Abstract
Purpose
Poor cosmetic outcome have been reported as a result of breast cancer operation due to lower quadrant breast tumors; this is particularly true for women with small, firm breasts. Herein, we report here on the use of superior based lateral breast rotation flap reconstruction to improve cosmetic outcome in patients with lower quadrant breast cancer.
Methods
We enrolled 33 patients with invasive breast cancer located in the lower quadrant of the breast, which were located more than 2 cm apart from the nipple. After completing a quadrantectomy, a single S-shaped or reverse S-shaped incision was made from axilla to tumor site. Two triangular skin islands, one on the axilla and one overlying the tumor were marked for excision. Once the fibroglandular tissues and the additional fatty tissue of the lateral chest wall were appropriately mobilized, the breast defect was closed at the mid-point of the parenchymal thickness in order to keep the natural position of the infra mammary fold.
Results
Median tumor size was 2.3 cm (range, 0.7-3.5 cm) and median resected volume was 35.5 g (range, 27.0-51.0 g). With a mean follow-up of 24.5 months (range, 9.0-33.5 months), cosmetic outcomes were good (94.0%) to fair (6.0%) at 6 months after the procedure, and there was no local or systemic recurrence during the short term follow-up period.
Conclusion
Clearly, this type of rotation flap reconstruction is an oncologically safe and a cosmetically sound procedure. Hopefully this rotation flap reconstruction technique will become more widely available and perhaps a standard procedure for lower quadrant breast tumors, especially for cosmetic treatment of small to medium-sized breasts.
The purpose of oncoplastic surgery for breast cancer is to improve the cosmetic outcome without impacting local control after resection. Generally, oncoplastic surgery consists of two components; wide local excision of the tumor and reshaping of the breast. When performing breast conserving operations, oncological surgeons should think about breast cancer distribution. Amano and colleagues [1] classified the distribution of breast cancer into three patterns based on observation of preoperative magnetic resonance imaging (MRI) and histopathological findings: localized (55%), segmentally extended (35%), and irregularly extended (10%). Histologically, pure or predominant ductal carcinoma in situ (DCIS) was distributed segmentally [1,2]. Localized tumors usually do not include an extensive intraductal component. Based on this observation, we thought a quadrantectomy could cover 90% of the breast cancer patients while lumpectomy could cover only 55%. This concept has been the reason we chose quadrantectomy as a breast conserving operation rather than lumpectomy, for oncological safety. Various available techniques are used to repair breast defects after surgery for breast cancer [3-5]. Usually the techniques include transferring adjacent breast parenchyma with or without overlying skin for volume replacement. This rearrangement of breast parenchyma makes later follow-up or re-excision of the tumor site difficult. Such approaches are especially important in all women with a small firm breast [6,7]. The location of the tumor can have a negative effect on cosmetic outcome. When performing breast conserving operations, inferior cosmetic outcome has been reported in lower half breast tumors [8,9]. Reduction mastopexy lumpectomy is commonly used for breast cancer that arise below the nipple, but this has not been optimal for women with small firm breasts [10,11]. Herein, we report here on the use of superior based lateral breast rotation flap reconstruction to improve cosmetic outcome in patients with lower quadrant breast tumors. In this procedure, incision lines resemble S-shape for the left breast and reverse S-shape for the right breast.
Preoperative breast imaging was performed to determine cancer distribution. On a preoperative basis, each breast cancer patient underwent the following imaging studies: mammography, ultrasonography, breast MRI, positron emission tomography integrated with computed tomography (PET/CT), and a whole-body bone scans. We included patients with invasive breast cancer that was located in the lower quadrant of the breasts, and the tumors were located more than 2 cm from the nipple (Figure 1). We excluded patients from the study, who had one or more of the following: large pendulous breast, tumors that were too large for the breast conserving operation, multicentricity, tumors involving the nipple, and patients with blood stained nipple discharge or diffuse microcalcification. After adjuvant chemotherapy, if that was indicated, the breast was treated with up to 50 Gy with conventional fraction of 2 Gy/day. A boost was administered to the tumor excision site to bring the total dose to 60 Gy when advised by the radiotherapist. After chemoradiation or 6 months after undergoing the operation, cosmesis was assessed by a panel of two surgeons when patients visited at outpatient department. Cosmesis was based on the following four parameters: symmetry, shape, scarring, and the position of the nipple-areolar complex (NAC). Cosmetic outcome was rated as good, fair, or poor according to sum of scores of the above four parameters (Table 1).
All surgical procedures were performed by one surgeon. Single S-shaped or reverse S-shaped incision lines were drawn on the skin from the axilla to the tumor site prior to beginning the procedure (Figures 2A and 3A). Triangular skin islands, one in the axilla for Burow's triangle and one overlying the tumor were marked for excision. Sentinel lymph node biopsy was performed through the small incision on the same line of the drawing at the axilla and was sent for frozen section. Tumors were then resected by radial-segmental lumpectomy of the fibroglandular tissue at full thickness with the overlying skin triangle (quadrantectomy) to avoid excessive redundant skin and postoperative skin-pectoral adherence deformities. The resected specimens were sent for frozen sections to define margin status. If the margin of the specimen was clear and the tumor was more than 1 cm in distance from the margin, the process continued to the oncoplastic procedure for the purpose of reconstructing breast defects.
After completing a quadrantectomy, a long skin incision was made from the axilla to the tumor site along the anterior axillary line and infra mammary fold (Figures 2B and 3B), and fibroglandular tissue of the breast was mobilized from the pectoral muscle for advancement to close the breast defect. The triangular axillary skin island (Burow's triangle) was removed along with the underlying axillary fat in order to reduce the tension on the breast, which resulted from the removal of the specimen with overlying skin and subsequent mobilization of the adjacent skin and subcutaneous tissue, as well as the mobilization of the breast from the pectoral muscle. During the mobilization procedure of the breast parenchyma, small branches from the lateral thoracic artery and the lateral half of the intercostal perforators were ligated, while parasternal perforators from the internal mammary arteries and the medial half of the intercostal perforators were preserved intact. Importantly, relatively redundant fatty tissue on the serratus anterior muscle was mobilized together with the fibroglandular tissue to increase the breast volume. By adding fatty tissue from the lateral chest wall, symmetry with the contralateral breast could be maintained after quarantectomy. Once the fibroglandular tissues and the additional fatty tissue of the lateral chest wall were appropriately mobilized, the breast parenchymal defect was closed at the mid-point of the parenchymal thickness in order to keep the natural position of the infra mammary fold.
A total of 33 patients with lower-half located breast cancer underwent operations. Patients, procedure, and tumor characteristics are summarized in Tables 2 and 3. The body mass index (BMI) of the entire patients ranged from 20.5 to 41.3. The median tumor size was 2.3 cm (range, 0.7-3.5 cm) and the median resected volume was 35.5 g (range, 27.0-51.0 g). All patients had negative resection margins on both frozen sections and permanent sections. Two patients showed free margins of 0.5 and 0.6 cm with a DCIS component at the nipple side. Twenty-one patients (63.6%) underwent sentinel lymph node biopsies and 12 patients (36.4%) underwent complete axillary dissections. The mean follow-up was 24.5 months, with a range from 9.0 to 33.5 months. Cosmetic outcomes were good (94.0%) to fair (6.0%) at 6 months after the operation. There were no local or systemic recurrence during the short-term follow-up period (Table 3). The NAC was preserved at the correct position for all patients, and symmetry was also well maintained (Figures 2, 3). One patient experienced breast edema and indentation of the incision line immediately after the radiation. However, this was completely corrected within 6 months. Another patient, who was the first case for this procedure, showed upward movement of the infra mammary fold and resultant indentation due to an overly sharp angled skin resection and a shortage of remaining skin. There was no relationship between tumor location, whether outer or inner quadrant, and cosmetic outcome. There were no complications associated with vascular compromise or infection.
Oncoplastic techniques allow extensive resections for breast-conserving therapy of breast carcinomas [12], but oncoplastic techniques commonly make it more difficult to excise further; most patients with involved margins will need mastectomy due to complexity of the techniques [12-14]. Our technique is divided into a 2-stage operation; first, radial-segmental lumpectomy with a verifying free margin through the frozen section and second, reconstruction with rotation flap advancement. Even if the margin is involved, further excision is very simple in our technique compared to other complex oncoplastic procedures. An advantage of this approach is the simplicity of execution. Full-thickness excision and removal of overlying skin can be done by oncological surgeons without formal training in plastic surgery [4]. Although the multidisciplinary teams are required for the oncoplastic approach, in reality, it's not always feasible to have different teams working closely on entire stages of the process. Other advantages include the ease of axillary dissection, avoiding bird beak deformity, maintaining symmetry by transferring fatty tissue from the lateral chest wall, and an invisible lateral and inferior scar from the front (Figures 2D and 3D). Not all oncoplastic surgery results in good cosmetic outcome. Five percent to twenty-five percent of patients could have poor cosmetic outcome after oncoplastic surgery [12-16]. Standard lumpectomy for cancer in the lower region of the breast causes a down-turning of the nipple, resulting in bird beak deformities. The ability to excise a wider margin may be limited in the patients with lower quadrant breast cancers due to the cosmetic concern. In a study of 50 patients with lower quadrant breast cancers, Nos et al. [17] reported a local recurrence rate of 7% at 5 years. Reduction mastopexy lumpectomy is commonly used in this situation, but is not the optimum procedure for women who have small firm breasts [10,11,15]. Quadrantectomy with superior based lateral breast rotation flap reconstruction could be a new option for lower quadrant breast cancer, especially for the small to medium sized breast. Grisotti suggests that NAC should not be moved closer than 7 cm to midline or higher than 16 cm to the sterna notch [4]. Continuous incisions to the axilla have proven to give more deviation and retraction in NAC after radiotherapy [18,19]. Importantly, there was no shifting of NAC in our technique in spite of making a continuous, long incision to the axilla, even after radiotherapy. Meticulous primary closure of the skin wound could prevent much scarring and resultant retraction of NAC. Our technique made it easy to locate the tumor bed, which would be the focus for boost irradiation, while other complex techniques did not [20]. Keeping the natural position of the inframammary fold is very important to preserve the normal breast contour. The density of the breast parenchyma in most Asian women, including Koreans, gives it some plasticity and tensile strength. For lower quadrant breast tumors, suturing of the defect at its deepest point, which is a technique used for women with relatively large sized breasts, could shift the inframammary fold to an unnatural position in women with small firm breasts [4,6]. This effect could be avoided by suturing at the mid-point of the parenchymal thickness when closing the breast defects. With regard to tumor size, we excluded breast tumors larger than 3.5 cm which are not suitable for the breast conserving operation when dealing with small to medium sized breasts. Abundant collateral circulation in the breast allows ligation of small intercostal perforators and somewhat larger vessels from lateral thoracic arteries. There was no vascular compromise in the outer half of the breast that is supplied by collaterals from intact internal mammary arteries and remaining intercostals perforators [4]. The analysis of our series shows that this approach is safe and reliable. Clearly, superior based lateral breast rotation flap reconstruction is an oncologically safe and a cosmetically sound procedure. Hopefully this rotation flap reconstruction will become more widely available and perhaps a standard procedure for lower half located breast tumors, especially in the small to medium sized breasts.
Figures and Tables
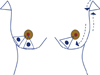 | Figure 1Operative design for patients with lower half located breast cancer. If margin status of the quadrantectomy specimen was adequate, a long skin incision was made from the axilla to the tumor site along the anterior axillary line and inframammary fold. The axillary triangles: axillary skin and underlying fibrofatty tissue which was later removed in this procedure. The triangles with dots: tumor sites where the overlying skin and the tumor down to the chest wall will be removed. Dotted lines indicate the direction of the motion of the skin along the long incision line. |
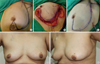 | Figure 2Quadrantectomy and axillary dissection with superior based lateral rotation flap reconstruction for left caudal located breast cancer. (A) Depict the skin triangles to be removed with underlying tissue, one in the axilla and one in the overlying the tumor. (B) After completing quadrantectomy, single S-shaped incision was made from axilla to tumor site and triangular skin islands in the axilla were removed. (C) Once the fibroglandular tissues and the additional fatty tissue of the lateral chest wall were appropriately mobilized, the breast defect was closed. (D, E) Postoperative photos from the front and those from the lateral show a good cosmetic outcome. |
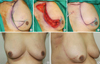 | Figure 3Quadrantectomy and sentinel lymph node biopsy with superior based lateral rotation flap reconstruction for right lower lateral located breast cancer. Operative procedures (A-C) and postoperative photos from the front and lateral (D, E) views show reverse S-shaped incision and scar. |
References
1. Amano G, Ohuchi N, Ishibashi T, Ishida T, Amari M, Satomi S. Correlation of three-dimensional magnetic resonance imaging with precise histopathological map concerning carcinoma extension in the breast. Breast Cancer Res Treat. 2000. 60:43–55.

2. Holland R, Hendriks JH, Vebeek AL, Mravunac M, Schuurmans Stekhoven JH. Extent, distribution, and mammographic/histological correlations of breast ductal carcinoma in situ. Lancet. 1990. 335:519–522.

3. de Andrade Urban C. New classification for oncoplastic procedures in surgical practice. Breast. 2008. 17:321–322.

4. Anderson BO, Masetti R, Silverstein MJ. Oncoplastic approaches to partial mastectomy: an overview of volume-displacement techniques. Lancet Oncol. 2005. 6:145–157.

5. Kronowitz SJ, Kuerer HM, Buchholz TA, Valero V, Hunt KK. A management algorithm and practical oncoplastic surgical techniques for repairing partial mastectomy defects. Plast Reconstr Surg. 2008. 122:1631–1647.

6. Tan M. Additional considerations in the application of oncoplastic approaches. Lancet Oncol. 2005. 6:355–356.

7. Jakes RW, Duffy SW, Ng FC, Gao F, Ng EH. Mammographic parenchymal patterns and risk of breast cancer at and after a prevalence screen in Singaporean women. Int J Epidemiol. 2000. 29:11–19.

8. Steeves RA, Phromratanapongse P, Wolberg WH, Tormey DC. Cosmesis and local control after irradiation in women treated conservatively for breast cancer. Arch Surg. 1989. 124:1369–1373.

9. Bartelink H, Horiot JC, Poortmans P, Struikmans H, Van den Bogaert W, Barillot I, et al. Recurrence rates after treatment of breast cancer with standard radiotherapy with or without additional radiation. N Engl J Med. 2001. 345:1378–1387.

10. Petit JY, Garusi C, Greuse M, Rietiens M, Youssef O, Luini A, et al. One hundred and eleven cases of breast conservation treatment with simultaneous reconstruction at the European Institute of Oncology (Milan). Tumori. 2002. 88:41–47.
11. Losken A, Styblo TM, Carlson GW, Jones GE, Amerson BJ. Management algorithm and outcome evaluation of partial mastectomy defects treated using reduction or mastopexy techniques. Ann Plast Surg. 2007. 59:235–242.

12. Clough KB, Lewis JS, Couturaud B, Fitoussi A, Nos C, Falcou MC. Oncoplastic techniques allow extensive resections for breast-conserving therapy of breast carcinomas. Ann Surg. 2003. 237:26–34.

13. Newman LA, Kuerer HM, McNeese MD, Hunt KK, Gurtner GC, Vlastos GS, et al. Reduction mammoplasty improves breast conservation therapy in patients with macromastia. Am J Surg. 2001. 181:215–220.

14. Spear SL, Pelletiere CV, Wolfe AJ, Tsangaris TN, Pennanen MF. Experience with reduction mammaplasty combined with breast conservation therapy in the treatment of breast cancer. Plast Reconstr Surg. 2003. 111:1102–1109.

15. Clough KB, Nos C, Salmon RJ, Soussaline M, Durand JC. Conservative treatment of breast cancers by mammaplasty and irradiation: a new approach to lower quadrant tumors. Plast Reconstr Surg. 1995. 96:363–370.

16. Asgeirsson KS, Rasheed T, McCulley SJ, Macmillan RD. Oncological and cosmetic outcomes of oncoplastic breast conserving surgery. Eur J Surg Oncol. 2005. 31:817–823.

17. Nos C, Fitoussi A, Bourgeois D, Fourquet A, Salmon RJ, Clough KB. Conservative treatment of lower pole breast cancers by bilateral mammoplasty and radiotherapy. Eur J Surg Oncol. 1998. 24:508–514.

18. Dewar JA, Benhamou S, Benhamou E, Arriagada R, Petit JY, Fontaine F, et al. Cosmetic results following lumpectomy, axillary dissection and radiotherapy for small breast cancers. Radiother Oncol. 1988. 12:273–280.

19. Munshi A, Kakkar S, Bhutani R, Jalali R, Budrukkar A, Dinshaw KA. Factors influencing cosmetic outcome in breast conservation. Clin Oncol (R Coll Radiol). 2009. 21:285–293.

20. Oden S, Thureau S, Baron M, Hanzen C. Conservative treatment for breast cancer: optimization of the tumor bed localization. Cancer Radiother. 2010. 14:96–102.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download