Abstract
The efficacy and safety of a single tablet regimen (STR) of elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil fumarate (E/C/F/TDF) were analyzed in Phase 3 clinical trials in antiretroviral therapy (ART)-naïve and ART-experienced Asian subjects infected with human immunodeficiency virus (HIV)-1. Studies GS-US-236-102 and GS-US-236-103 were randomized, double-blind, placebo-controlled, 144-week studies conducted in ART-naïve subjects, comparing E/C/F/TDF versus efavirenz (EFV)/F/TDF or ritonavir-boosted atazanavir (ATV+RTV) plus emtricitabine/tenofovir DF (F/TDF), respectively. Studies GS-US-236-115 and GS-US-236-121 were randomized, open-label, 96-week long conducted in ART-experienced subjects, who switched to E/C/F/TDF from ritonavir-boosted protease inhibitors (PI+RTV)+F/TDF, or non-nucleoside reverse transcriptase inhibitors (NNRTI)+F/TDF regimens. The E/C/F/TDF appeared to have sustained efficacy and safety and was well tolerated in the small number of ART-naïve and ART-experienced Asian subjects.
The single tablet regimen (STR) containing elvitegravir, cobicistat, emtricitabine and tenofovir disoproxil fumarate (E/C/F/TDF) is a recommended regimen in the guidelines of the US Department of Health and Human Services and the European acquired immunodeficiency syndrome (AIDS) Clinical Society [12]. In two Phase 3 randomized, double-blind, placebo-controlled clinical trials in ART-naïve adults infected with the human immunodeficiency virus (HIV)-1, E/C/F/TDF (n = 701) demonstrated non-inferior efficacy at week 48, 96, and 144 compared to the STR of efavirenz (EFV)/F/TDF (GS-US-236-0102, Study 102) and the ritonavir-boosted atazanavir (ATV+RTV) plus emtricitabine/tenofovir DF (F/TDF, GS-US-236-0103, Study 103) as well as favorable safety and tolerability [3456789]. Studies GS-US-236-115 (STRATEGY-PI) and GS-US-236-121 (STRATEGY-NNRTI) examined the efficacy, safety, and tolerability of switching to E/C/F/TDF from ritonavir-boosted protease inhibitors (PI+RTV)+ F/TDF or non-nucleoside reverse transcriptase inhibitors (NNRTI)+F/TDF regimens, respectively, in virologically suppressed (HIV-1 RNA<50 copies/mL), ART-experienced adult subjects [10111213]. At week 48 and 96, the STRATEGY-PI study showed that switching to E/C/F/TDF from a PI+RTV-based regimen was associated with significantly higher rates of virological success, lower triglyceride levels, and improvements in self-reported diarrhea and bloating[14]. The STRATEGY-NNRTI study showed the non-inferior efficacy of E/C/F/TDF versus remaining on the NNRTI+ F/TDF regimen, improvement in patient-reported outcomes (PROs) related to NNRTI-associated neuropsychiatric side effects, and greater treatment satisfaction scores [15]. There is limited data on the efficacy and safety of current antiretroviral therapies in Asian subjects infected with HIV-1. Here, we report a sub-analysis of E/C/F/TDF efficacy and safety data in Asian subjects enrolled in Studies 102 and 103 at week 144 as well as Studies 115 and 121 at week 96.
In the two ART-naïve studies, 1,408 subjects (E/C/F/TDF, n = 701 vs. EFV/FTC/TDF, n = 352 vs. ATV+RTV+ F/TDF, n = 355) were enrolled and received at least one dose of a study drug. In the two studies with ART-experienced, virologically suppressed subjects, 867 (Study 115: E/C/F/TDF, n = 293 vs. PI+RTV+ F/TDF, n = 140 and Study 121: E/C/F/TDF, n = 291 vs. NNRTI+ F/TDF, n=143) were enrolled and received at least one dose of a study drug. In these four clinical trials, 72 Asian subjects consisting of 50 ART-naïve (E/C/F/TDF, n=23; EFV/F/TDF, n = 10; and ATV+RTV+ F/TDF, n = 17) and 22 ART-experienced, virologically suppressed (Study 115: E/C/F/TDF, n = 7 and PI+RTV+ F/TDF, n=2; and Study 121: E/C/F/TDF and n = 4; NNRTI+ F/TDF, n = 9) were included in this sub-analysis of the E/C/F/TDF data [16].
The baseline demographics and disease characteristics of the ART-naïve (Studies 102 and 103, pooled) and ART-experienced (Study 115 and Study 121, separately) subjects on E/C/F/TDF were as follows: median age 33, 33, and 45 years; male: 83%, 71%, and 100%; mean CD4 count: 374, 548, and 402 cells/mm3; and median estimated glomerular filtration rate (eGFR) using the Cockcroft-Gault method: 100, 104, and 125 mL/min, respectively (Table 1).
Similar high rates of virological success (HIV-RNA <50 copies/mL, FDA Snapshot Analysis) and comparable immunological outcomes were observed in the ART-naïve and ART-experienced subjects on E/C/F/TDF. In the ART-naïve subjects, the virological success (HIV-1 RNA <50 copies/mL) was 91, 80, and 76% with E/C/F/TDF, EFV/F/TDF, ATV+RTV+ F/TDF, respectively at week 144. The mean CD4+ T cell count (/mm3) increased from baseline through week 144 (235, E/C/F/TDF; 161, EFV/F/TDF; and 250, ATV+RTV+ F/TDF). In the ART-experienced, virologically suppressed subjects, the virological success (maintenance of HIV-1 RNA<50 copies/mL) of the E/C/F/TDF regimen was 86 and 100% (Studies 115 and 121, respectively) at week 96. The virological success rates for subjects who remained on the PI+RTV+ F/TDF or NNRTI+ F/TDF regimens were 100 and 67%, respectively, both at week 96. The mean CD4+ T cell count increases were similar for E/C/F/TDF in both Studies 115 and 121: 61 and 71 cells/mm3 compared to -30 and 162 cells/mm3 for patients who remained on the PI+RTV+ F/TDF and NNRTI+ F/TDF regimens, respectively (Fig. 1).
The overall safety and tolerability of E/C/F/TDF in both ART-naïve and ART-experienced subjects were similar to that of E/C/F/TDF in the overall populations enrolled in the four studies. In the ART naïve subjects, the most common study drug-related adverse events (AEs) with E/C/F/TDF were nausea (n = 4 vs. EFV/F/TDF, n = 0 and ATV+RTV+ F/TDF, n = 5), abnormal dreams (n = 2 vs. 5 and 1, respectively), diarrhea (n = 2 vs. 0 and 3, respectively) and dizziness (n = 1 vs. 2 and 2, respectively). Grade 3AEs occurred in subjects with similar frequency between E/C/F/TDF (17%, n = 4) and EFV/F/TDF (20%, n = 2) while no subjects in the ATV+RTV+ F/TDF treatment arm experienced this Grade. Furthermore, there were no Grade 4 AEs. There were few study drug discontinuations due to AEs in the ART-naïve subjects and the frequency was similar between the treatment arms [E/C/F/TDF, n = 1 due to lymphoma; EFV/F/TDF, n=1 due to presyncope; and ATV+RTV+ F/TDF, n = 2 due to gastrointestinal disorder consisting of diarrhea (one subject) and nausea, vomiting, and flatulence (one subject, who also had dizziness)]. In the ART-experienced subjects, the most common study drug-related AEs with E/C/F/TDF in Study 115 were, enlarged parotid gland and alopecia (n = 1 each) and in Study 121, increased weight (n = 1) and GFR (n = 1 in male, 47 years old, observed as Grade 1). Only one Grade 3-4 AE occurred in the ART-experienced subjects, which was a subject with a clavicle fracture treated on E/C/F/TDF. There were no subjects on E/C/F/TDF in the ART-experienced studies that discontinued the study drug because of AEs and no subjects in either the ART-naïve or ART-experienced discontinued E/C/F/TDF because of renal AEs. The treatment emergent Grade 3-4 laboratory abnormalities observed with E/C/F/TDF were all Grade 3 (alanine transaminase [ALT], n = 2; aspartate transaminase [AST], n = 1; and hyperuricemia, n = 1) in ART-experienced subjects.
Cobicistat induced a slight increase in serum creatinine (SCr) with a consequent reduction in the eGFR in Phase 1 and 2 clinical trials [171819]. The changes in SCr are caused by the inhibition of tubular creatinine secretion with no effect on the actual glomerular filtration rate, as measured by the clearance of iohexol [18]. In this sub-analysis in Asian subjects, the median changes from baseline in SCr were similar to that observed in the overall study population on E/C/F/TDF. The median change from baseline in the SCr of the ART-naïve subjects was 0.14 mg/dL with E/C/F/TDF vs. -0.03 mg/dL with EFV/F/TDF and 0.04 mg/dL with ATV+RTV+F/TDF. Furthermore, the values for the ART-experienced subjects were 0.06 mg/dL with E/C/F/TDF vs. -0.18 mg/dL with PI+RTV+F/TDF (Study 115) and 0.05 mg/dL with both E/C/F/TDF and NNRTI+F/TDF (Study 121).
In Asian subjects, the median changes from baseline in the fasting lipid parameters [total cholesterol (TC) and high-density lipoprotein (HDL)] were slight with E/C/F/TDF, which resulted in minimal median changes in the TC:HDL ratio [ART-naïve (0.1) and ART-experienced (Studies 115 and 121, 0.2 and 0.0, respectively)] (Fig. 2)
A limitation of this sub-analysis is the small number of Asian subjects, who accounted for 4 and 3% of the overall population of the participants enrolled in the ART-naïve and ART-experienced studies. This restricted the definitive assessment of the safety and tolerability of E/C/F/TDF in Asian subjects, and may limit generalization of the results.
In these sub-studies of Phase 3 clinical trials of E/C/F/TDF, the Asian subjects with HIV-1 infections who were either initiating E/C/F/TDF therapy or switching to E/C/F/TDF from RTV-boosted PIs or NNRTIs (both combined with F/TDF) demonstrated a high (>90%) efficacy (HIV-1 RNA<50 copies/mL). The safety and tolerability profile of E/C/F/TDF in the Asian subjects was similar to that of E/C/F/TDF in the overall study populations investigated [891213]. No new or unique safety concerns were observed in the ART-naïve and ART-experienced Asian subjects on E/C/F/TDF. The AEs leading to study drug discontinuation were uncommon with no renal AEs leading to discontinuation with E/C/F/TDF. Changes in the SCr of Asian subjects on E/C/F/TDF were consistent with those observed in the overall populations enrolled in the ART-naïve and ART-experienced studies. The changes from baseline in SCr observed at week 144 and 96 (ART-naïve and ART-experienced, respectively) were similar to those observed from baseline to week 48, which suggests the initial increase in SCr with E/C/F/TDF was caused by the inhibitory effect of cobicistat on the renal tubular creatinine secretion, which then stabilized [20]. The slight changes in the lipid parameters in Asian subjects on E/C/F/TDF resulted in minimal changes in the TC:HDL ratio and were similar to those in the overall study population. In summary, from this sub-analysis in a small number of Asian subjects, E/C/F/TDF appears to have sustained efficacy and is safe and well-tolerated based on the available data in both the ART-naïve and ART-experienced Asian subjects.
Figures and Tables
Figure 1
Virological and immunological outcomes studies 102 and 103: Asian subset through week 144
HIV, human immunodeficiency virus; RNA, ribonucleic acid; FDA, food and drug administration; E, elvitegravir; C, cobicistat; F, emtricitabine; TDF, tenofovir disoproxil fumarate; EFV, efavirenz; FTC, emtricitabine; ATV, atazanavir; RTV, ritonavir; TVD, truvada; PI, protease inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor
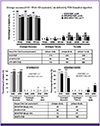
Figure 2
Median change in fasting lipids from baseline
TC, total cholesterol; LDL, low-density lipoprotein; TG, triglycerides; HDL, high-density lipoprotei
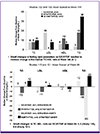
Table 1
Baseline Characteristics: Asian Subpopulation Studies

aOther: E/C/F/TDF; Australia (2); ATV+RTV+TVD; Australia (7); Canada (2)
bOther: E/C/F/TDF: Switzerland (1); NNRTI+TVD: Australia (1)
E, elvitegravir; C, cobicistat; F, emtricitabine; TDF, tenofovir disoproxil fumarate; EFV, efavirenz; ATV, atazanavir; RTV, ritonavir; PI, protease inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; HBV, hepatitis B virus; HCV, hepatitis C virus; HIV, human immunodeficiency virus; GFR, glomerular filtration rate
Acknowledgments
The authors would like to acknowledge and thank the patients who participated in these studies, as well as the site and study management staff whose effort made this study possible. All investigators and sites that participated in these studies have previously published their respective results [3-15].
References
1. Department for Health and Human Services (DHHS). Guidelines for the Use of Antiretroviral Agents in HIV-1 Infected Adults and Adolescents. Accessed 28 January 2016. Available at: https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-treatment-guidelines/0.
2. European AIDS Clinical Society (EACS). EACS Guidelines Version 8.0. Accessed 10 January 2016. Available at: http://www.eacsociety.org/guidelines/eacs-guidelines/eacs-guidelines.html.
3. Sax PE, DeJesus E, Mills A, Zolopa A, Cohen C, Wohl D, Gallant JE, Liu HC, Zhong L, Yale K, White K, Kearney BP, Szwarcberg J, Quirk E, Cheng AK. GS-US-236-0102 study team. Co-formulated elvitegravir, cobicistat, emtricitabine, and tenofovir versus co-formulated efavirenz, emtricitabine, and tenofovir for initial treatment of HIV-1 infection: a randomised, double-blind, phase 3 trial, analysis of results after 48 weeks. Lancet. 2012; 379:2439–2448.

4. DeJesus E, Rockstroh JK, Henry K, Molina JM, Gathe J, Ramanathan S, Wei X, Yale K, Szwarcberg J, White K, Cheng AK, Kearney BP. GS-236-0103 Study Team. Co-formulated elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate versus ritonavir-boosted atazanavir plus co-formulated emtricitabine and tenofovir disoproxil fumarate for initial treatment of HIV-1 infection: a randomised, double-blind, phase 3, non-inferiority trial. Lancet. 2012; 379:2429–2438.

5. Zolopa A, Sax PE, DeJesus E, Mills A, Cohen C, Wohl D, Gallant JE, Liu HC, Plummer A, White KL, Cheng AK, Rhee MS, Szwarcberg J. GS-US-236-0102 Study Team. A randomized double-blind comparison of coformulated elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil fumarate versus efavirenz/emtricitabine/tenofovir disoproxil fumarate for initial treatment of HIV-1 infection: analysis of week 96 results. J Acquir Immune Defic Syndr. 2013; 63:96–100.

6. Rockstroh JK, DeJesus E, Henry K, Molina JM, Gathe J, Ramanathan S, Wei X, Plummer A, Abram M, Cheng AK, Fordyce MW, Szwarcberg J. GS-236-0103 Study Team. A randomized, double-blind comparison of co-formulated elvitegravir/cobicistat/emtricitabine/tenofovir versus ritonavir-boosted atazanavir plus co-formulated emtricitabine and tenofovir DF for initial treatment of HIV-1 infection: analysis of week 96 results. J Acquir Immune Defic Syndr. 2013; 62:483–486.

7. Rockstroh J. A randomized, double-blind comparison of coformulated elvitegravir/cobicistat/emtricitabine/tenofovir DF vs ritonavir-boosted atazanavir plus coformulated emtricitabine and tenofovir DF for initial treatment of HIV-1 infection: analysis of week 96 results. [erratum]. J Acquir Immune Defic Syndr. 2013; 63:e171.
8. Wohl DA, Cohen C, Gallant JE, Mills A, Sax PE, Dejesus E, Zolopa A, Liu HC, Plummer A, White KL, Cheng AK, Rhee MS, Szwarcberg J. GS-US-236-0102 Study Team. A randomized, double-blind comparison of single-tablet regimen elvitegravir/cobicistat/emtricitabine/tenofovir DF versus single-tablet regimen efavirenz/emtricitabine/renofovir DF for initial treatment of HIV-1 infection: analysis of week 144 results. J Acquir Immune Defic Syndr. 2014; 65:e118–20.
9. Clumeck N, Molina JM, Henry K, Gathe J, Rockstroh JK, DeJesus E, Wei X, White K, Fordyce MW, Rhee MS, Szwarcberg J. GS-236-0103 Study Team. A randomized, double-blind comparison of single-tablet regimenelvitegravir/cobicistat/emtricitabine/tenofovir DF vs ritonavir-boosted atazanavir plusemtricitabine/tenofovir DF for initial treatment of HIV-1 infection: analysis of week144 results. J Acquir Immune Defic Syndr. 2014; 65:e121–4.
10. Arribas JR, Pialoux G, Gathe J, Di Perri G, Reynes J, Tebas P, Nguyen T, Ebrahimi R, White K, Piontkowsky D. Simplification to coformulated elvitegravir, cobicistat, emtricitabine, and tenofovir versus continuation of ritonavir-boosted protease inhibitor with emtricitabine and tenofovir in adults with virologically suppressed HIV (STRATEGY-PI): 48 week results of a randomised, open-label, phase 3b, non-inferiority trial. Lancet Infect Dis. 2014; 14:581–589.

11. Pozniak A, Markowitz M, Mills A, Stellbrink HJ, Antela A, Domingo P, Girard PM, Henry K, Nguyen T, Piontkowsky D, Garner W, White K, Guyer B. Switching to coformulated elvitegravir, cobicistat, emtricitabine, and tenofovir versus continuation of non-nucleoside reverse transcriptase inhibitor with emtricitabine and tenofovir in virologically suppressed adults with HIV (STRATEGY-NNRTI): 48 week results of a randomised, open-label, phase 3b non-inferiority trial. Lancet Infect Dis. 2014; 14:590–599.

12. In : Arribas J, DeJesus E, Van Lunzen J, Zurawski C, Doroana M, Towner W, Lazzarin A, Nelson M, Benn P, Chu H, McColl D, Swamy R, Nguyen T, editors. Simplification to the STRIBILD single tablet regimen from PI + RTV + FTC/TDF multi-pill regimens maintains durable HIV suppression: week 96 results of STRATEGY-PI (Study 115) [Poster P1]. 21st Annual Conference of the British HIV Association; 2015 April 21-24; Brighton, UK.
13. In : Pozniak A, Flamm J, Antinori A, Bloch M, Ward D, Berenguer J, Cote P, Smith A, Andreatta K, Garner W, Szwarcberg J, Piontkowsky D, editors. Switch to STRIBILD from NNRTI plus FTC/TDF regimens maintains HIV suppression and is well-tolerated: week 96 results of STRATEGY-NNRTI (Study 121) [Poster P5]. 21st Annual Conference of the British HIV Association; 2015 April 21-24; Brighton, UK.
14. Gathe J, Arribas JR, Van Lunzen J, Garner W, Speck RM, Bender R, Shreay S, Nguyen T. Patient-reported symptoms over 48 weeks in a randomized, open-label, phase 3b non-inferiority rrial of adults with HIV switching to coformulated elvitegravir, cobicistat, emtricitabine, and tenofovir DF versus continuation of ritonavir-boosted protease inhibitor with emtricitabine and tenofovir DF. Patient. 2015; 8:445–454.

15. Mills A, Garner W, Pozniak A, Berenguer J, Speck RM, Bender R, Nguyen T. Patient-reported symptoms over 48 weeks in a randomized, open-label, phase IIIb non-inferiority trial of adults with HIV switching to co-formulated elvitegravir, cobicistat, emtricitabine, and tenofovir DF versus continuation of non-nucleoside reverse transcriptase inhibitor with emtricitabine and tenofovir DF. Patient. 2015; 8:359–371.

16. In : Choi JY, Sungkanuparaph S, Anekthananon T, Sax P, DeJesus E, Edelstein H, Nelson M, DeMorin J, Liu H, Swamy R, Bahn J, Ng C, Piontkowsky D, editors. Efficacy and safety of elvitegravir/cobicistat/emtricitabine/tenofovir DF in HIV-1 infected, Asian subjects: a sub-analysis of phase 3 clinical trials [Presentation OB-6]. Interscience Conference on Infection and Chemotherapy (ICIC); 2015 November 5-7; Seoul, Korea.
17. Cohen C, Elion R, Ruane P, Shamblaw D, DeJesus E, Rashbaum B, Chuck SL, Yale K, Liu HC, Warren DR, Ramanathan S, Kearney BP. Randomized, phase 2 evaluation of two single-tablet regimens elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil fumarate versus efavirenz/emtricitabine/tenofovir disoproxil fumarate for the initial treatment of HIV infection. AIDS. 2011; 25:F7–12.

18. German P, Lui C, Warren D, Hepner-Harris M, Andrews J, Kearney BP, Mathias A. Effect of cobicistat on glomerular filtration rate (GFR) in subjects with normal and impaired renal function [Poster H2-804]. In : 51st Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); 2011 September 17-20; Chicago, IL, USA.
19. Elion R, Cohen C, Gathe J, Shalit P, Hawkins T, Liu HC, Mathias AA, Chuck SL, Kearney BP, Warren DR. GS-US-216-0105 Study Team. Phase 2 study of cobicistat versus ritonavir each with once-daily atazanavir and fixed-dose emtricitabine/tenofovir df in the initial treatment of HIV infection. AIDS. 2011; 25:1881–1886.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download