Abstract
Study Design
In vivo study to determine the immune effects to adenoviral vector encoding LMP- 1 cDNA in rabbit.
Objective
To quantify the immune effect of A d5- LMP- 1 in the rabbit during the therapeutic gene transfer.
Summary of Literature Review
One of the major limitations in the use of adenoviral vector for gene therapy is the immune response and it made the poor transduction efficiency when re- administrated. A denoviral antigen plus those derived from transgene expression in transduced cell contribute to cellular, humoral and non- specific immune response constitutes barriers to successful gene therapy. Therefore, the animal immune model will be mandatory to study the immune impact.
Materials and Method
We i.v. injected A d5- β Gal to total 24 adult NZW rabbits; 1x108, 1x109, 1x1010, 1x1011 v.p. to each 6 rabbits allowed them to develop immune response. Six non- immunized animals were used as control. A denovirus antibodies were measured at 0, 4, 8, 16, 20 weeks. Group I. 6 control rabbit underwent spinal arthrodesis at 4 weeks (n=3) and 16 weeks (n=3) with 4 million cells and MOI of 4. Group II. 6 rabbit underwent spinal arthrodesis at 4 weeks after injection of 108 p.f.u virus (n=3) and 16 weeks (n=3). Group III. six 109 immunized rabbits, Group IV. six 1010 immunized rabbits, Group V. six 1011 immunized rabbits, underwent spinal arthrodesis at 4 and 16 weeks after injection. Total anti- Ad Ig and neutralizing antibody titer was measured on the 0. 4. 8, 16, 20 weeks after injection.
Results
Group I. All 6 non- immunized rabbits had solid spine fusions at 4 and 16 weeks. Group II. All 3 immunized rabbits had not spine fusions at 4 weeks and all three had solid spine fusion at 16 weeks. Group III. None of them (n=6) immunized rabbits had spine fusion at 4 and 16 weeks, but some bone formation was observed at 16 weeks. Group IV, V. None of them immunized rabbits had bone formation. The anti- A d5 Ig and neutralizing A b were detected and peaked at the 4 weeks and significantly dropped off 16 weeks after injection.
Go to : 
REFERENCES
1). Boden SD, Schimandle JH, Hutton WC. An experimental lumbar intertransverse process spinal fusion model: Radiographic, histologic, and biomechanical healing characteristics. Spine. 20:412–420. 1995.
2). Boden SD, Schimandle JH, Hutton WC. 1995 Volvo Award in Basic Sciences. The use of an osteoinductive growth factor for lumbar spinal fusion. Part II: Study of dose, carrier, and species. Spine. 20:2633–2644. 1995.
3). Chen P, Kovesdi I, Bruder JT. Effective repeat administration with adenovirus vectors to the muscle. Gene Therapy. 7:587–595. 2000.

4). Chirmule N, Raper SE, Burkly L, Thomas D, Tazelaar J, Hughes JV, Wilson JM. Readministration of adenovirus vector in nonhuman primate lungs by blockade of CD40-CD40 ligand interactions. J Virol. 74:3345–3352. 2000.

5). Christ M, Lusky M, Stoeckel F, Dreyer D, Dieterle A, Michou AI, Pavirani A, Mehtali M. Gene therapy with recombinant adenovirus vectors: evaluation of the host immune response. Immunology Letters. 57:19–25. 1997.

6). Crystal R. Transfer of genes to humans: early lessons and obstacles to success. Science. 270:404–410. 1995.

7). Gahery-Segard H, Farace F, Godfrin D, Gaston J, Lengagne R, Tursz T, Boulanger P, Guillet JG. Immune response to recombinant capsid proteins of adenovirus in humans: antifiber and anti-penton base antibodies have a synergistic effect on neutralizing activity. J Virol. 72:2388–2397. 1998.
8). Gahery-Segard H, Juillard V, Gaston J, Lengagne R, Pavirani A, Boulanger P, Guillet JG. Humoral immune response to the capsid components of recombinant adenoviruses: routes of immunization modulate virus-induced Ig subclass shifts. Eur J Immunol. 27:653–659. 1997.
9). Kagami H, Atkinson JC, Michalek SM, Handelman B, Yu S, Baum BJ, O'Connell B. Repetitive adenovirus administration to the parotid gland: role of immunological barriers and induction of oral tolerance. Hum Gene Ther. 10:305–313. 1998.

10). Kuriyama S, Tominaga K, Mitoro A, Tsujinoue H, Na-katani T, Yamazaki M, Nagao S, Toyokawa Y, Okamo-to S, Fukui H. Immunomodulation with FK506 around the time of intravenous re-administration of an adenoviral vector facilities gene transfer into rat liver. Int J Cancer. 85:839–844. 2000.
11). Mittereder N, March KL, Trapnell BC. Evaluation of the concentration and bioactivity of adenovirus vectors for gene therapy. J Virol. 70:7498–7509. 1996.

12). Moffatt S, Hays J, HogenEsch H, Mittal SK. Circum -vention of vector specific neutralizing antibody response by alternating use of human and non-human adenoviruses: Implications in gene therapy. Virology. 272:159–167. 2000.
13). Molinier-Frenkel V, Gahery-Segard H, Mehtali M, Le Boulaire C, Ribault S, Boulanger P, Tursz T, Guillet JG, Farace F. Immune response to recombinant adenovirus in humans: Capsid components from viral input are targets for vector specific cytotoxic T lymphocyte. J Virol. 74:7678–7682. 2000.
14). Turner RJ, Held SDE, Hirst JE, Billinghurst G, Woot-ton RJ. An immunological assessment of group housed rabbits. Laboratory Animals. 31:362–372. 1997.
15). Viggeswarapu M, Boden SD, Liu Y, Hair GA, Ugbo JL, Murakami H, Kim HS, Mayer MT, Hutton WC, Titus L. Adenoviral delivery of LIM mineralization protrin-1 induces new-bone formation in vitro and in vivo. J Bone Joint Surg Am. 83:364–376. 2001.
16). Waldsworth SC, Zhou H, Smith AE, Kaplan JM. adenovirus vector-infected cells can escape adenovirus antigen specific T-lymphocyte killing in vivo. J Virol. 71:5189–5196. 1997.
Go to : 
Figures and Tables%
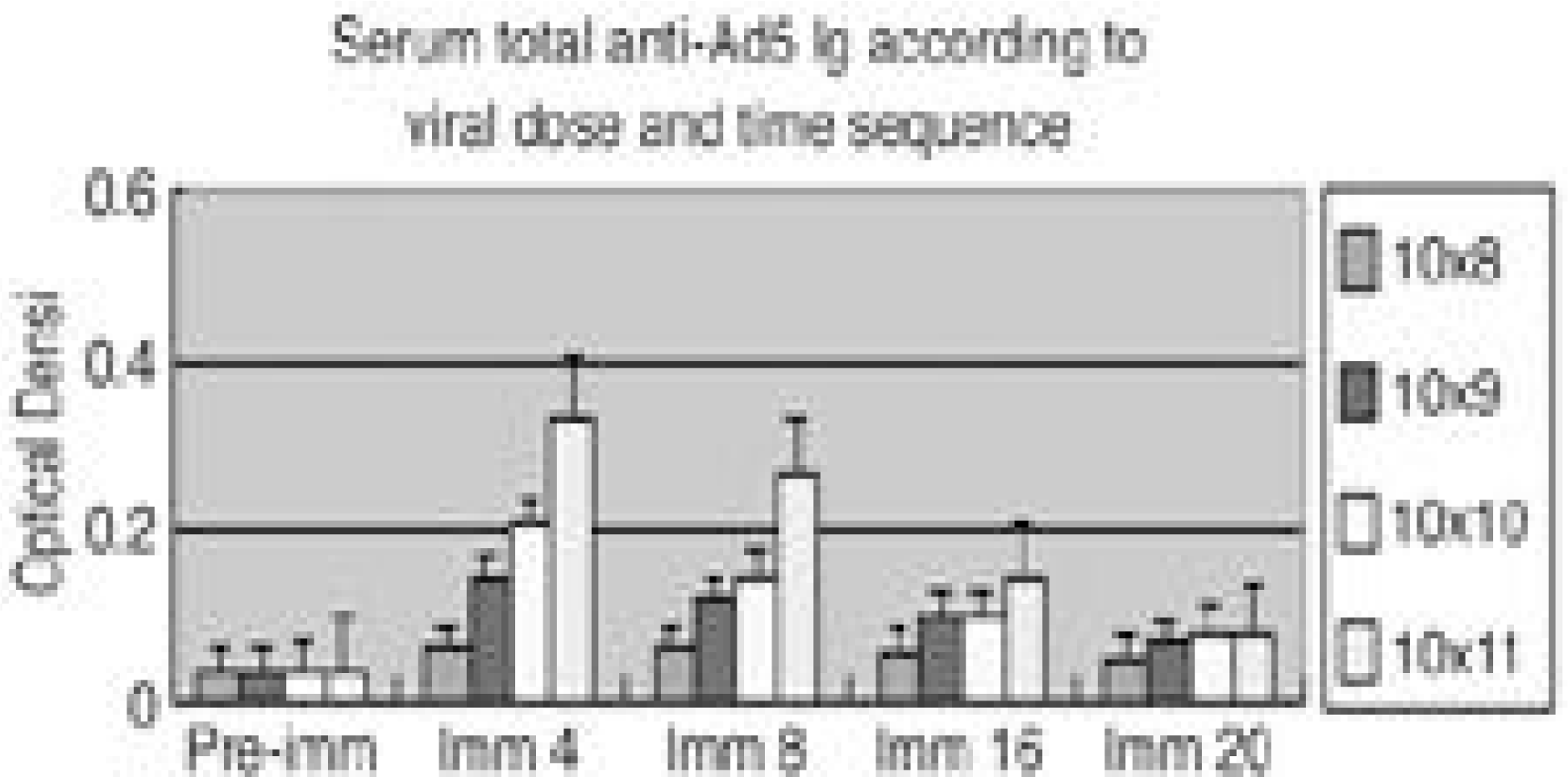 | Fig. 1.Kinetics of adenovirus specific antibody response according to viral dose and time sequence. NZW rabbit were immunized with i.v. injections of non-virus, 1×108, 1× 109, 1× 101 0, 1× 101 1 v.p. of Ad5-ßGal(CMV promotor). At the indicated times after administration, serum anti-adenovirus immune globulin were measured. Era bars indicated the standard deviation of the mean(n=5). |
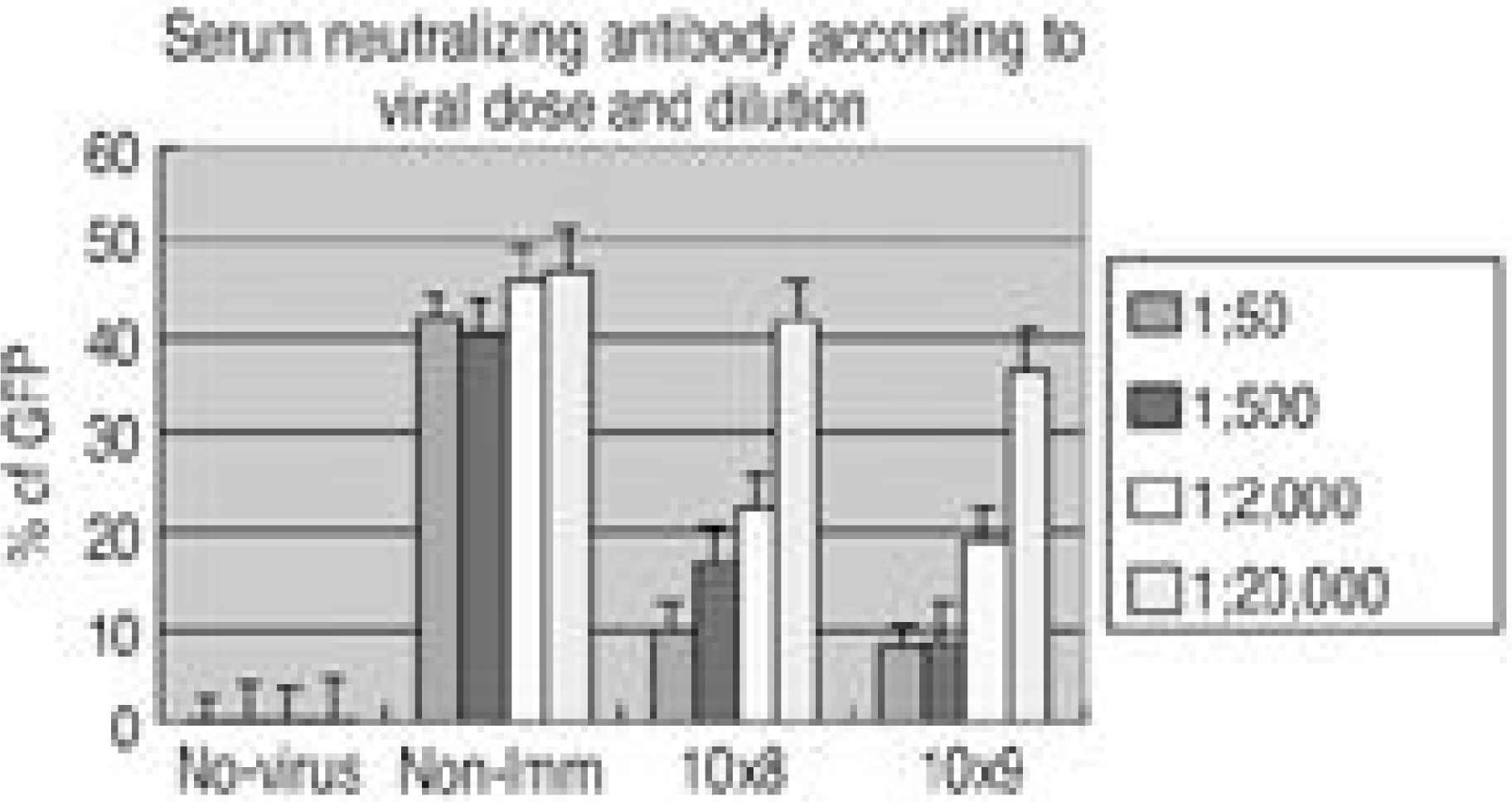 | Fig. 2.Inhibitory effects of immune-rabbit sera on adenovirus mediated in vitro infectivity. A293 cell were infected with Ad5-CMV-GFP △ E 1 at an 10 particle per unit(p.f.u.) per cell(10 M.O.I.), in the presence of serial dilution of serum, and the fluorescence emission at 538 nm recorded after infection. Titers are compared to the GFP fluorescence relative to the total recorded in absence of serum. The number of % GFP fluorescence infected with adenovirus with immunized sera was significantly smaller than that infected with adenovirus with non-immunized sera in 1:50, 500, 2,000 dilution. |
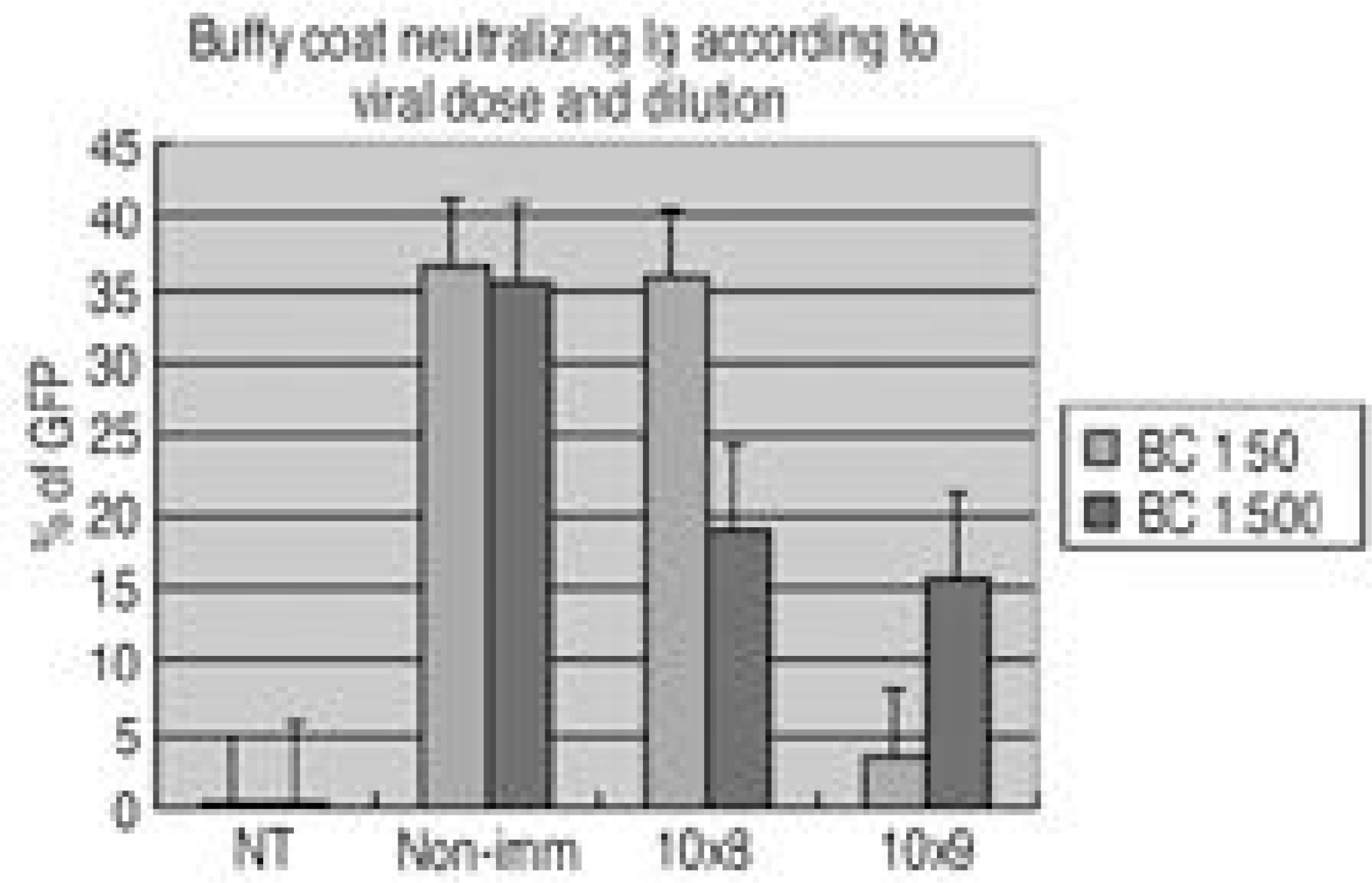 | Fig. 3.Inhibitory effects of immune-rabbit buffy coat on an adenovirus mediated in vitro infectivity using same method of figure 2. The number of % GFP fluorescence infected with adenovirus with immunized buffy coat was significantly smaller than infected with adenovirus with non-immunized buffy coat in 1:50 dilution in 1× 108 Ad5-ßGal and 1× 1 09 A-5ßGal. However, there was no significant difference between 1:500 dilution in 1× 108 Ad5-ßGal and 1× 109 ßGal immunized buffy coat. |
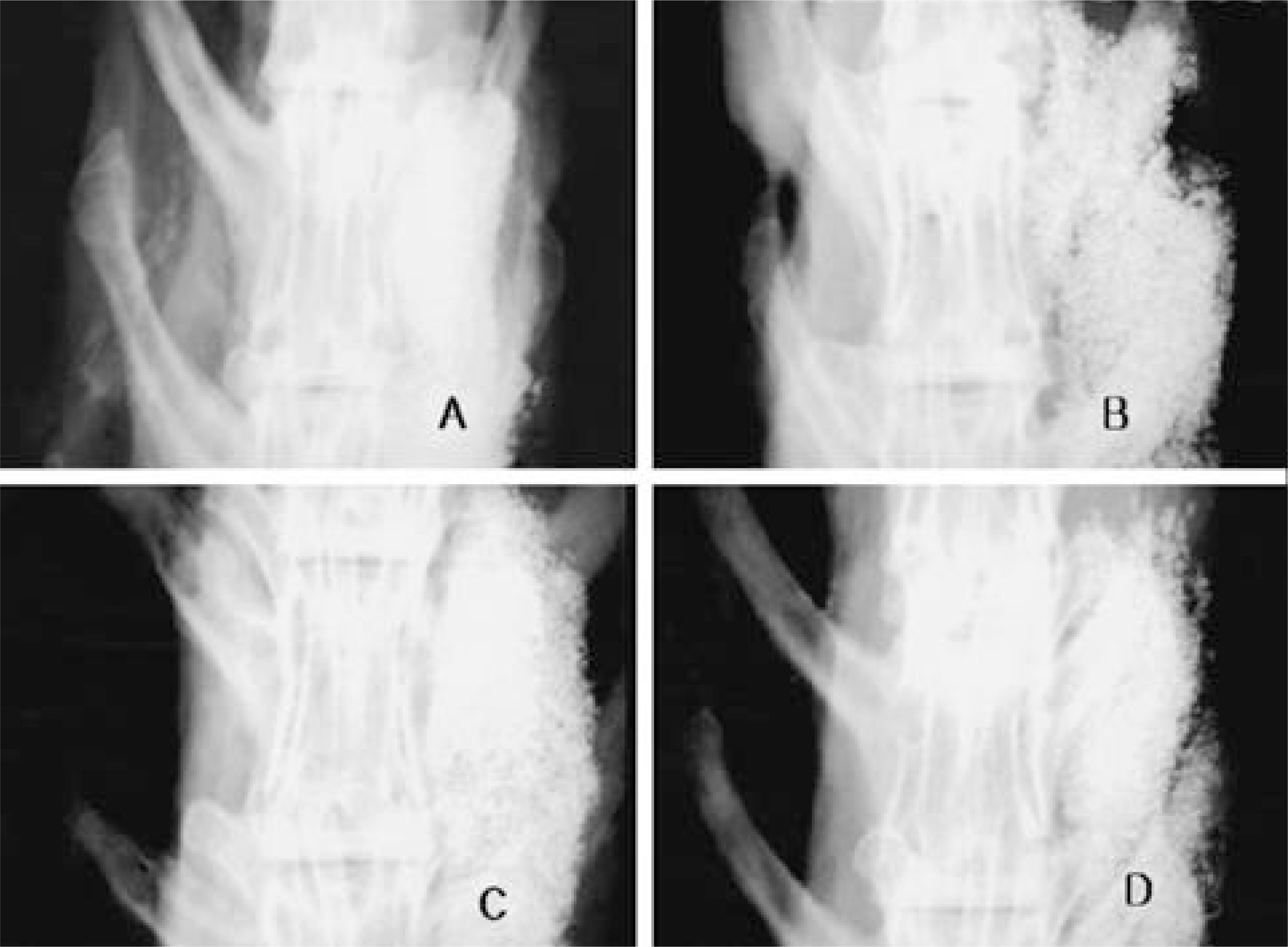 | Fig. 4.Comparison of plain radiography 4 weeks after immunization according to immunized viral doses. A is the 1× 108 v.p. immunized rabbit, B 1× 109 v.p., C 1× 1010 v.p., D 1 × 1011 v.p. Generally, the bone formation were not observed and small bone formation could be observed at the 1× 108 v.p. immunized rabbit. It represents that recent any viral doses effect strong inhibition |
 | Fig. 5.Comparison of plain radiography 16 weeks after immunization according to immunized viral doses. A is the 1× 108 v.p. immunized rabbit, B 1× 109 v.p., C 1× 1010 v.p., D 1× 1011 v.p. Generally, the more bone formation were observed in small viral doses(A, B) It represents that large viral doses effect more strong inhibition in bone formation. |
 | Fig. 6.Comparison of histology at 4 weeks and 16 weeks after immunization in 1× 108 Ad5-ßGal immunized group II. Left figure is the histology of 4 weeks after immunization. Right figure is the histology of 16 weeks after immunization. The lots of bone formation were observed in left, but right. It represents that time sequence blunted the impact of immune reaction. |
Table 1.
In vivo spine fusion rate in the NZW rabbit according to viral doses and time sequence.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download