Abstract
Vertebral osteomyelitis caused by nontuberculous mycobacteria (NTM) is rarely reported, especially in an immunocompetent host. NTM are usually not susceptible in vitro to antituberculous drugs, and appropriate antimicrobial therapy for treatment of NTM infection is based on susceptibility results, which vary between different NTM species; therefore, treatment of vertebral osteomyelitis caused by NTM is challenging. We report on the first case of vertebral osteomyelitis caused by M. abscessus in an otherwise healthy individual, confirmed by cultures of bone tissue obtained during surgery. Clinical cure was achieved with a combination of antimicrobial therapy and surgery. We also review previous reports of vertebral osteomyelitis caused by NTM.
Nontuberculous mycobacteria (NTM) are free-living organisms that are ubiquitous in the environment. These organisms can also inhabit body surfaces or secretions without causing significant disease. Thus, occasional isolates of NTM are largely considered colonizers or contaminants. However, with the decreasing incidence of tuberculosis and development of new microbiological diagnostic methods, the importance of NTM in human disease has become increasingly evident. NTM cause distinct clinical syndromes, such as pulmonary disease, superficial lymphadenitis, skin and soft tissue infections, and disseminated disease in severely immunocompromised patients [1].
Rapidly growing mycobacteria (RGM) include three clinically relevant species, Mycobacterium fortuitum, M. chelonae, and M. abscessus. RGM are environmental organisms found worldwide; compared with other mycobacteria, RGM usually show rapid growth in subculture within one week. Among RGM, M. abscessus is the most pathogenic; RGM are likely to cause pulmonary infection, primarily in patients with underlying lung disease. RGM also cause infections of the musculoskeletal system from either hematogenous seeding or percutaneous inoculation due to trauma or surgery. The clinical course is indolent, slowly progressive, and destructive, due in part to delay in diagnosis. Herein, we report on a case of vertebral osteomyelitis caused by M. abscessus in an immunocompetent patient with no risk factors.
A 55-year-old female patient presented with a ten-month history of progressive lower-back pain. She did not have a history of trauma, penetrating injury, or invasive procedure, such as acupuncture, immunocompromised status, diabetes, or steroid use. In another hospital, she was diagnosed with infectious spondylitis and was initially treated with cefazolin and then switched to ciprofloxacin for a total of two months.
However, her lower-back pain did not show improvement. Therefore, she was transferred to our hospital.
A physical examination showed diffuse tenderness over the mid-lumbar region, however, no other signs of neurological involvement were observed. She was afebrile and had a negative psoas sign.
Results of laboratory studies showed a WBC count of 6,700/mm3 and a C-reactive protein (CRP) level of 6.04 mg/dL. A human immunodeficiency virus serology test showed a negative result, and the tuberculin skin test also showed a negative result. Findings on an initial CT scan showed infectious spondylitis of L2 and L3 and disc bulging with annular tears at L3-L4 and L5-S1. MRI of the spine showed enhancement of the L2, L3 vertebral bodies, the intervertebral disc space between L2-L3, as well as paraspinal abscesses (Fig. 1). She was diagnosed with vertebral osteomyelitis and underwent debridement and drainage of the paraspinal abscess, corpectomy of L2 and L3, total discectomy of L2 and L3, and anterior lumbar interbody fusion from L2 to L3 with an autologous iliac bone graft. A specimen obtained surgically showed acute osteomyelitis of the bone, chronic inflammation, and myositis of the soft tissue. Results of AFB stain, cultures, and NTM PCR (polymerase chain reaction) of the collected disc, muscle, and abscess indicated M. abscessus.
The patient was treated with an initial antimycobacterial regimen that included IV amikacin, IV imipenem, IV moxifloxacin, and oral clarithromycin. After the first month of treatment, by a drug sensitivity test of M. abscessus referred to the Korean Institute of Tuberculosis, M. abscessus isolated from the patient was found to be susceptible only to clarithromycin and was resistant to ciprofloxacin, doxycycline, and trimethoprim-sulfamethoxazole. The patient was discharged from the hospital, taking clarithromycin and rifampin.
At her two-month follow-up, she reported that her back pain had gradually subsided, her CRP level had normalized, and follow-up radiographs showed stabilization of the involved segments. The patient took clarithromycin and rifampicin for six months.
NTM are an extremely rare cause of vertebral osteomyelitis. A MedLine review of the literature published between 1965 and June 2012 found only 53 case reports and one report on a nosocomial outbreak [2-15]. The NTM species identified most frequently was M. avium complex (n=17), followed by M. xenopi (n=12), M. fortuitum (n=9), M. abscessus (n=4), M. chelonae (n=2), and M. kansasii (n=3); single cases involving M. bovis, M. flavescens, M. heckeshornense, M. simiae, and two unidentified NTM were also reported (Table 1). Various degrees of immunosuppression (systemic lupus erythematosus with steroids, n=8; HIV infection, n=9; interferon receptor defect, n=1; carcinoma, n=1; diabetes, n=1; renal failure, n=1; liver cirrhosis, n=1; chronic granulomatous disease, n=1; rheumatoid arthritis with etanercept, n=1; and renal transplantation recipient, n=1) were reported in 25 (47.2%) of 53 patients with vertebral osteomyelitis caused by NTM. Seven patients (13.2%) developed vertebral osteomyelitis caused by NTM after blunt trauma or surgery to the back. Other reported causes of vertebral osteomyelitis were chronic lung disease (n=2) and intravenous drug abuse (n=2). Vertebral osteomyelitis caused by M. abscessus is none in an immunocompetent patient. We report on the first case of vertebral osteomyelitis caused by M. abscessus in an otherwise healthy individual.
Diagnosis of vertebral osteomyelitis, based on clinical, laboratory, and radiologic features, can be difficult. Due to the rarity of the disease, the insidious onset of symptoms, and the high frequency of lower back pain in the general population, it is often delayed or missed. In diagnosis, biopsy or culture of the vertebral lesion can provide important clues, and must be performed during the initial stages.
Diagnosis of NTM vertebral osteomyelitis can be easily missed in patients without risk factors. A high index of suspicion is required for prompt diagnosis in order to ensure improved long-term outcomes when the clinical course is indolent, slowly progressive, destructive, and not responsive to empirical antibacterial agents. Laboratory tests and imaging studies are not sufficient for diagnosis and differentiation from other infections. Organisms are isolated from sterile tissue or bony specimens for diagnosis of NTM vertebral osteomyelitis [16]. If the specimen shows a positive result for AFB stain, it will then be analyzed for M. tuberculosis using MTB-PCR. In the case of a positive result for MTB-PCR, it can be tentatively diagnosed as M. tuberculosis. However, the possibility of NTM should be considered in cases showing a negative result of MTB-PCR and the results of the AFB culture will be necessary in the final diagnosis.
RGM, in particular, are highly resistant to multiple antimycobacterial drugs. A previous study reported that that all isolates of M. abscessus and M. chelonae and approximately 80 percent of isolates of M. fortuitum are susceptible in vitro to the macrolide clarithromycin [17].
Because of these resistance patterns, routine susceptibility testing to antituberculosis drugs is not recommended for RGM [1]. Our results from antimicrobial susceptibility testing suggest that clarithromycin is the only effective agent against M. abscessus. Imipenem, cefoxitin, and amikacin may have a role in multiple drug regimens [18]. Monotherapy has proven disappointing for treatment of infections due to M. abscessus, and a combination of at least two to three active antibiotics is required.
No randomized controlled studies have evaluated the duration of antibiotic treatment for vertebral osteomyelitis. Previous experience suggests that in cases involving disseminated or deep-seated M. abscessus infection, antimicrobial treatment should be continued for a minimum of six weeks after clinical resolution of the primary infection; this usually requires six months of treatment [19]. A more prolonged course lasting years may be necessary in severely immunocompromised patients, and those with extensive bone destruction and/or paravertebral infection. A consensus on guidelines concerning treatment of spinal infections caused by NTM does not exist, however, limited clinical experience suggests that aggressive surgical debridement of all involved tissues is critical, and performance of sequential surgical procedures is often required. Indications for surgery in patients with pyogenic osteomyelitis include progression of disease despite adequate directed or empiric antimicrobial therapy, epidural or paravertebral abscess formation, and threatened or actual cord compression due to vertebral collapse and/or spinal instability [20]; because there is no consensus regarding indications for surgery in patients with vertebral osteomyelitis caused by NTM, these can be applied to patients with vertebral osteomyelitis caused by NTM. The best way to reduce the morbidity and mortality associated with vertebral osteomyelitis is to shorten the time between onset of symptoms and initiation of appropriate therapy.
Our case illustrates the unusual occurrence of vertebral osteomyelitis caused by one NTM, M. abscessus. Only four cases of vertebral osteomyelitis caused by M. abscessus have been reported in the literature; all patients had comorbid conditions, including steroid use, intravenous cocaine abuse, diabetes, and wounds from blunt trauma or surgery to the back. To the best of our knowledge, this is the first case of vertebral osteomyelitis caused by M. abscessus in an otherwise healthy individual. Owing to the negative AFB cultures and NTM PCR of the collected bony specimens, the possibility of contamination by NTM cannot be completely excluded. However, we suppose that M. abscessus is the true pathogen because the clinical course of the patient showed improvement after treatment for NTM.
Figures and Tables
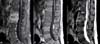 | Figure 1Spondylitis of the L2, L3 bodies with a prevertebral abscess is shown as low signal intensity on a T1-weighted image, high signal intensity on a T2-weighted image, and enhancement on a contrast media-enhanced T1-weighted image. (A) T1-weighted image, (B) T2-weighted image, (C) contrast media-enhanced T1-weighted image). |
References
1. Griffith DE, Aksamit T, Brown-Elliott BA, Catanzaro A, Daley C, Gordin F, Holland SM, Horsburgh R, Huitt G, Iademarco MF, Iseman M, Olivier K, Ruoss S, von Reyn CF, Wallace RJ Jr, Winthrop K. ATS Mycobacterial Diseases Subcommittee. American Thoracic Society. Infectious Disease Society of America. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007. 175:367–416.

2. Duttaroy B, Agrawal C, Pendse A. Spinal tuberculosis due to dissemination of atypical mycobacteria. Indian J Med Sci. 2004. 58:203–205.
3. Petitjean G, Fluckiger U, Scharen S, Laifer G. Vertebral osteomyelitis caused by non-tuberculous mycobacteria. Clin Microbiol Infect. 2004. 10:951–953.

4. Yano T, Okuda S, Kato K, Kato K, Kishimoto T. Mycobacterium kansasii osteomyelitis in a patient with AIDS on highly active antiretroviral therapy. Intern Med. 2004. 43:1084–1086.

5. Meybeck A, Fortin C, Abgrall S, Adle-Biassette H, Hayem G, Ruimy R, Yeni P. Spondylitis due to Mycobacterium xenopi in a human immunodeficiency virus type 1-infected patient: case report and review of the literature. J Clin Microbiol. 2005. 43:1465–1466.

6. Tortoli E, Mantella A, Mariottini A, Mazzarelli G, Pecile P, Rogasi PG, Sterrantino G, Fantoni E, Leoncini F. Successfully treated spondylodiscitis due to a previously unreported mycobacterium. J Med Microbiol. 2006. 55:119–121.

7. Corrales-Medina V, Symes S, Valdivia-Arenas M, Boulanger C. Localized Mycobacterium avium complex infection of vertebral and paravertebral structures in an HIV patient on highly active antiretroviral therapy. South Med J. 2006. 99:174–177.

8. Korres DS, Papagelopoulos PJ, Zahos KA, Kolia MD, Poulakou GG, Falagas ME. Multifocal spinal and extra-spinal Mycobacterium chelonae osteomyelitis in a renal transplant recipient. Transpl Infect Dis. 2007. 9:62–65.

9. Danaviah S, Govender S, Gordon ML, Cassol S. Atypical mycobacterial spondylitis in HIV-negative patients identified by genotyping. J Bone Joint Surg Br. 2007. 89:346–348.

10. Sobottke R, Zarghooni K, Seifert H, Faetkenheuer G, Koriller M, Michael JW, Delank KS, Eysel P. Spondylodiscitis caused by Mycobacterium xenopi. Arch Orthop Trauma Surg. 2008. 128:1047–1053.
11. Hirakawa A, Miyamoto K, Ohno Y, Hioki A, Ogawa H, Nishimoto H, Yokoi T, Hosoe H, Shimizu K. Two-stage (posterior and anterior) surgical treatment of spinal osteomyelitis due to atypical mycobacteria and associated thoracolumbar kyphoscoliosis in a nonimmunocompromised patient. Spine (Phila Pa 1976). 2008. 33:E221–E224.

12. Wong NM, Sun LK, Lau PY. Spinal infection caused by Mycobacterium avium complex in a patient with no acquired immune deficiency syndrome: a case report. J Orthop Surg (Hong Kong). 2008. 16:359–363.

13. Rahman I, Bhatt H, Chillag S, Duffus W. Mycobacterium chelonae vertebral osteomyelitis. South Med J. 2009. 102:1167–1169.
14. Takakuwa O, Oguri T, Kasai D, Nakamura A, Sato S, Ueda R. A case of spinal osteomyelitis due to Mycobacterium intracellulare with pulmonary Mycobacterium avium complex. Nihon Kokyuki Gakkai Zasshi. 2010. 48:759–764.
15. Kim BH, Park JS, Jang YR, Kim JY, Cho YK. A case of Mycobacterium abscessus spondylitis. Korean J Med. 2012. 83:277–282.
16. Chan ED, Kong PM, Fennelly K, Dwyer AP, Iseman MD. Vertebral osteomyelitis due to infection with nontuberculous Mycobacterium species after blunt trauma to the back: 3 examples of the principle of locus minoris resistentiae. Clin Infect Dis. 2001. 32:1506–1510.

17. Brown BA, Wallace RJ Jr, Onyi GO, De Rosas V, Wallace RJ 3rd. Activities of four macrolides, including clarithromycin, against Mycobacterium fortuitum, Mycobacterium chelonae, and M. chelonae-like organisms. Antimicrob Agents Chemother. 1992. 36:180–184.

18. Wallace RJ Jr, Brown BA, Onyi GO. Susceptibilities of Mycobacterium fortuitum biovar. fortuitum and the two subgroups of Mycobacterium chelonae to imipenem, cefmetazole, cefoxitin, and amoxicillin-clavulanic acid. Antimicrob Agents Chemother. 1991. 35:773–775.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download