1. Fischbach MA, Walsh CT. Antibiotics for emerging pathogens. Science. 2009; 325:1089–1093.

2. McDonald LC, Killgore GE, Thompson A, Owens RC Jr, Kazakova SV, Sambol SP, Johnson S, Gerding DN. An epidemic, toxin gene-variant strain of
Clostridium difficile. N Engl J Med. 2005; 353:2433–2441.

3. Dorman SE, Chaisson RE. From magic bullets back to the magic mountain: the rise of extensively drug-resistant tuberculosis. Nat Med. 2007; 13:295–298.

4. Munoz-Price LS, Weinstein RA. Acinetobacter infection. N Engl J Med. 2008; 358:1271–1281.

5. Oliver A, Canton R, Campo P, Baquero F, Blázquez J. High frequency of hypermutable
Pseudomonas aeruginosa in cystic fibrosis lung infection. Science. 2000; 288:1251–1254.

6. Lautenbach E, Synnestvedt M, Weiner MG, Bilker WB, Vo L, Schein J, Kim M. Imipenem resistance in
Pseudomonas aeruginosa: emergence, epidemiology, and impact on clinical and economic outcomes. Infect Control Hosp Epidemiol. 2010; 31:47–53.

7. Garner MJ, Carson C, Lingohr EJ, Fazil A, Edge VL, Trumble Waddell J. An assessment of antimicrobial resistant disease threats in Canada. PLoS One. 2015; 10:e0125155.

8. Rosenthal VD, Bijie H, Maki DG, Mehta Y, Apisarnthanarak A, Medeiros EA, Leblebicioglu H, Fisher D, Álvarez-Moreno C, Khader IA, Del Rocío González Martínez M, Cuellar LE, Navoa-Ng JA, Abouqal R, Guanche Garcell H, Mitrev Z, Pirez García MC, Hamdi A, Dueñas L, Cancel E, Gurskis V, Rasslan O, Ahmed A, Kanj SS, Ugalde OC, Mapp T, Raka L, Yuet Meng C, Thu le TA, Ghazal S, Gikas A, Narváez LP, Mejía N, Hadjieva N, Gamar Elanbya MO, Guzmán Siritt ME, Jayatilleke K. INICC members. International Nosocomial Infection Control Consortium (INICC) report, data summary of 36 countries, for 2004-2009. Am J Infect Control. 2012; 40:396–407.

9. Schneider H, Geginat G, Hogardt M, Kramer A, Dürken M, Schroten H, Tenenbaum T.
Pseudomonas aeruginosa outbreak in a pediatric oncology care unit caused by an errant water jet into contaminated siphons. Pediatr Infect Dis J. 2012; 31:648–650.

10. Nagao M, Iinuma Y, Igawa J, Saito T, Yamashita K, Kondo T, Matsushima A, Takakura S, Takaori-Kondo A, Ichiyama S. Control of an outbreak of carbapenem-resistant
Pseudomonas aeruginosa in a haemato-oncology unit. J Hosp Infect. 2011; 79:49–53.

11. Caselli D, Cesaro S, Ziino O, Zanazzo G, Manicone R, Livadiotti S, Cellini M, Frenos S, Milano GM, Cappelli B, Licciardello M, Beretta C, Aricò M, Castagnola E. Infection Study Group of the Associazione Italiana Ematologia Oncologia Pediatrica (AIEOP). Multidrug resistant
Pseudomonas aeruginosa infection in children undergoing chemotherapy and hematopoietic stem cell transplantation. Haematologica. 2010; 95:1612–1615.

12. Bergen PJ, Bulitta JB, Forrest A, Tsuji BT, Li J, Nation RL. Pharmacokinetic/pharmacodynamic investigation of colistin against
Pseudomonas aeruginosa using an
in vitro model. Antimicrob Agents Chemother. 2010; 54:3783–3789.

13. Johansen HK, Moskowitz SM, Ciofu O, Pressler T, Høiby N. Spread of colistin resistant non-mucoid
Pseudomonas aeruginosa among chronically infected Danish cystic fibrosis patients. J Cyst Fibros. 2008; 7:391–397.

14. Zavascki AP, Carvalhaes CG, Picão RC, Gales AC. Multidrug-resistant
Pseudomonas aeruginosa and
Acinetobacter baumannii: resistance mechanisms and implications for therapy. Expert Rev Anti Infect Ther. 2010; 8:71–93.

15. Queenan AM, Bush K. Carbapenemases: the versatile beta-lactamases. Clin Microbiol Rev. 2007; 20:440–458. table of contents.
16. Kitao T, Tada T, Tanaka M, Narahara K, Shimojima M, Shimada K, Miyoshi-Akiyama T, Kirikae T. Emergence of a novel multidrug-resistant
Pseudomonas aeruginosa strain producing IMP-type metallo-beta-lactamases and AAC(6')-Iae in Japan. Int J Antimicrob Agents. 2012; 39:518–521.

17. Poirel L, Nordmann P, Lagrutta E, Cleary T, Munoz-Price LS. Emergence of KPC-producing
Pseudomonas aeruginosa in the United States. Antimicrob Agents Chemother. 2010; 54:3072.

18. Wang C, Cai P, Chang D, Mi Z. A
Pseudomonas aeruginosa isolate producing the GES-5 extended-spectrum beta-lactamase. J Antimicrob Chemother. 2006; 57:1261–1262.

19. Lee K, Lim JB, Yum JH, Yong D, Chong Y, Kim JM, Livermore DM.
blaVIM-2 cassette-containing novel integrons in metallo-beta-lactamase-producing
Pseudomonas aeruginosa and
Pseudomonas putida isolates disseminated in a Korean hospital. Antimicrob Agents Chemother. 2002; 46:1053–1058.

20. Jovcic B, Lepsanovic Z, Suljagic V, Rackov G, Begovic J, Topisirovic L, Kojic M. Emergence of NDM-1 metallo-beta-lactamase in
Pseudomonas aeruginosa clinical isolates from Serbia. Antimicrob Agents Chemother. 2011; 55:3929–3931.

21. Potron A, Poirel L, Nordmann P. Plasmid-mediated transfer of the
blaNDM-1 gene in Gram-negative rods. FEMS Microbiol Lett. 2011; 324:111–116.

22. Yezli S, Shibl AM, Memish ZA. The molecular basis of beta-lactamase production in Gram-negative bacteria from Saudi Arabia. J Med Microbiol. 2015; 64:127–136.

23. Martins AF, Zavascki AP, Gaspareto PB, Barth AL. Dissemination of
Pseudomonas aeruginosa producing SPM-1-like and IMP-1-like metallo-beta-lactamases in hospitals from southern Brazil. Infection. 2007; 35:457–460.

24. Sevillano E, Gallego L, García-Lobo JM. First detection of the OXA-40 carbapenemase in
P. aeruginosa isolates, located on a plasmid also found in
A. baumannii. Pathol Biol (Paris). 2009; 57:493–495.

25. El Garch F, Bogaerts P, Bebrone C, Galleni M, Glupczynski Y. OXA-198, an acquired carbapenem-hydrolyzing class D beta-lactamase from
Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2011; 55:4828–4833.

26. Mendes RE, Bell JM, Turnidge JD, Castanheira M, Jones RN. Emergence and widespread dissemination of OXA-23, -24/40 and -58 carbapenemases among
Acinetobacter spp. in Asia-Pacific nations: report from the SENTRY Surveillance Program. J Antimicrob Chemother. 2009; 63:55–59.

27. Voor In 't Holt AF, Severin JA, Lesaffre EM, Vos MC. A systematic review and meta-analyses show that carbapenem use and medical devices are the leading risk factors for carbapenem-resistant Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2014; 58:2626–2637.
28. Yoneyama H, Nakae T. Mechanism of efficient elimination of protein D2 in outer membrane of imipenem-resistant
Pseudomonas aeruginosa. Antimicrob Agents Chemother. 1993; 37:2385–2390.

29. Borgianni L, Prandi S, Salden L, Santella G, Hanson ND, Rossolini GM, Docquier JD. Genetic context and biochemical characterization of the IMP-18 metallo-beta-lactamase identified in a
Pseudomonas aeruginosa isolate from the United States. Antimicrob Agents Chemother. 2011; 55:140–145.

30. Hocquet D, Plésiat P, Dehecq B, Mariotte P, Talon D, Bertrand X. ONERBA. Nationwide investigation of extended-spectrum beta-lactamases, metallo-beta-lactamases, and extended-spectrum oxacillinases produced by ceftazidime-resistant
Pseudomonas aeruginosa strains in France. Antimicrob Agents Chemother. 2010; 54:3512–3515.

31. Koh TH, Khoo CT, Tan TT, Arshad MA, Ang LP, Lau LJ, Hsu LY, Ooi EE. Multilocus sequence types of carbapenem-resistant
Pseudomonas aeruginosa in Singapore carrying metallo-beta-lactamase genes, including the novel
blaIMP-26 gene. J Clin Microbiol. 2010; 48:2563–2564.

32. Quale J, Bratu S, Gupta J, Landman D. Interplay of efflux system, ampC, and oprD expression in carbapenem resistance of
Pseudomonas aeruginosa clinical isolates. Antimicrob Agents Chemother. 2006; 50:1633–1641.

33. Bradford PA. Extended-spectrum beta-lactamases in the 21st century: characterization, epidemiology, and detection of this important resistance threat. Clin Microbiol Rev. 2001; 14:933–951. table of contents.

34. Jacoby GA, Munoz-Price LS. The new beta-lactamases. N Engl J Med. 2005; 352:380–391.
35. Walsh TR. Clinically significant carbapenemases: an update. Curr Opin Infect Dis. 2008; 21:367–371.

36. Partridge SR, Tsafnat G, Coiera E, Iredell JR. Gene cassettes and cassette arrays in mobile resistance integrons. FEMS Microbiol Rev. 2009; 33:757–784.

37. Edelstein MV, Skleenova EN, Shevchenko OV, D'Souza JW, Tapalski DV, Azizov IS, Sukhorukova MV, Pavlukov RA, Kozlov RS, Toleman MA, Walsh TR. Spread of extensively resistant VIM-2-positive ST235
Pseudomonas aeruginosa in Belarus, Kazakhstan, and Russia: a longitudinal epidemiological and clinical study. Lancet Infect Dis. 2013; 13:867–876.

38. Watanabe M, Iyobe S, Inoue M, Mitsuhashi S. Transferable imipenem resistance in
Pseudomonas aeruginosa. Antimicrob Agents Chemother. 1991; 35:147–151.

39. Lauretti L, Riccio ML, Mazzariol A, Cornaglia G, Amicosante G, Fontana R, Rossolini GM. Cloning and characterization of
blaVIM, a new integron-borne metallo-beta-lactamase gene from a
Pseudomonas aeruginosa clinical isolate. Antimicrob Agents Chemother. 1999; 43:1584–1590.

40. Wachino J, Yoshida H, Yamane K, Suzuki S, Matsui M, Yamagishi T, Tsutsui A, Konda T, Shibayama K, Arakawa Y. SMB-1, a novel subclass B3 metallo-beta-lactamase, associated with IS
CR1 and a class 1 integron, from a carbapenem-resistant
Serratia marcescens clinical isolate. Antimicrob Agents Chemother. 2011; 55:5143–5149.

41. El Salabi A, Borra PS, Toleman MA, Samuelsen Ø, Walsh TR. Genetic and biochemical characterization of a novel metallo-beta-lactamase, TMB-1, from an
Achromobacter xylosoxidans strain isolated in Tripoli, Libya. Antimicrob Agents Chemother. 2012; 56:2241–2245.

42. Potron A, Poirel L, Nordmann P. Emerging broad-spectrum resistance in
Pseudomonas aeruginosa and
Acinetobacter baumannii: Mechanisms and epidemiology. Int J Antimicrob Agents. 2015; 45:568–585.

43. Willmann M, Bezdan D, Zapata L, Susak H, Vogel W, Schröppel K, Liese J, Weidenmaier C, Autenrieth IB, Ossowski S, Peter S. Analysis of a long-term outbreak of XDR
Pseudomonas aeruginosa: a molecular epidemiological study. J Antimicrob Chemother. 2015; 70:1322–1330.

44. Walsh TR, Toleman MA, Poirel L, Nordmann P. Metallo-beta-lactamases: the quiet before the storm? Clin Microbiol Rev. 2005; 18:306–325.
45. Nordmann P, Poirel L. Emerging carbapenemases in Gram-negative aerobes. Clin Microbiol Infect. 2002; 8:321–331.

46. Toleman MA, Simm AM, Murphy TA, Gales AC, Biedenbach DJ, Jones RN, Walsh TR. Molecular characterization of SPM-1, a novel metallo-beta-lactamase isolated in Latin America: report from the SENTRY antimicrobial surveillance programme. J Antimicrob Chemother. 2002; 50:673–679.

47. Gales AC, Menezes LC, Silbert S, Sader HS. Dissemination in distinct Brazilian regions of an epidemic carbapenem-resistant
Pseudomonas aeruginosa producing SPM metallo-beta-lactamase. J Antimicrob Chemother. 2003; 52:699–702.

48. Salabi AE, Toleman MA, Weeks J, Bruderer T, Frei R, Walsh TR. First report of the metallo-beta-lactamase SPM-1 in Europe. Antimicrob Agents Chemother. 2010; 54:582.

49. Jones RN, Biedenbach DJ, Sader HS, Fritsche TR, Toleman MA, Walsh TR. Emerging epidemic of metallo-beta-lactamase-mediated resistances. Diagn Microbiol Infect Dis. 2005; 51:77–84.
50. Toleman MA, Bennett PM, Walsh TR. IS
CR elements: novel gene-capturing systems of the 21st century? Microbiol Mol Biol Rev. 2006; 70:296–316.

51. Castanheira M, Toleman MA, Jones RN, Schmidt FJ, Walsh TR. Molecular characterization of a beta-lactamase gene,
blaGIM-1, encoding a new subclass of metallo-beta-lactamase. Antimicrob Agents Chemother. 2004; 48:4654–4661.

52. Wendel AF, Brodner AH, Wydra S, Ressina S, Henrich B, Pfeffer K, Toleman MA, Mackenzie CR. Genetic characterization and emergence of the metallo-beta-lactamase GIM-1 in
Pseudomonas spp. and
Enterobacteriaceae during a long-term outbreak. Antimicrob Agents Chemother. 2013; 57:5162–5165.

53. Yong D, Toleman MA, Giske CG, Cho HS, Sundman K, Lee K, Walsh TR. Characterization of a new metallo-beta-lactamase gene,
blaNDM-1, and a novel erythromycin esterase gene carried on a unique genetic structure in
Klebsiella pneumoniae sequence type 14 from India. Antimicrob Agents Chemother. 2009; 53:5046–5054.

54. Flateau C, Janvier F, Delacour H, Males S, Ficko C, Andriamanantena D, Jeannot K, Merens A, Rapp C. Recurrent pyelonephritis due to NDM-1 metallo-beta-lactamase producing Pseudomonas aeruginosa in a patient returning from Serbia, France, 2012. Euro Surveill. 2012; 17:pii: 20311.
55. Janvier F, Jeannot K, Tessé S, Robert-Nicoud M, Delacour H, Rapp C, Mérens A. Molecular characterization of
blaNDM-1 in a sequence type 235
Pseudomonas aeruginosa isolate from France. Antimicrob Agents Chemother. 2013; 57:3408–3411.

56. Khajuria A, Praharaj AK, Kumar M, Grover N. Emergence of NDM - 1 in the clinical isolates of Pseudomonas aeruginosa in India. J Clin Diagn Res. 2013; 7:1328–1331.
57. Carattoli A, Fortini D, Galetti R, Garcia-Fernandez A, Nardi G, Orazi D, Capone A, Majolino I, Proia A, Mariani B, Parisi G, Morrone A, Petrosillo N. Isolation of NDM-1-producing Pseudomonas aeruginosa sequence type ST235 from a stem cell transplant patient in Italy, May 2013. Euro Surveill. 2013; 18:pii: 20633.
58. Zafer MM, Amin M, El Mahallawy H, Ashour MS, Al Agamy M. First report of NDM-1-producing Pseudomonas aeruginosa in Egypt. Int J Infect Dis. 2014; 29:80–81.
59. Kulkova N, Babalova M, Sokolova J, Krcmery V. First report of New Delhi metallo-beta-lactamase-1-producing strains in Slovakia. Microb Drug Resist. 2015; 21:117–120.

60. Pollini S, Maradei S, Pecile P, Olivo G, Luzzaro F, Docquier JD, Rossolini GM. FIM-1, a new acquired metallo-beta-lactamase from a
Pseudomonas aeruginosa clinical isolate from Italy. Antimicrob Agents Chemother. 2013; 57:410–416.

61. Yong D, Shin HB, Kim YK, Cho J, Lee WG, Ha GY, Choi TY, Jeong SH, Lee K, Chong Y. KONSAR group. Increase in the prevalence of carbapenem-resistant
Acinetobacter isolates and ampicillin-resistant non-typhoidal
Salmonella species in Korea: A KONSAR study conducted in 2011. Infect Chemother. 2014; 46:84–93.

62. Poirel L, Naas T, Nicolas D, Collet L, Bellais S, Cavallo JD, Nordmann P. Characterization of VIM-2, a carbapenem-hydrolyzing metallo-beta-lactamase and its plasmid- and integron-borne gene from a
Pseudomonas aeruginosa clinical isolate in France. Antimicrob Agents Chemother. 2000; 44:891–897.

63. Lee K, Lee WG, Uh Y, Ha GY, Cho J, Chong Y. Korean Nationwide Surveillance of Antimicrobial Resistance Group. VIM- and IMP-type metallo-beta-lactamase-producing
Pseudomonas spp and
Acinetobacter spp. in Korean hospitals. Emerg Infect Dis. 2003; 9:868–871.

64. Lee K, Ha GY, Shin BM, Kim JJ, Kang JO, Jang SJ, Yong D, Chong Y. Korean Nationwide Surveillance of Antimicrobial Resistance (KONSAR) group. Metallo-beta-lactamase-producing Gram-negative bacilli in Korean Nationwide Surveillance of Antimicrobial Resistance group hospitals in 2003: continued prevalence of VIM-producing
Pseudomonas spp. and increase of IMP-producing
Acinetobacter spp. Diagn Microbiol Infect Dis. 2004; 50:51–58.

65. Lee K, Park AJ, Kim MY, Lee HJ, Cho JH, Kang JO, Yong D, Chong Y. KONSAR group. Metallo-beta-lactamase-producing
Pseudomonas spp. in Korea: high prevalence of isolates with VIM-2 type and emergence of isolates with IMP-1 type. Yonsei Med J. 2009; 50:335–339.

66. Seok Y, Bae IK, Jeong SH, Kim SH, Lee H, Lee K. Dissemination of IMP-6 metallo-beta-lactamase-producing
Pseudomonas aeruginosa sequence type 235 in Korea. J Antimicrob Chemother. 2011; 66:2791–2796.

67. Hong JS, Kim JO, Lee H, Bae IK, Jeong SH, Lee K. Characteristics of metallo-beta-lactamase-producing
Pseudomonas aeruginosa in Korea. Infect Chemother. 2015; 47:33–40.

68. Valenza G, Joseph B, Elias J, Claus H, Oesterlein A, Engelhardt K, Turnwald D, Frosch M, Abele-Horn M, Schoen C. First survey of metallo-beta-lactamases in clinical isolates of
Pseudomonas aeruginosa in a German university hospital. Antimicrob Agents Chemother. 2010; 54:3493–3497.

69. Yano H, Kuga A, Okamoto R, Kitasato H, Kobayashi T, Inoue M. Plasmid-encoded metallo-beta-lactamase (IMP-6) conferring resistance to carbapenems, especially meropenem. Antimicrob Agents Chemother. 2001; 45:1343–1348.

70. Xu J, Duan X, Wu H, Zhou Q. Surveillance and correlation of antimicrobial usage and resistance of Pseudomonas aeruginosa: a hospital population-based study. PLoS One. 2013; 8:e78604.
71. Kakeya H, Yamada K, Nakaie K, Takizawa E, Okada Y, Fujita A, Nakamura Y, Abe J, Hirose A, Kaneko Y, Hino M. A comparison of susceptibility of Pseudomonas aeruginosa clinical isolates to carbapenem antibiotics in our hospital. Jpn J Antibiot. 2014; 67:241–248.
72. Lee HS, Loh YX, Lee JJ, Liu CS, Chu C. Antimicrobial consumption and resistance in five Gram-negative bacterial species in a hospital from 2003 to 2011. J Microbiol ImmunolInfect. 2014; [Epub ahead of print].

73. Kiratisin P, Chongthaleong A, Tan TY, Lagamayo E, Roberts S, Garcia J, Davies T. Comparative in vitro activity of carbapenems against major Gram-negative pathogens: results of Asia-Pacific surveillance from the COMPACT II study. Int J Antimicrob Agents. 2012; 39:311–316.

74. Mendes RE, Mendoza M, Banga Singh KK, Castanheira M, Bell JM, Turnidge JD, Lin SS, Jones RN. Regional resistance surveillance program results for 12 Asia-Pacific nations (2011). Antimicrob Agents Chemother. 2013; 57:5721–5726.

75. Farajzadeh Sheikh A, Rostami S, Jolodar A, Tabatabaiefar MA, Khorvash F, Saki A, Shoja S, Sheikhi R. Detection of metallo-beta lactamases among carbapenem-resistant Pseudomonas aeruginosa. Jundishapur J Microbiol. 2014; 7:e12289.
76. Castanheira M, Deshpande LM, Costello A, Davies TA, Jones RN. Epidemiology and carbapenem resistance mechanisms of carbapenem-non-susceptible
Pseudomonas aeruginosa collected during 2009-11 in 14 European and Mediterranean countries. J Antimicrob Chemother. 2014; 69:1804–1814.

77. Mansour SA, Eldaly O, Jiman-Fatani A, Mohamed ML, Ibrahim EM. Epidemiological characterization of
P. aeruginosa isolates of intensive care units in Egypt and Saudi Arabia. East Mediterr Health J. 2013; 19:71–80.

78. Liakopoulos A, Mavroidi A, Katsifas EA, Theodosiou A, Karagouni AD, Miriagou V, Petinaki E. Carbapenemase-producing Pseudomonas aeruginosa from central Greece: molecular epidemiology and genetic analysis of class I integrons. BMC Infect Dis. 2013; 13:505.
79. Sefraoui I, Berrazeg M, Drissi M, Rolain JM. Molecular epidemiology of carbapenem-resistant
Pseudomonas aeruginosa clinical strains isolated from western Algeria between 2009 and 2012. Microb Drug Resist. 2014; 20:156–161.

80. Pitout JD, Revathi G, Chow BL, Kabera B, Kariuki S, Nordmann P, Poirel L. Metallo-beta-lactamase-producing
Pseudomonas aeruginosa isolated from a large tertiary centre in Kenya. Clin Microbiol Infect. 2008; 14:755–759.

81. Brink A, Moolman J, da Silva MC, Botha M. National Antibiotic Surveillance Forum. Antimicrobial susceptibility profile of selected bacteraemic pathogens from private institutions in South Africa. S Afr Med J. 2007; 97:273–279.
82. Mataseje LF, Bryce E, Roscoe D, Boyd DA, Embree J, Gravel D, Katz K, Kibsey P, Kuhn M, Mounchili A, Simor A, Taylor G, Thomas E, Turgeon N, Mulvey MR. Canadian Nosocomial Infection Surveillance Program. Carbapenem-resistant Gram-negative bacilli in Canada 2009-10: results from the Canadian Nosocomial Infection Surveillance Program (CNISP). J Antimicrob Chemother. 2012; 67:1359–1367.

83. Morfin-Otero R, Tinoco-Favila JC, Sader HS, Salcido-Gutierrez L, Perez-Gomez HR, Gonzalez-Diaz E, Petersen L, Rodriguez-Noriega E. Resistance trends in gram-negative bacteria: surveillance results from two Mexican hospitals, 2005-2010. BMC Res Notes. 2012; 5:277.

84. Perez F, Hujer AM, Marshall SH, Ray AJ, Rather PN, Suwantarat N, Dumford D 3rd, O'Shea P, Domitrovic TN, Salata RA, Chavda KD, Chen L, Kreiswirth BN, Vila AJ, Haussler S, Jacobs MR, Bonomo RA. Extensively drug-resistant
pseudomonas aeruginosa isolates containing
blaVIM-2 and elements of
Salmonella genomic island 2: a new genetic resistance determinant in Northeast Ohio. Antimicrob Agents Chemother. 2014; 58:5929–5935.

85. Toval F, Guzmán-Marte A, Madriz V, Somogyi T, Rodríguez C, García F. Predominance of carbapenem-resistant
Pseudomonas aeruginosa isolates carrying
blaIMP and
blaVIM metallo-beta-lactamases in a major hospital in Costa Rica. J Med Microbiol. 2015; 64:37–43.

86. Labarca JA, Salles MJ, Seas C, Guzmán-Blanco M. Carbapenem resistance in Pseudomonas aeruginosa and Acinetobacter baumannii in the nosocomial setting in Latin America. Crit Rev Microbiol. 2014; [Epub ahead of print].
87. Mano Y, Saga T, Ishii Y, Yoshizumi A, Bonomo RA, Yamaguchi K, Tateda K. Molecular analysis of the integrons of metallo-beta-lactamase-producing Pseudomonas aeruginosa isolates collected by nationwide surveillance programs across Japan. BMC Microbiol. 2015; 15:41.
88. Qu TT, Zhang JL, Wang J, Tao J, Yu YS, Chen YG, Zhou JY, Li LJ. Evaluation of phenotypic tests for detection of metallo-beta-lactamase-producing
Pseudomonas aeruginosa strains in China. J Clin Microbiol. 2009; 47:1136–1142.

89. Ozgumus OB, Caylan R, Tosun I, Sandalli C, Aydin K, Koksal I. Molecular epidemiology of clinical
Pseudomonas aeruginosa isolates carrying IMP-1 metallo-beta-lactamase gene in a University Hospital in Turkey. Microb Drug Resist. 2007; 13:191–198.

90. Koh TH, Wang GC, Sng LH. Clonal spread of IMP-1-producing
Pseudomonas aeruginosa in two hospitals in Singapore. J Clin Microbiol. 2004; 42:5378–5380.

91. Boonkerd N, Pibalpakdi P, Tiloklurs M, Niumsup PR. Class 1 integron containing metallo beta-lactamase gene
blaIMP-1 in carbapenem-resistant
Pseudomonas aeruginosa in Thailand. J Infect Chemother. 2009; 15:257–261.

92. Fallah F, Borhan RS, Hashemi A. Detection of blaIMP and blaVIM metallo-beta-lactamases genes among Pseudomonas aeruginosa strains. Int J Burns Trauma. 2013; 3:122–124.
93. Shibata N, Doi Y, Yamane K, Yagi T, Kurokawa H, Shibayama K, Kato H, Kai K, Arakawa Y. PCR typing of genetic determinants for metallo-beta-lactamases and integrases carried by gram-negative bacteria isolated in Japan, with focus on the class 3 integron. J Clin Microbiol. 2003; 41:5407–5413.

94. Bert F, Vanjak D, Leflon-Guibout V, Mrejen S, Delpierre S, Redondo A, Nicolas-Chanoine MH. IMP-4-producing
Pseudomonas aeruginosa in a French patient repatriated from Malaysia: impact of early detection and control measures. Clin Infect Dis. 2007; 44:764–765.

95. Peleg AY, Franklin C, Bell JM, Spelman DW. Dissemination of the metallo-beta-lactamase gene
blaIMP-4 among gram-negative pathogens in a clinical setting in Australia. Clin Infect Dis. 2005; 41:1549–1556.

96. Hawkey PM, Xiong J, Ye H, Li H, M'Zali FH. Occurrence of a new metallo-beta-lactamase IMP-4 carried on a conjugative plasmid in Citrobacter youngae from the People's Republic of China. FEMS Microbiol Lett. 2001; 194:53–57.

97. Brízio A, Conceição T, Pimentel M, Da Silva G, Duarte A. High-level expression of IMP-5 carbapenemase owing to point mutation in the -35 promoter region of class 1 integron among
Pseudomonas aeruginosa clinical isolates. Int J Antimicrob Agents. 2006; 27:27–31.

98. Ryoo NH, Lee K, Lim JB, Lee YH, Bae IK, Jeong SH. Outbreak by meropenem-resistant
Pseudomonas aeruginosa producing IMP-6 metallo-beta-lactamase in a Korean hospital. Diagn Microbiol Infect Dis. 2009; 63:115–117.

99. Chen Y, Sun M, Wang M, Lu Y, Yan Z. Dissemination of IMP-6-producing
Pseudomonas aeruginosa ST244 in multiple cities in China. Eur J Clin Microbiol Infect Dis. 2014; 33:1181–1187.

100. Gibb AP, Tribuddharat C, Moore RA, Louie TJ, Krulicki W, Livermore DM, Palepou MF, Woodford N. Nosocomial outbreak of carbapenem-resistant
Pseudomonas aeruginosa with a new
blaIMP allele,
blaIMP-7. Antimicrob Agents Chemother. 2002; 46:255–258.

101. Ho SE, Subramaniam G, Palasubramaniam S, Navaratnam P. Carbapenem-resistant
Pseudomonas aeruginosa in malaysia producing IMP-7 beta-lactamase. Antimicrob Agents Chemother. 2002; 46:3286–3287.

102. Ohlasova D, Kmet V, Niks M. First report of the carbapenem-resistant
Pseudomonas aeruginosa producing IMP-7 metallo-beta-lactamase in Slovakia. Int J Antimicrob Agents. 2007; 30:370–371.

103. Kouda S, Kuwahara R, Ohara M, Shigeta M, Fujiwara T, Komatsuzawa H, Usui T, Sugai M. First isolation of blaIMP-7 in a Pseudomonas aeruginosa in Japan. J Infect Chemother. 2007; 13:276–277.
104. Hrabák J, Fridrichová M, Stolbová M, Bergerová T, Zemlickova H, Urbaskova P. First identification of metallo-beta-lactamase-producing Pseudomonas aeruginosa in the Czech Republic. Euro Surveill. 2009; 14:pii: 19102.
105. Hammerum AM, Jakobsen L, Hansen F, Stegger M, Sørensen LA, Andersen PS, Wang M. Characterisation of an IMP-7-producing ST357
Pseudomonas aeruginosa isolate detected in Denmark using whole genome sequencing. Int J Antimicrob Agents. 2015; 45:200–201.

106. Xiong J, Hynes MF, Ye H, Chen H, Yang Y, M'Zali F, Hawkey PM.
blaIMP-9 and its association with large plasmids carried by
Pseudomonas aeruginosa isolates from the People's Republic of China. Antimicrob Agents Chemother. 2006; 50:355–358.

107. Lim KT, Yasin RM, Yeo CC, Puthucheary SD, Balan G, Maning N, Wahab ZA, Ismail N, Tan EA, Mustaffa A, Thong KL. Genetic fingerprinting and antimicrobial susceptibility profiles of Pseudomonas aeruginosa hospital isolates in Malaysia. J Microbiol Immunol Infect. 2009; 42:197–209.
108. Iyobe S, Kusadokoro H, Takahashi A, Yomoda S, Okubo T, Nakamura A, O'Hara K. Detection of a variant metallo-beta-lactamase, IMP-10, from two unrelated strains of
Pseudomonas aeruginosa and an alcaligenes xylosoxidans strain. Antimicrob Agents Chemother. 2002; 46:2014–2016.

109. Zhao WH, Chen G, Ito R, Hu ZQ. Relevance of resistance levels to carbapenems and integron-borne
blaIMP-1,
blaIMP-7,
blaIMP-10 and
blaVIM-2 in clinical isolates of
Pseudomonas aeruginosa. J Med Microbiol. 2009; 58:1080–1085.

110. Duljasz W, Gniadkowski M, Sitter S, Wojna A, Jebelean C. First organisms with acquired metallo-beta-lactamases (IMP-13, IMP-22, and VIM-2) reported in Austria. Antimicrob Agents Chemother. 2009; 53:2221–2222.

111. Pagani L, Colinon C, Migliavacca R, Labonia M, Docquier JD, Nucleo E, Spalla M, Li Bergoli M, Rossolini GM. Nosocomial outbreak caused by multidrug-resistant
Pseudomonas aeruginosa producing IMP-13 metallo-beta-lactamase. J Clin Microbiol. 2005; 43:3824–3828.

112. Fournier D, Jeannot K, Robert-Nicoud M, Muller E, Cholley P, van der Mee-Marquet N, Plésiat P. Spread of the
blaIMP-13 gene in French
Pseudomonas aeruginosa through sequence types ST621, ST308 and ST111. Int J Antimicrob Agents. 2012; 40:571–573.

113. Naas T, Bogaerts P, Kostyanev T, Cuzon G, Huang TD, Ozsu S, Nordmann P, Glupczynski Y. Silent spread of IMP-13-producing
Pseudomonas aeruginosa belonging to sequence type 621 in Belgium. J Antimicrob Chemother. 2011; 66:2178–2179.

114. Piyakul C, Tiyawisutsri R, Boonbumrung K. Emergence of metallo-beta-lactamase IMP-14 and VIM-2 in
Pseudomonas aeruginosa clinical isolates from a tertiary-level hospital in Thailand. Epidemiol Infect. 2012; 140:539–541.

115. Quinones-Falconi F, Galicia-Velasco M, Marchiaro P, Mussi MA, Ballerini V, Vila AJ, Viale AM, Bermejo-Morales K, Limansky AS. Emergence of
Pseudomonas aeruginosa strains producing metallo-beta-lactamases of the IMP-15 and VIM-2 types in Mexico. Clin Microbiol Infect. 2010; 16:126–131.

116. Gilarranz R, Juan C, Castillo-Vera J, Chamizo FJ, Artiles F, Álamo I, Oliver A. First detection in Europe of the metallo-beta-lactamase IMP-15 in clinical strains of Pseudomonas putida and Pseudomonas aeruginosa. Clin Microbiol Infect. 2013; 19:E424–E427.
117. Mendes RE, Toleman MA, Ribeiro J, Sader HS, Jones RN, Walsh TR. Integron carrying a novel metallo-beta-lactamase gene,
blaIMP-16, and a fused form of aminoglycoside-resistant gene
aac(6')-30/aac(6')-Ib': report from the SENTRY Antimicrobial Surveillance Program. Antimicrob Agents Chemother. 2004; 48:4693–4702.

118. Hanson ND, Hossain A, Buck L, Moland ES, Thomson KS. First occurrence of a
Pseudomonas aeruginosa isolate in the United States producing an IMP metallo-beta-lactamase, IMP-18. Antimicrob Agents Chemother. 2006; 50:2272–2273.

119. Garza-Ramos U, Tinoco P, Silva-Sanchez J, Morfin-Otero R, Rodriguez-Noriega E, Leon-Garnica G, Sader HS, Jones RN. Metallo-beta-lactamase IMP-18 is located in a class 1 integron (In96) in a clinical isolate of
Pseudomonas aeruginosa from Mexico. Int J Antimicrob Agents. 2008; 31:78–80.

120. Wolter DJ, Khalaf N, Robledo IE, Vázquez GJ, Santé MI, Aquino EE, Goering RV, Hanson ND. Surveillance of carbapenem-resistant
Pseudomonas aeruginosa isolates from Puerto Rican Medical Center Hospitals: dissemination of KPC and IMP-18 beta-lactamases. Antimicrob Agents Chemother. 2009; 53:1660–1664.

121. Pollini S, Antonelli A, Venturelli C, Maradei S, Veggetti A, Bracco S, Rumpianesi F, Luzzaro F, Rossolini GM. Acquisition of plasmid-borne
blaIMP-19 gene by a VIM-1-positive
Pseudomonas aeruginosa of the sequence type 235 epidemic lineage. J Antimicrob Chemother. 2013; 68:722–724.

122. Pellegrini C, Mercuri PS, Celenza G, Galleni M, Segatore B, Sacchetti E, Volpe R, Amicosante G, Perilli M. Identification of
blaIMP-22 in
Pseudomonas spp. in urban wastewater and nosocomial environments: biochemical characterization of a new IMP metallo-enzyme variant and its genetic location. J Antimicrob Chemother. 2009; 63:901–908.

123. Kim MJ, Bae IK, Jeong SH, Kim SH, Song JH, Choi JY, Yoon SS, Thamlikitkul V, Hsueh PR, Yasin RM, Lalitha MK, Lee K. Dissemination of metallo-beta-lactamase-producing
Pseudomonas aeruginosa of sequence type 235 in Asian countries. J Antimicrob Chemother. 2013; 68:2820–2824.

124. Jeannot K, Poirel L, Robert-Nicoud M, Cholley P, Nordmann P, Plésiat P. IMP-29, a novel IMP-type metallo-beta-lactamase in
Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2012; 56:2187–2190.

125. Pegg KM, Liu EM, Lacuran AE, Oelschlaeger P. Biochemical characterization of IMP-30, a metallo-beta-lactamase with enhanced activity toward ceftazidime. Antimicrob Agents Chemother. 2013; 57:5122–5126.

126. Pfennigwerth N, Geis G, Gatermann SG, Kaase M. Description of IMP-31, a novel metallo-beta-lactamase found in an ST235 Pseudomonas aeruginosa strain in Western Germany. J Antimicrob Chemother. 2015; [Epub ahed of print].
127. Deshpande LM, Davies TA, Blandino G, Nicoletti G, Jones RN, Castanheira M. IMP-33, a New IMP variant detected in
Pseudomonas aeruginosa from Sicily. Antimicrob Agents Chemother. 2013; 57:6401–6403.

128. Pournaras S, Köck R, Mossialos D, Mellmann A, Sakellaris V, Stathopoulos C, Friedrich AW, Tsakris A. Detection of a phylogenetically distinct IMP-type metallo-beta-lactamase, IMP-35, in a CC235
Pseudomonas aeruginosa from the Dutch-German border region (Euregio). J Antimicrob Chemother. 2013; 68:1271–1276.

129. Tada T, Miyoshi-Akiyama T, Shimada K, Shimojima M, Kirikae T. IMP-43 and IMP-44 metallo-beta-lactamases with increased carbapenemase activities in multidrug-resistant
Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2013; 57:4427–4432.

130. Wang Y, Wang X, Schwarz S, Zhang R, Lei L, Liu X, Lin D, Shen J. IMP-45-producing multidrug-resistant
Pseudomonas aeruginosa of canine origin. J Antimicrob Chemother. 2014; 69:2579–2581.

131. Corvec S, Poirel L, Decousser JW, Allouch PY, Drugeon H, Nordmann P. Emergence of carbapenem-hydrolysing metallo-beta-lactamase VIM-1 in
Pseudomonas aeruginosa isolates in France. Clin Microbiol Infect. 2006; 12:941–942.

132. Tsakris A, Pournaras S, Woodford N, Palepou MF, Babini GS, Douboyas J, Livermore DM. Outbreak of infections caused by
Pseudomonas aeruginosa producing VIM-1 carbapenemase in Greece. J Clin Microbiol. 2000; 38:1290–1292.

133. Mansour W, Poirel L, Bettaieb D, Bouallegue O, Boujaafar N, Nordmann P. Metallo-beta-lactamase-producing Pseudomonas aeruginosa isolates in Tunisia. Diagn Microbiol Infect Dis. 2009; 64:458–461.
134. Castanheira M, Bell JM, Turnidge JD, Mathai D, Jones RN. Carbapenem resistance among
Pseudomonas aeruginosa strains from India: evidence for nationwide endemicity of multiple metallo-beta-lactamase clones (VIM-2, -5, -6, and -11 and the newly characterized VIM-18). Antimicrob Agents Chemother. 2009; 53:1225–1227.

135. Libisch B, Watine J, Balogh B, Gacs M, Muzslay M, Szabó G, Füzi M. Molecular typing indicates an important role for two international clonal complexes in dissemination of VIM-producing
Pseudomonas aeruginosa clinical isolates in Hungary. Res Microbiol. 2008; 159:162–168.

136. Khosravi Y, Tee Tay S, Vadivelu J. Metallo-beta-lactamase-producing imipenem-resistant
Pseudomonas aeruginosa clinical isolates in a university teaching hospital in Malaysia: detection of IMP-7 and first identification of IMP-4, VIM-2, and VIM-11. Diagn Microbiol Infect Dis. 2010; 67:294–296.

137. Yatsuyanagi J, Saito S, Harata S, Suzuki N, Ito Y, Amano K, Enomoto K. Class 1 integron containing metallo-beta-lactamase gene
blaVIM-2 in
Pseudomonas aeruginosa clinical strains isolated in Japan. Antimicrob Agents Chemother. 2004; 48:626–628.

138. Poirel L, Collet L, Nordmann P. Carbapenem-hydrolyzing metallo-beta-lactamase from a nosocomial isolate of Pseudomonas aeruginosa in France. Emerg Infect Dis. 2000; 6:84–85.
139. Poole K. Overcoming multidrug resistance in gram-negative bacteria. Curr Opin Investig Drugs. 2003; 4:128–139.
140. Pallecchi L, Riccio ML, Docquier JD, Fontana R, Rossolini GM. Molecular heterogeneity of
blaVIM-2-containing integrons from
Pseudomonas aeruginosa plasmids encoding the VIM-2 metallo-beta-lactamase. FEMS Microbiol Lett. 2001; 195:145–150.

141. Cardoso O, Leitão R, Figueiredo A, Sousa JC, Duarte A, Peixe LV. Metallo-beta-lactamase VIM-2 in clinical isolates of
Pseudomonas aeruginosa from Portugal. Microb Drug Resist. 2002; 8:93–97.

142. Prats G, Miro E, Mirelis B, Poirel L, Bellais S, Nordmann P. First isolation of a carbapenem-hydrolyzing beta-lactamase in
Pseudomonas aeruginosa in Spain. Antimicrob Agents Chemother. 2002; 46:932–933.

143. Sardelic S, Pallecchi L, Punda-Polic V, Rossolini GM. Carbapenem-resistant
Pseudomonas aeruginosa-carrying VIM-2 metallo-beta-lactamase determinants, Croatia. Emerg Infect Dis. 2003; 9:1022–1023.

144. Walsh TR, Toleman MA, Hryniewicz W, Bennett PM, Jones RN. Evolution of an integron carrying
blaVIM-2 in Eastern Europe: report from the SENTRY Antimicrobial Surveillance Program. J Antimicrob Chemother. 2003; 52:116–119.

145. Zafer MM, Al-Agamy MH, El-Mahallawy HA, Amin MA, El Din Ashour S. Dissemination of VIM-2 producing Pseudomonas aeruginosa ST233 at tertiary care hospitals in Egypt. BMC Infect Dis. 2015; 15:122.
146. Yan JJ, Hsueh PR, Ko WC, Luh KT, Tsai SH, Wu HM, Wu JJ. Metallo-beta-lactamases in clinical
Pseudomonas isolates in Taiwan and identification of VIM-3, a novel variant of the VIM-2 enzyme. Antimicrob Agents Chemother. 2001; 45:2224–2228.

147. Libisch B, Gacs M, Csiszár K, Muzslay M, Rókusz L, Füzi M. Isolation of an integron-borne
blaVIM-4 type metallo-beta-lactamase gene from a carbapenem-resistant
Pseudomonas aeruginosa clinical isolate in Hungary. Antimicrob Agents Chemother. 2004; 48:3576–3578.

148. Castanheira M, Bell JM, Turnidge JD, Mendes RE, Jones RN. Dissemination and genetic context analysis of
blaVIM-6 among
Pseudomonas aeruginosa isolates in Asian-Pacific Nations. Clin Microbiol Infect. 2010; 16:186–189.

149. Toleman MA, Rolston K, Jones RN, Walsh TR.
blaVIM-7, an evolutionarily distinct metallo-beta-lactamase gene in a
Pseudomonas aeruginosa isolate from the United States. Antimicrob Agents Chemother. 2004; 48:329–332.

150. Crespo MP, Woodford N, Sinclair A, Kaufmann ME, Turton J, Glover J, Velez JD, Castañeda CR, Recalde M, Livermore DM. Outbreak of carbapenem-resistant
Pseudomonas aeruginosa producing VIM-8, a novel metallo-beta-lactamase, in a tertiary care center in Cali, Colombia. J Clin Microbiol. 2004; 42:5094–5101.

151. Woodford N, Zhang J, Kaufmann ME, Yarde S, Tomas Mdel M, Faris C, Vardhan MS, Dawson S, Cotterill SL, Livermore DM. Detection of
Pseudomonas aeruginosa isolates producing VEB-type extended-spectrum beta-lactamases in the United Kingdom. J Antimicrob Chemother. 2008; 62:1265–1268.

152. Marchiaro P, Tomatis PE, Mussi MA, Pasteran F, Viale AM, Limansky AS, Vila AJ. Biochemical characterization of metallo-beta-lactamase VIM-11 from a
Pseudomonas aeruginosa clinical strain. Antimicrob Agents Chemother. 2008; 52:2250–2252.

153. Juan C, Beceiro A, Gutiérrez O, Albertí S, Garau M, Pérez JL, Bou G, Oliver A. Characterization of the new metallo-beta-lactamase VIM-13 and its integron-borne gene from a
Pseudomonas aeruginosa clinical isolate in Spain. Antimicrob Agents Chemother. 2008; 52:3589–3596.

154. Mazzariol A, Mammina C, Koncan R, Di Gaetano V, Di Carlo P, Cipolla D, Corsello G, Cornaglia G. A novel VIM-type metallo-beta-lactamase (VIM-14) in a
Pseudomonas aeruginosa clinical isolate from a neonatal intensive care unit. Clin Microbiol Infect. 2011; 17:722–724.

155. Schneider I, Keuleyan E, Rasshofer R, Markovska R, Queenan AM, Bauernfeind A. VIM-15 and VIM-16, two new VIM-2-like metallo-beta-lactamases in
Pseudomonas aeruginosa isolates from Bulgaria and Germany. Antimicrob Agents Chemother. 2008; 52:2977–2979.

156. Siarkou VI, Vitti D, Protonotariou E, Ikonomidis A, Sofianou D. Molecular epidemiology of outbreak-related
Pseudomonas aeruginosa strains carrying the novel variant
blaVIM-17 metallo-beta-lactamase gene. Antimicrob Agents Chemother. 2009; 53:1325–1330.

157. Riera E, Cabot G, Mulet X, García-Castillo M, del Campo R, Juan C, Cantón R, Oliver A.
Pseudomonas aeruginosa carbapenem resistance mechanisms in Spain: impact on the activity of imipenem, meropenem and doripenem. J Antimicrob Chemother. 2011; 66:2022–2027.

158. El-Mahdy TS. Identification of a novel metallo-beta-lactamase VIM-28 located within unusual arrangement of class 1 integron structure in
Pseudomonas aeruginosa isolates from Egypt. Jpn J Infect Dis. 2014; 67:382–384.

159. Iraz M, Duzgun AO, Cicek AC, Bonnin RA, Ceylan A, Saral A, Nordmann P, Sandalli C. Characterization of novel VIM carbapenemase, VIM-38, and first detection of GES-5 carbapenem-hydrolyzing beta-lactamases in
Pseudomonas aeruginosa in Turkey. Diagn Microbiol Infect Dis. 2014; 78:292–294.

160. Poirel L, Magalhaes M, Lopes M, Nordmann P. Molecular analysis of metallo-beta-lactamase gene
blaSPM-1-surrounding sequences from disseminated
Pseudomonas aeruginosa isolates in Recife, Brazil. Antimicrob Agents Chemother. 2004; 48:1406–1409.

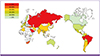






 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download