Abstract
Posttransplant lymphoproliferative disorder (PTLD) is documented as one of the serious complications leading to mortality particularly in organ transplant recipients receiving immunosuppressive therapy. Extant literature confirms beyond doubt that the most common site of involvement of PTLD is lymph nodes, and rarely involved is the gastrointestinal tract. It is a well-known fact that Epstein-Barr virus (EBV) is a risk factor for PTLD development. In this study, we report a case of PTLD presented as small bowel perforation without EBV infection after long-term immunosuppressive therapy in a renal transplant recipient.
Go to : 
REFERENCES
1). Sampaio MS, Cho YW, Qazi Y, Bunnapradist S, Hut-chinson IV, Shah T. Posttransplant malignancies in solid organ adult recipients: an analysis of the U.S. National Transplant Database. Transplantation. 2012; 94:990–8.
2). Dharnidharka VR, Tejani AH, Ho PL, Harmon WE. Posttransplant lymphoproliferative disorder in the United States: young Caucasian males are at highest risk. Am J Transplant. 2002; 2:993–8.

3). Caillard S, Lamy FX, Quelen C, Dantal J, Lebranchu Y, Lang P, et al. Epidemiology of posttransplant lymphoproliferative disorders in adult kidney and kidney pancreas recipients: report of the French registry and analysis of subgroups of lymphomas. Am J Transplant. 2012; 12:682–93.

4). Ferry JA, Jacobson JO, Conti D, Delmonico F, Harris NL. Lymphoproliferative disorders and hematologic malignancies following organ transplantation. Mod Pathol. 1989; 2:583–92.
5). Starzl TE, Nalesnik MA, Porter KA, Ho M, Iwatsuki S, Griffith BP, et al. Reversibility of lymphomas and lymphoproliferative lesions developing under cyclosporin- steroid therapy. Lancet. 1984; 1:583–7.
6). Buell JF, Gross TG, Woodle ES. Malignancy after transplantation. Transplantation. 2005; 80(2 Suppl):S254–64.

7). Cockfield SM. Identifying the patient at risk for post- transplant lymphoproliferative disorder. Transpl Infect Dis. 2001; 3:70–8.
8). Shroff R, Rees L. The posttransplant lymphoproliferative disorder: a literature review. Pediatr Nephrol. 2004; 19:369–77.
9). Yoon SO, Yu E, Cho YM, Suh C, Kim KM, Han DJ, et al. Posttransplant lymphoproliferative disorders: clin-icopathological analysis of 43 cases in a single center, 1990-2009. Clin Transplant. 2012; 26:67–73.

10). Glotz D, Chapman JR, Dharnidharka VR, Hanto DW, Castro MC, Hirsch HH, et al. The Seville expert work-shop for progress in posttransplant lymphoproliferative disorders. Transplantation. 2012; 94:784–93.

11). Briggs JD. Causes of death after renal transplantation. Nephrol Dial Transplant. 2001; 16:1545–9.

12). Nelson BP, Nalesnik MA, Bahler DW, Locker J, Fung JJ, Swerdlow SH. Epstein-Barr virus-negative posttransplant lymphoproliferative disorders: a distinct entity? Am J Surg Pathol. 2000; 24:375–85.
13). Cox KL, Lawrence-Miyasaki LS, Garcia-Kennedy R, Lennette ET, Martinez OM, Krams SM, et al. An increased incidence of Epstein-Barr virus infection and lymphoproliferative disorder in young children on FK506 after liver transplantation. Transplantation. 1995; 59:524–9.
14). Cruz RJ Jr, Ramachandra S, Sasatomi E, DiMartini A, de Vera M, Fontes P, et al. Surgical management of gastrointestinal posttransplant lymphoproliferative disorders in liver transplant recipients. Transplantation. 2012; 94:417–23.

15). Opelz G, Daniel V, Naujokat C, Fickenscher H, Döhler B. Effect of cytomegalovirus prophylaxis with immunog-lobulin or with antiviral drugs on posttransplant non- Hodgkin lymphoma: a multicentre retrospective analysis. Lancet Oncol. 2007; 8:212–8.
Go to : 
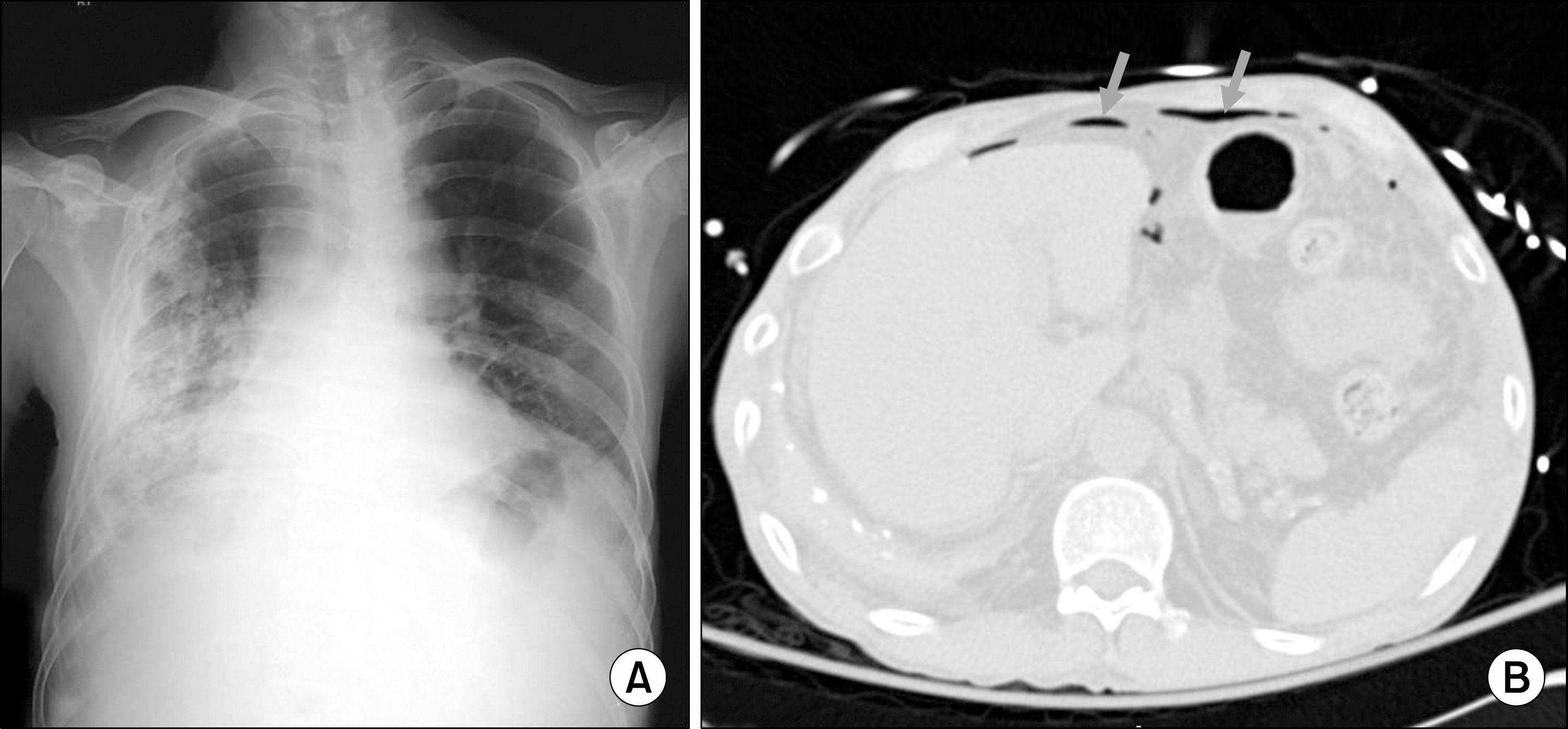 | Fig. 1.(A) Chest X-ray and (B) abdominal computed tomography (CT) scan in the emergency room. In chest X-ray did not show the subdiaphragmatic free air repre-senting bowel perforation. Right lung showed the posttuberculosis sequelae with fibrosis and ate-lectasis combined with pleural ef-fusion. In CT scan showed the in-traperitoneal free air (arrows), and the focus of perforation was not defined. |
 | Fig. 2.The perforation site of small intestine were identified by gross pathology (arrows). (A) Extraluminal view. (B) Intraluminal view. |
 | Fig. 3.(A) Transmural involvement of the small bowel by the lesion (black arrows) (HE stain, ×12.5). (B) The mass consists of predom-inantly large transformed cells containing huge cytoplasm (immunehistochemistry stain, ×200). (C) The large transformed cells were positive for CD20 (immunehistochemistry, ×40). (D) The transformed cells were also positive for blc-2 (immunehistochemistry, ×40). |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download