Abstract
Background
The most effective treatment for end-stage renal disease is kidney transplantation, and the number of kidney transplantations has shown a rapid increase. The aim of this study was to determine graft survival and functional outcome of 1,500 kidney transplant cases in a single center. We also investigated the factors affecting graft failure after kidney transplantation.
Methods
We retrospectively reviewed the clinical data of 1,500 pairs of donors and subsequent recipients who underwent kidney transplantation in Samsung Medical Center, from February 1995 to January 2012.
Results
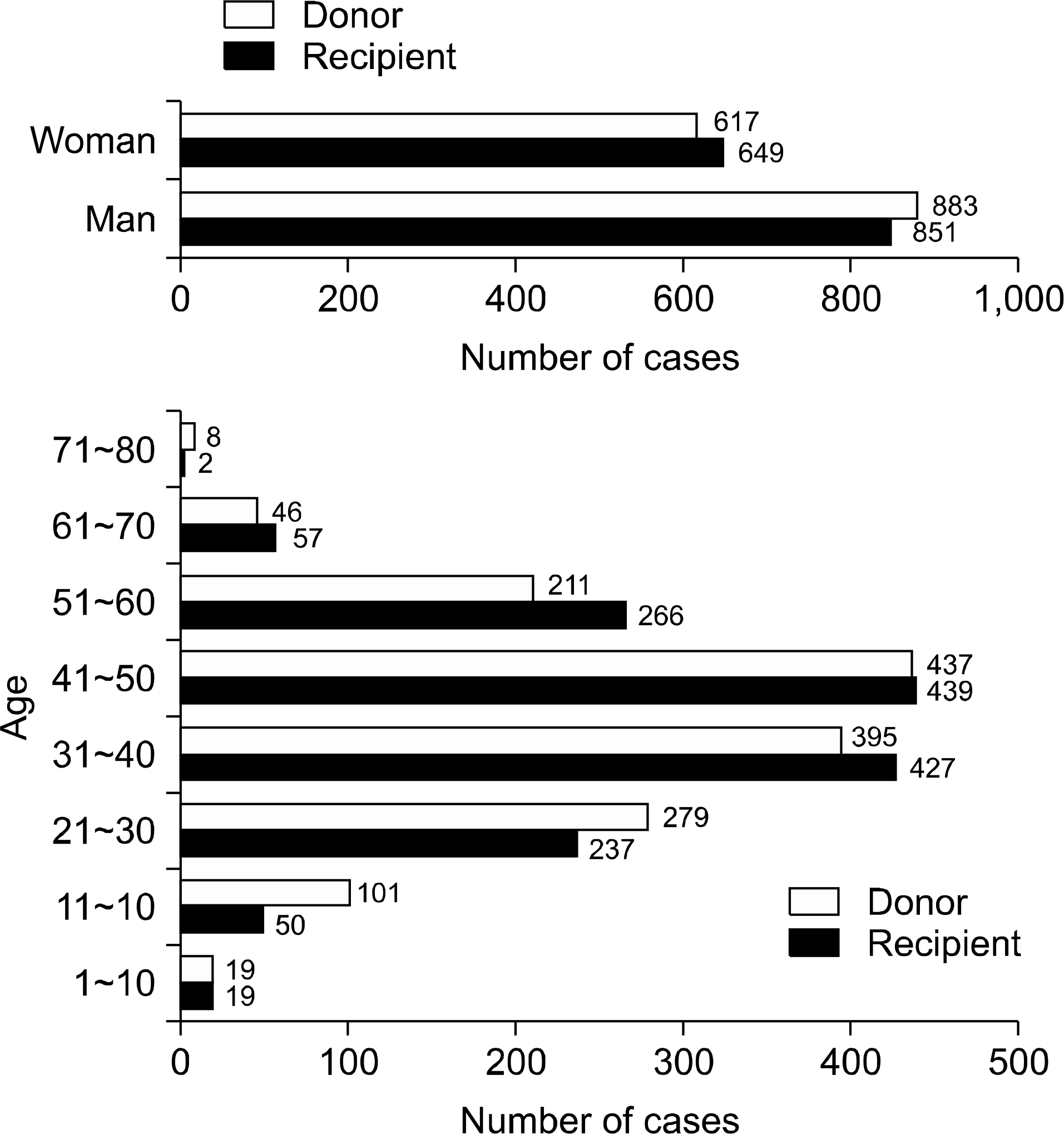
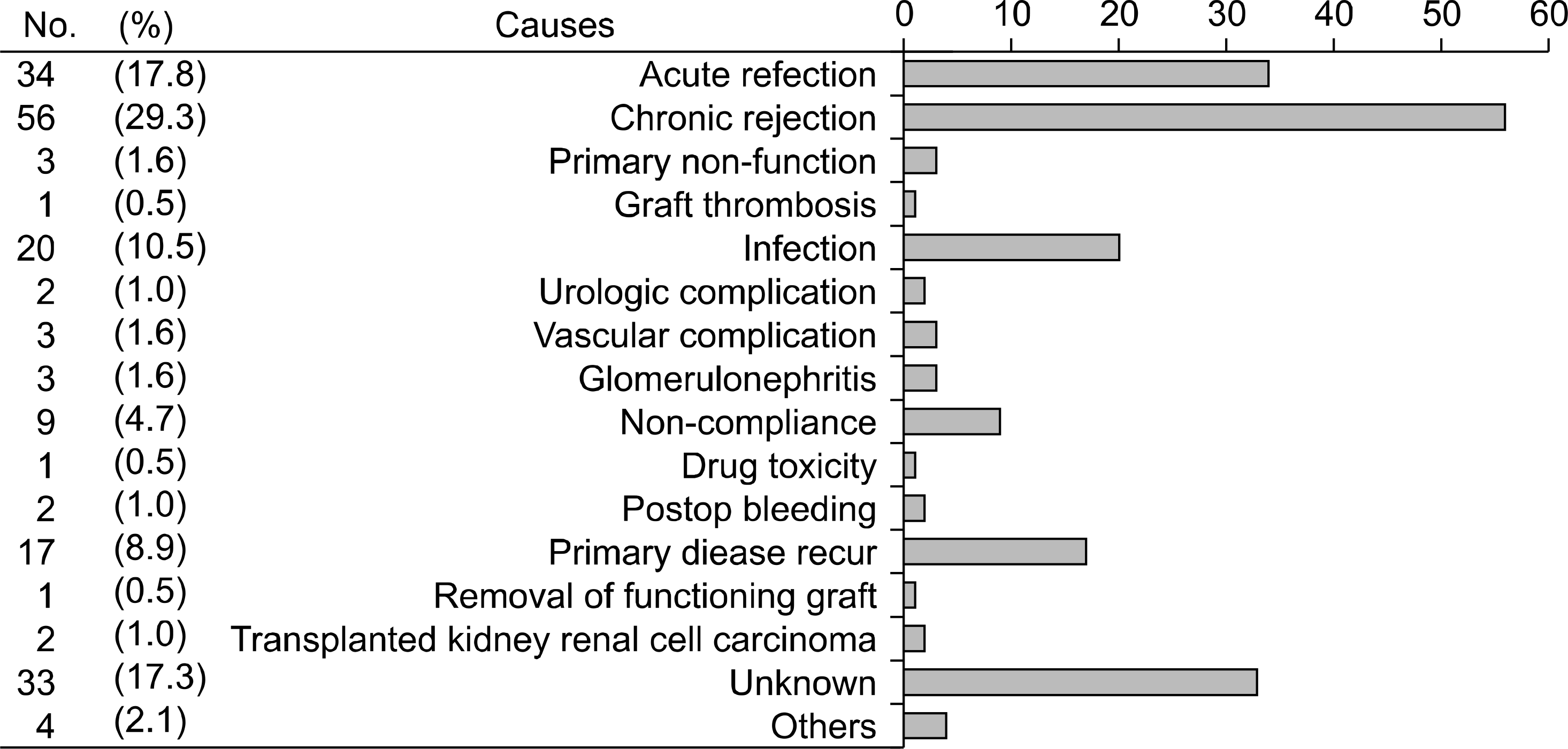
The mean follow-up period was 2,241.5±1,609.4 days. There were 851 (56.7%) male recipients; 62 (4.1%) recipients were younger than 19 years old. Eleven (0.7%) cases were ABO blood group incompatible kidney transplant. A total of 531 (35%) deceased and 969 (65%) living donors were included. Among them, 191 (12.7%) recipients were experienced in graft failure. The most common cause of graft loss was chronic allograft nephropathy. One-year, 5-year, 10-year, and 15-year graft survival were 97.3%, 92.8%, 81.6%, and 75.1% (85.2% for living, 75.4% for deceased donor), respectively. Higher incidence of graft failure was observed in recipients who received deceased donor kidneys or experienced a rejection episode.
References
1). Korean Network for Organ Sharing (KONOS). KONOS Annual Report 2011 [Internet]. Seoul: KONOS;2011. [cited 2014 Jan 15]. Available from:. http://www.konos.go.kr.
2). McDonald SP, Russ GR. Survival of recipients of cadaveric kidney transplants compared with those receiving dialysis treatment in Australia and New Zealand, 1991–2001. Nephrol Dial Transplant. 2002; 17:2212–9.

3). Schnuelle P, Lorenz D, Trede M, Van Der Woude FJ. Impact of renal cadaveric transplantation on survival in end-stage renal failure: evidence for reduced mortality risk compared with hemodialysis during longterm followup. J Am Soc Nephrol. 1998; 9:2135–41.

4). Wolfe RA, Ashby VB, Milford EL, Ojo AO, Ettenger RE, Agodoa LY, et al. Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant. N Engl J Med. 1999; 341:1725–30.

5). Jung HT, Jung GO, Choi GS, Kwon CH, Kim SJ, Joh JW, et al. Report of 1,000 kidney transplants at the Sungkyunkwan University of Korea. J Korean Soc Transplant. 2009; 23:141–8. (정희택, 정금오, 최규성, 권준혁, 김성주, 조재원, 등. 성균관대학교 의과대학, 삼성서울병원에서 시행한 신장이식 1,000 예의 임상성적 보고. 대한이식학회지 2009;23: 141–8.).

6). Ekberg H, Tedesco-Silva H, Demirbas A, Vítko S, Nashan B, Gürkan A, et al. Reduced exposure to calcineurin inhibitors in renal transplantation. N Engl J Med. 2007; 357:2562–75.

7). Srinivas TR, Meier-Kriesche HU. Minimizing immunosuppression, an alternative approach to reducing side effects: objectives and interim result. Clin J Am Soc Nephrol. 2008; 3(Suppl 2):S101–16.

8). Park JB, Kim SJ, Oh HY, Han YS, Kim DJ, Park JW, et al. Steroid withdrawal in living donor renal transplant recipients using tacrolimus and cyclosporine: a randomized prospective study. Transpl Int. 2006; 19:478–84.

9). Kim CK, Song JH, Kim SM, Peck KR, Oh W, Huh W, et al. Clinical usefulness of human cytomegalovirus antigenemia assay after kidney transplantation. Transplantation. 2003; 75:2151–5.

10). Port FK, Merion RM, Roys EC, Wolfe RA. Trends in organ donation and transplantation in the United States, 1997–2006. Am J Transplant. 2008; 8(4 Pt 2):911–21.

11). Dahmane D, Audard V, Hiesse C, Pessione F, Bentaarit B, Barrou B, et al. Retrospective follow-up of transplantation of kidneys from ‘marginal' donors. Kidney Int. 2006; 69:546–52.

12). Pascual J, Zamora J, Pirsch JD. A systematic review of kidney transplantation from expanded criteria donors. Am J Kidney Dis. 2008; 52:553–86.

13). Nankivell BJ, Borrows RJ, Fung CL, O'Connell PJ, Allen RD, Chapman JR. The natural history of chronic allograft nephropathy. N Engl J Med. 2003; 349:2326–33.

14). Moon JI, Lee CM, Kim SI, Kim MS, Kim YS, Park K. The impact of acute rejection on longterm graft outcome in renal allograft recipient. J Korean Soc Transplant. 1998; 12:67–74. (문장일, 이창목, 김순일, 김명수, 김유선, 박기일. 급성 거부 반응이 이식신의 장기 생존율에 미치는 영향. 대한이식학회지 1998;12: 67–74.).
15). Sellarés J, de Freitas DG, Mengel M, Reeve J, Einecke G, Sis B, et al. Understanding the causes of kidney transplant failure: the dominant role of antibody-mediated rejection and nonadherence. Am J Transplant. 2012; 12:388–99.

16). Humar A, Michaels M. AST ID Working Group on Infectious Disease Monitoring. American Society of Transplantation recommendations for screening, monitoring and reporting of infectious complications in immunosuppression trials in recipients of organ transplantation. Am J Transplant. 2006; 6:262–74.

17). Tolkoff-Rubin NE, Rubin RH. Clinical approach to viral and fungal infections in the renal transplant patient. Semin Nephrol. 1992; 12:364–75.
18). Ramos E, Drachenberg CB, Portocarrero M, Wali R, Klassen DK, Fink JC, et al. BK virus nephropathy diagnosis and treatment: experience at the University of Maryland Renal Transplant Program. Clin Transpl. 2002; 143–53.
19). Rubin RH, Fang LS, Cosimi AB, Herrin JT, Varga PA, Russell PS, et al. Usefulness of the antibody-coated bacteria assay in the management of urinary tract infection in the renal transplant patient. Transplantation. 1979; 27:18–20.

20). Myerowitz RL, Medeiros AA, O'Brien TF. Bacterial infection in renal homotransplant recipients: a study of fif-ty-three bacteremic episodes. Am J Med. 1972; 53:308–14.
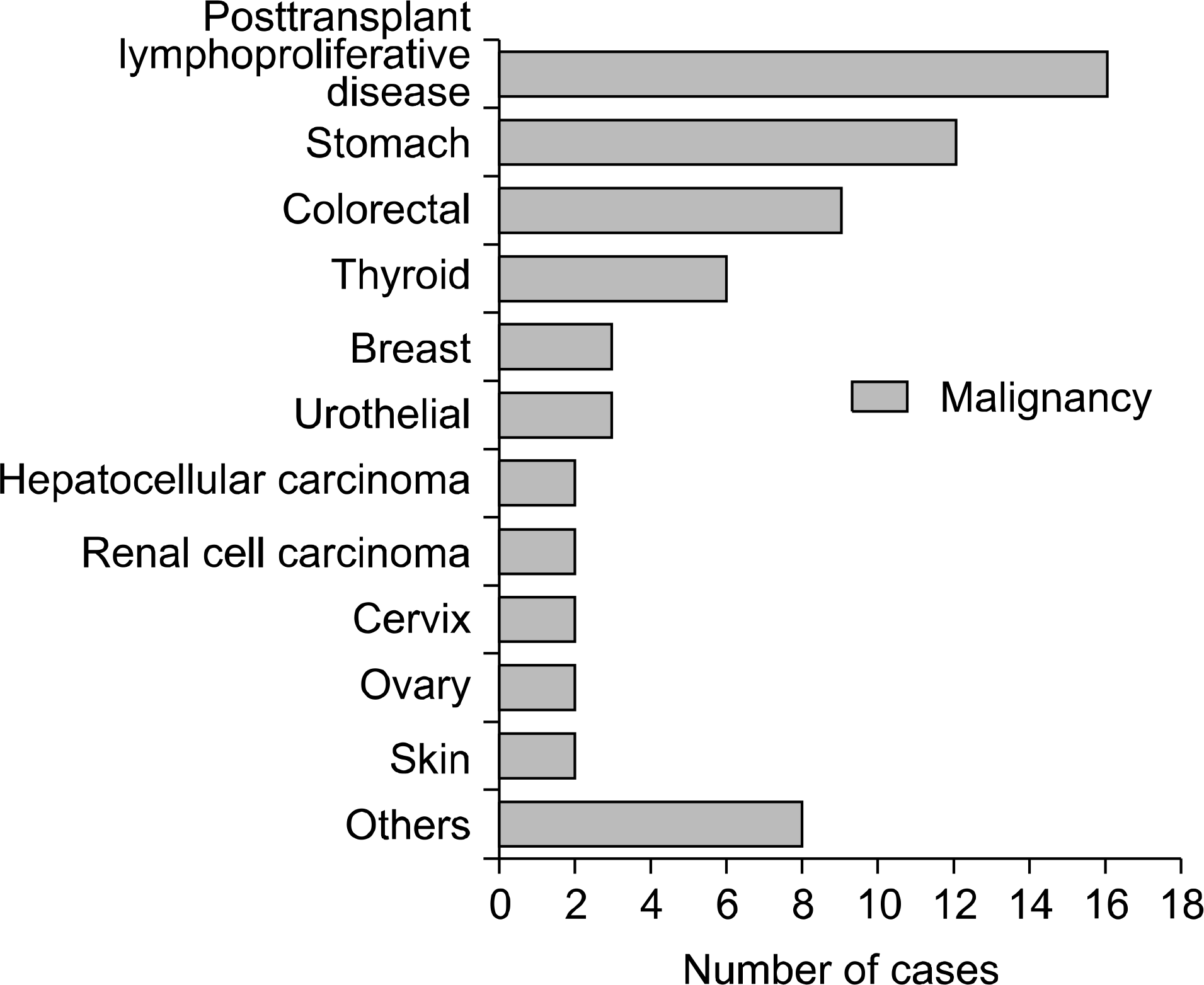
21). Mazuecos A, Muñoz Terol JM, García Alvárez T, Sola E, Rodríguez Benot A, Dsuna A, et al. Increase in malignancies as cause of death in renal transplant patients. Transplant Proc. 2009; 41:2159–62.

22). Buell JF, Gross TG, Woodle ES. Malignancy after transplantation. Transplantation. 2005; 80(2 Suppl):S254–64.

23). National Cancer Information Center annual report 2013 [Internet]. Seoul: National Cancer Information Center;2013. [cited 2014 Jan 15]. Available from:. http://cancer.go.kr.
Fig. 3.
Relationship of donors and recipients. (A) Donor type. (B) Relationship of donor and recipients in living related kidney transplantation. (C) Relationship of donor and recipients in living unrelated kidney transplantation. (D) Donor criteria in deceased donor.
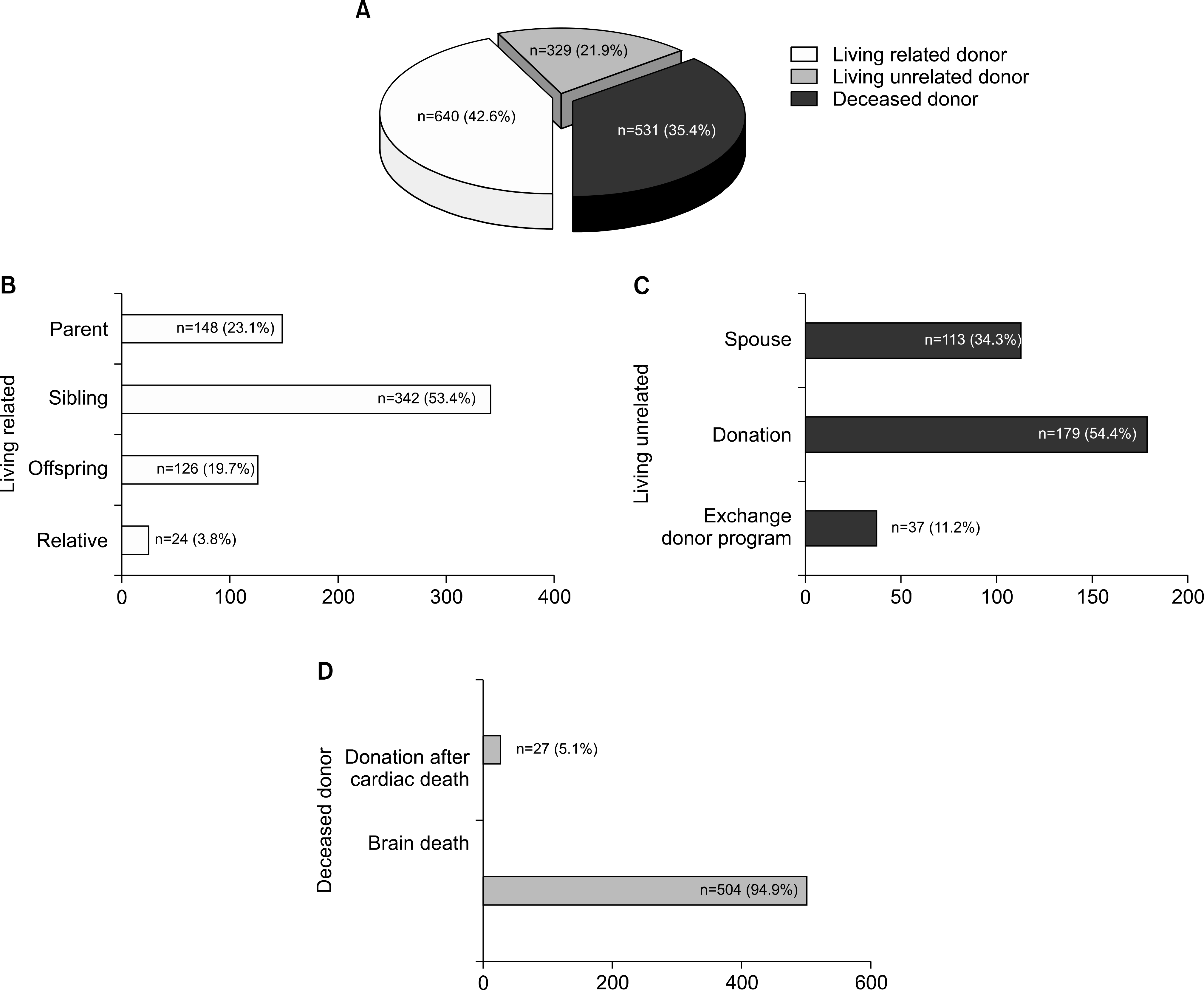
Fig. 5.
Long-term outcomes after kidney transplantation: graft survival. (A) Overall graft survival. (B) Living donor kidney transplantation (LDKT): deceased donor kidney transplantation (DDKT) graft survival.
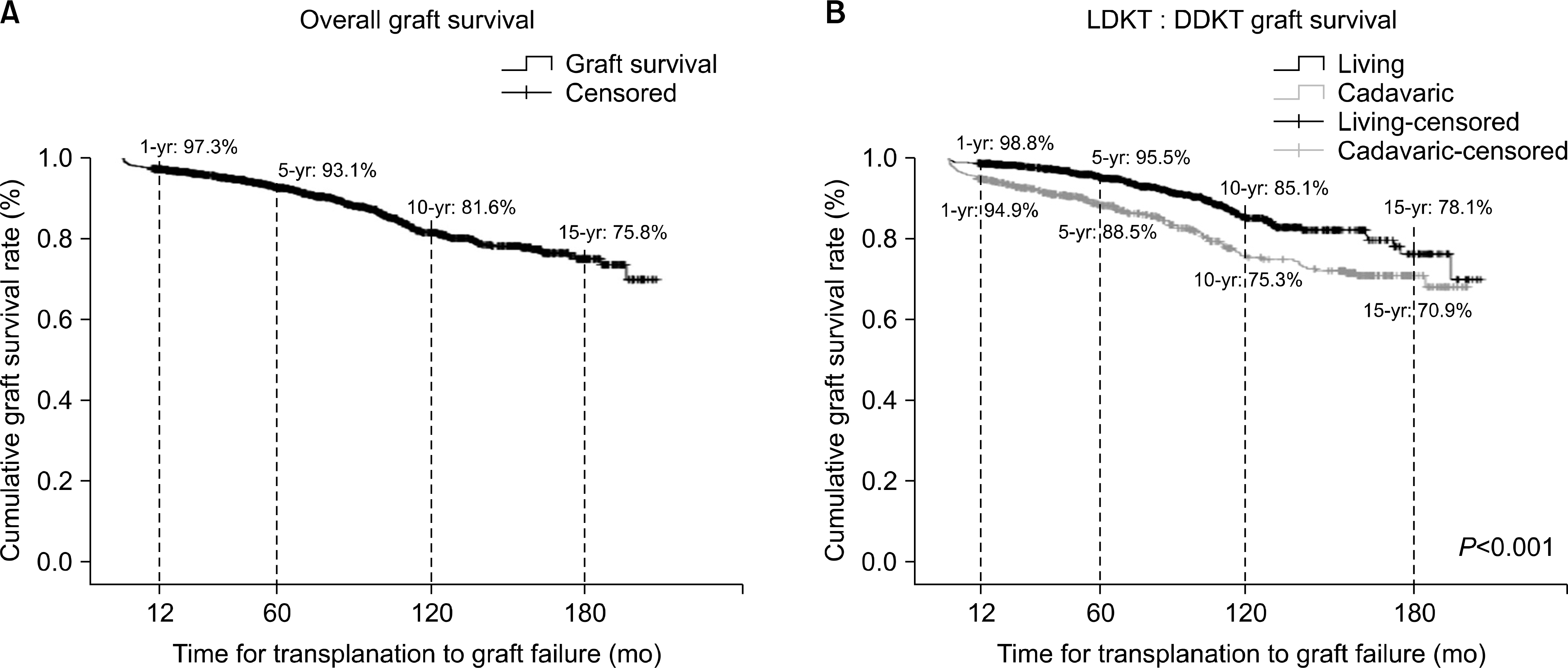
Fig. 7.
Long-term outcomes after kidney transplantation: patient survival. (A) Overall patient survival. (B) Living donor kidney transplantation (LDKT): deceased donor kidney transplantation (DDKT) patient survival.
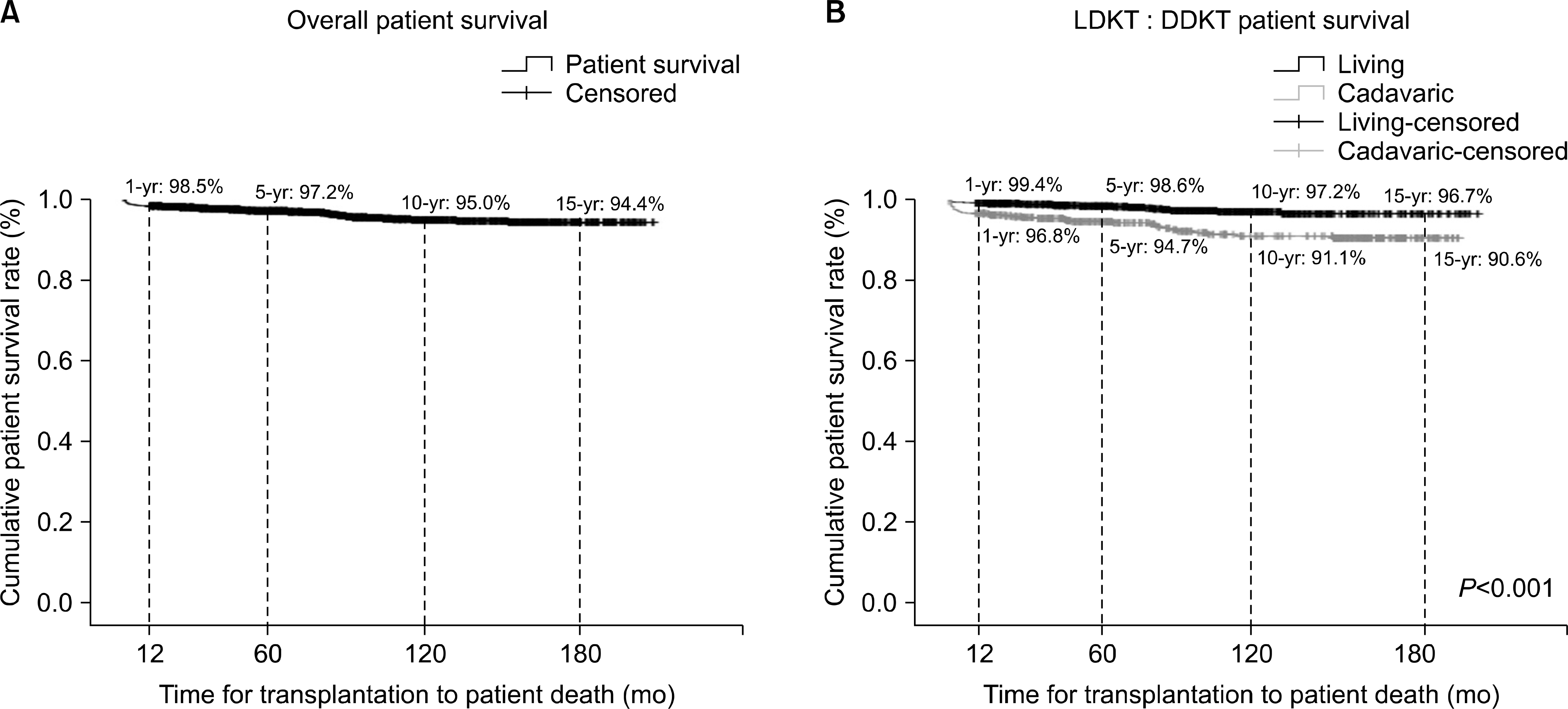
Table 1.
Baseline demographics of kidney transplantation
Table 2.
Use of immunosuppressive agents
Table 3.
Immediate and late complications after kidney transplantation
Table 4.
Infectious complications after kidney transplantation
Table 5.
Causes of patient death after kidney transplantation
Table 6.
Impact factors affecting graft failure after kidney transplantation




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download