et al. FDG PET and PET/CT: EANM procedure guidelines for tumour PET imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2010; 37:181–200.
15. Nakajima N, Kataoka M, Sugawara Y, Ochi T, Kiyoto S, Ohsumi S, et al. Volume-based parameters of 18F-fluorodeoxyglucose positron emission tomography/computed tomography improve disease recurrence prediction in postmastectomy breast cancer patients with 1 to 3 positive axillary lymph nodes. Int J Radiat Oncol Biol Phys. 2013; 87:738–746.

16. Hudis CA, Barlow WE, Costantino JP, Gray RJ, Pritchard KI, Chapman JA, et al. Proposal for standardized definitions for efficacy end points in adjuvant breast cancer trials: the STEEP system. J Clin Oncol. 2007; 25:2127–2132.

17. Seok JW, Kim Y, An YS, Kim BS. The clinical value of tumor FDG uptake for predicting axillary lymph node metastasis in breast cancer with clinically negative axillary lymph nodes. Ann Nucl Med. 2013; 27:546–553.

18. Elston CW, Ellis IO. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology. 1991; 19:403–410.

19. Robbins P, Pinder S, de Klerk N, Dawkins H, Harvey J, Sterrett G, et al. Histological grading of breast carcinomas: a study of interobserver agreement. Hum Pathol. 1995; 26:873–879.

20. Harvey JM, Clark GM, Osborne CK, Allred DC. Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol. 1999; 17:1474–1481.

21. Goldhirsch A, Winer EP, Coates AS, Gelber RD, Piccart-Gebhart M, Thürlimann B, et al. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol. 2013; 24:2206–2223.

22. Wolff AC, Hammond ME, Schwartz JN, Hagerty KL, Allred DC, Cote RJ, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol. 2007; 25:118–145.

23. Okereke IC, Gangadharan SP, Kent MS, Nicotera SP, Shen C, DeCamp MM. Standard uptake value predicts survival in non-small cell lung cancer. Ann Thorac Surg. 2009; 88:911–915.

24. Nguyen XC, So Y, Chung JH, Lee WW, Park SY, Kim SE. High correlations between primary tumours and loco-regional metastatic lymph nodes in non-small-cell lung cancer with respect to glucose transporter type 1-mediated 2-deoxy-2-F18-fluoro-D-glucose uptake. Eur J Cancer. 2008; 44:692–698.

25. Song BI, Lee SW, Jeong SY, Chae YS, Lee WK, Ahn BC, et al. 18F-FDG uptake by metastatic axillary lymph nodes on pretreatment PET/CT as a prognostic factor for recurrence in patients with invasive ductal breast cancer. J Nucl Med. 2012; 53:1337–1344.

26. Ohara M, Shigematsu H, Tsutani Y, Emi A, Masumoto N, Ozaki S, et al. Role of FDG-PET/CT in evaluating surgical outcomes of operable breast cancer: usefulness for malignant grade of triple-negative breast cancer. Breast. 2013; 22:958–963.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download