Abstract
Study design
This is a mechanical study of polymethylmetacrylate(PMMA) mixed with blood as a filler.
Objective
We tried to change the properties of PMMA so that it is more suitable to use for percutaneous vertebroplasty (PVP).
Summary of the Literature Review
The mechanical changes by adding a filler into PMMA were expected to decrease the Young's modulus, the polymerization temperature and the setting time. These changes of PMMA were considered to be more suitable and adaptable conditions for PVP for treating osteoporotic vertebral compression fracture.
Materials and Methods
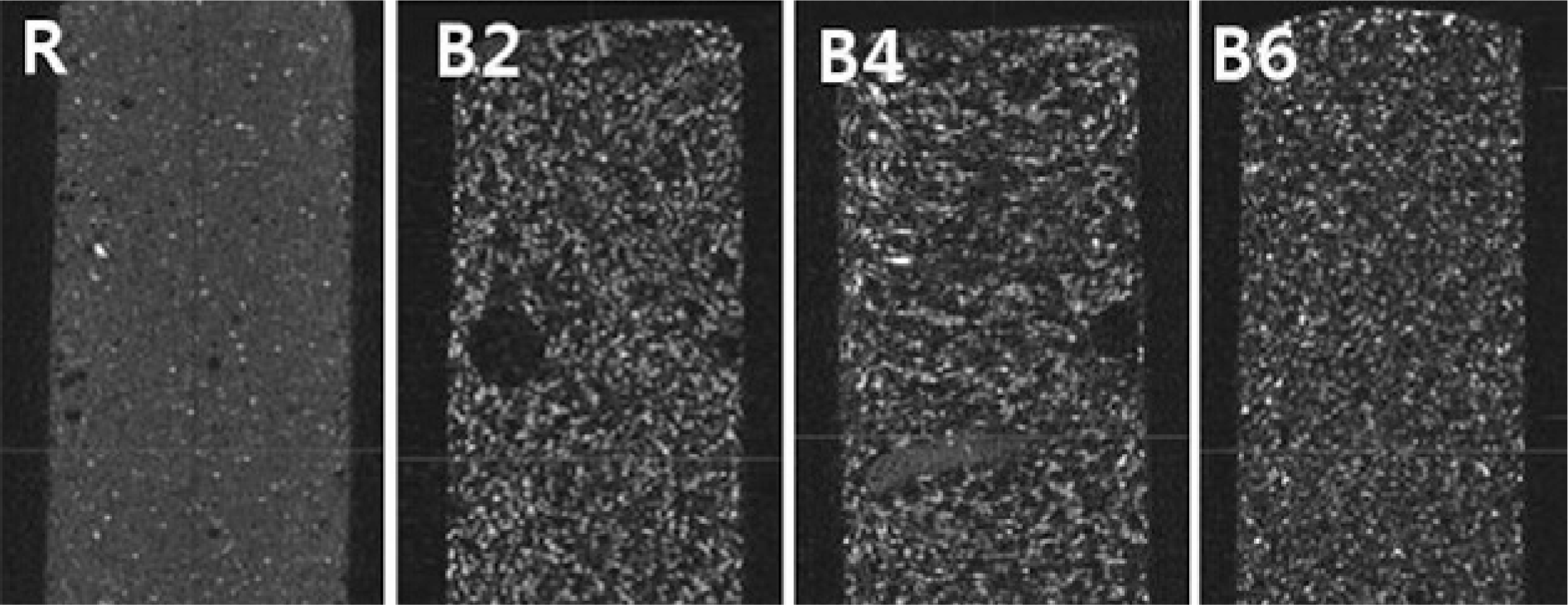
Porous PMMA was produced by mixing 2 ml (B2), 4 ml (B4) and 6 ml (B6)-blood as a filler, and the mechanical properties were investigated in comparison with regular PMMA(R) in view of Young's modulus, the polymerization temperature, the setting time and the optimal passing-time within the injectable viscosity (20~50N-needed) through a 2.8mm-diameter cement-filler tube. Porosity was inspected by performing microcomputated tomography (micro-CT).
Results
Young's modulus was decreased from 919.5 MPa (R) to 701 MPa (B2), 693.5 MPa (B4) and 545.6 MPa (B6) in each group. The polymerization temperature decreased from 74.2℃ (R) to 59.8℃ (B2), 54.2℃ (B4) and 47.5℃(B6), respectively. The setting time decreased from 1065sec (R) to 624sec (B2), 678sec(B4) and 606sec (B6), respectively, and the optimal passing-time decreased from 75.6sec (R) to 46.6sec (B2), 65.0sec (B4) and 79.0sec(B6), respectively. The porosity increased from 4.2%(R) to 27.6%(B2), 27.5%(B4) and 29.5%(B6), respectively. A homogenous microstructure with very fine pores was seen on inspection of all the blood-mixed PMMAs.
Conclusion
Blood mixed with PMMA was considered as an excellent filler that was easy to make and had good biocompatibility. The 6ml blood-mixed PMMA (B6) showed more suitable mechanical properties, including a decreased elastic modulus due to more porosity, less heating and a retarded optimal passing-time by the serum barrier, which diminished the friction between the PMMA and a cement-filler tube.
Go to : 
REFERENCES
01). Lin EP., Ekholm S., Hiwatashi A., Westesson PL. Vertebroplasty: Cement leakage into the disk increase the risk of new fracture of adjacent vertebral body. Am J Neruora-diol. 2004. 25:175–180.
02). Teng MM., Cheng H., Ho DM., Chang CY. Intraspinal leakage of bone cement after vertebroplasty: a report of 3 cases. Am J Neuroradiol. 2006. 27:224–229.
03). Chen JK., Lee HM., Shih JT., Hung ST. Combined extraforaminal and intradiscal cement leakage following percutaneous vertebroplasty. Spine. 2007. 32:358–362.

04). Lim SH., Kim H., Kim HK., Baek MJ. Multiple cardiac perforations and pulmonary embolism caused by cement leakage after percutaneous vertebroplasty. Eur J Cardio-thorac Surg. 2008. 33:510–512.

05). McLaren AC., McLaren SG., McLemore R., Vernon BL. Particle size of fillers affects permeability of polymethylmethacrylate. Clin Orthop Relat Res. 2007. 461:64–67.

06). Chiu FY., Lin CF. Antibiotic-impregnated cement in revision total knee arthroplasty. A prospective cohort study of one hundred and eighty-three knees. J Bone Joint Surg Am. 2009. 91:628–633.
07). Belkoff SM., Sanders JC., Jasper LE. The effect of the monomer-to-powder ratio on the material properties of acrylic bone cement. J Biomed Mater Res. 2002. 63:396–399.

08). Boger A., Bohner M., Heini P., Verrier S., Schneider E. Properties of an injectable low modulus PMMA bone cement for osteoporotic bone. J Biomed Mater Res B Appl Biomater. 2008. 86:474–482.

09). Loeffel M., Ferguson SJ., Nolte LP., Kowal JH. Vertebroplasty: experimental characterization of polymethyl-methacrylate bone cement spreading as a function of viscosity, bone porosity, and flow rate. Spine. 2008. 20:1352–1359.
10). Frankel BM., Monroe T., Wang C. Percutaneous vertebral augmentation: an elevation in adjacent-level fracture risk in kyphoplasty as compared with vertebroplasty. Spine J. 2007. 7:575–582.

11). Lin WC., Lee YC., Lee CH, et al. Refractures in cemented vertebrae after percutaneous vertebroplasty: a retrospective analysis. Eur Spine J. 2008. 17:592–599.

12). Hu X., Zhai X., Hirt T. A new concept for more biocompli-ant bone cements for vertebroplasty and kyphoplasty. Macromol Biosci. 2009. 9:195–202.

13). Grafe IA., Baier M., No¨ldge G, et al. Calcium-phosphate and polymethylmethacrylate cement in long-term outcome after kyphoplasty of painful osteoporotic vertebral fractures. Spine. 2008. 33:1284–1290.

14). Boger A., Bisig A., Bohner M., Heini P., Schneider E. Variation of the mechanical properties of PMMA to suit osteoporotic cancellous bone. J Biomater Sci Polym Ed. 2008. 19:1125–1142.

15). Hexip B., Nakaoka R., Tsuchiya T. Safety evaluation of surgical materials by cytotoxicity testing. Artif Organs. 2008. 11:204–211.
16). Boger A., Wheeler K., Montali A., Gruskin E. NMP-modified PMMA bone cement with adapted mechanical and hardening properties for the use in cancellous bone augmentation. J Biomed Mater Res B Appl Biomater. 2009. Early view (Published-Online).

17). Baroud G., Crookshank M., Bohner M. High-viscosity cement significantly enhances uniformity of cement filling in vertebroplasty: an experimental model and study on cement leakage. Spine. 2006. 31:2562–2568.

18). Gravius S., Kraska N., Maus U., Mumme T., Berdel P., Wei�kopf M. Intravertebral Pressure during Vertebroplasty - An in-vitro Study. Z Orthop Unfall. 2009. 147:43–47.
Go to : 
 | Fig. 1.Measurement methods of pressure-related manipulation time. A cement filler with 2.8 mm of diameter is connected with FGP-5. A jig can hold a cement filler tightly along the parallel axis to FGP-5 not to cause unexpected resistant pressure during cement-pushing. |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download