Abstract
Background
We wanted to explore performing hepatic arterial reconstruction in living donor liver transplantation (LDLT) using right lobe liver grafts and cadaveric liver transplantation (CLT) in a single center.
Methods
Thirty five LDLTs were performed from April 2005 to August 2009. The back wall support suture without twisting was used in most cases. A single RHA was anastomosed to the RHA in 24 patients, to the proper HA in 2 patients, to the RAHA in 4 patients, to the LHA in 2 patients and to an aberrant RHA arising from the SMA in 3 patients. The diameter of the donor RHA was between 1.5 mm and 3.0 mm (mean: 2.5 mm). In the 34 patients who underwent CLT, most of the arterial anastomoses were usually performed using two cuffs at the recipient HA and the GDA bifurcation and a branching point on the donor CHA with running and intermittent stay suture.
Results
The total incidence of HA complication was 4.34% (3/69): 1 HAT (2.85%) occurred in a case of LDLT and 2 HAS (5.88%) occurred in a case of CLT. HAT occurred in 1 recipient on the 1st day following LDLT and 2 HAS occurred in CLT recipients at one and two months, respectively, following LDLT.
Conclusions
HA complications occurred as a mild type of late complication and these complications might not be fatal in CLT. A low incidence of HAT can be achieved with using non-twisting method-guided microsurgical techniques for creating hepatic arterial anastomosis in LDLT. When early HAT occurs, early surgical reconstruction is mandatory for preventing the loss of the graft. Back wall sutures with only single needle suture might be a feasible method for HA microsurgical reconstruction.
Go to : 
References
1). Stange BJ, Glanemann M, Nuessler NC, Settmacher U, Steinmüller T, Neuhaus P. Hepatic artery thrombosis after adult liver transplantation. Liver Transpl. 2003; 9:612–20.
2). Bhattacharjya S, Gunson BK, Mirza DF, Mayer DA, Buckels JA, McMaster P, et al. Delayed hepatic artery thrombosis in adult orthotopic liver transplantation-a 12-year experience. Transplantation. 2001; 71:1592–6.

3). Settmacher U, Stange B, Haase R, Heise M, Steinmüller T, Bechstein WO, et al. Arterial complications after liver transplantation. Transpl Int. 2000; 13:372–8.

4). Takatsuki M, Chiang YC, Lin TS, Wang CC, Concejero A, Lin CC, et al. Anatomical and technical aspects of hepatic artery reconstruction in living donor liver transplantation. Surgery. 2006; 140:824–8.

5). Uchiyama H, Hashimoto K, Hiroshige S, Harada N, Soejima Y, Nishizaki T, et al. Hepatic artery reconstruction in living-donor liver transplantation: a review of its techniques and complications. Surgery. 2002; 131(1 Suppl):S200–4.

6). Wei WI, Lam LK, Ng RW, Liu CL, Lo CM, Fan ST, et al. Microvascular reconstruction of the hepatic artery in live donor liver transplantation: experience across a decade. Arch Surg. 2004; 139:304–7.
7). Broelsch CE, Whitington PF, Emond JC, Heffron TG, Thistlethwaite JR, Stevens L, et al. Liver transplantation in children from living related donors. Surgical techniques and results. Ann Surg. 1991; 214:428–37.
8). Miyagi S, Enomoto Y, Sekiguchi S, Kawagishi N, Sato A, Fujimori K, et al. Microsurgical back wall support suture technique with double needle sutures on hepatic artery reconstruction in living donor liver transplantation. Transplant Proc. 2008; 40:2521–2.

9). Shirouzu Y, Kasahara M, Morioka D, Sakamoto S, Taira K, Uryuhara K, et al. Vascular reconstruction and complications in living donor liver transplantation in infants weighing less than 6 kilograms: the Kyoto experience. Liver Transpl. 2006; 12:1224–32.

10). Kim BW, Won JH, Lee BM, Ko BH, Wang HJ, Kim MW. Intraarterial thrombolytic treatment for hepatic artery thrombosis immediately after living donor liver transplantation. Transplant Proc. 2006; 38:3128–31.

11). Kaneko J, Sugawara Y, Akamatsu N, Kishi Y, Niiya T, Kokudo N, et al. Prediction of hepatic artery thrombosis by protocol Doppler ultrasonography in pediatric living donor liver transplantation. Abdom Imaging. 2004; 29:603–5.

12). Katz E, Fukuzawa K, Schwartz M, Mor E, Miller C. The splenic artery as the inflow in arterial revascularization of the liver graft in clinical liver transplantation. Transplantation. 1992; 53:1373–4.

13). Cherqui D, Riff Y, Rotman N, Julien M, Fagniez PL. The recipient splenic artery for arterialization in orthotopic liver transplantation. Am J Surg. 1994; 167:327–30.

14). Ikegami T, Kawasaki S, Hashikura Y, Miwa S, Kubota T, Mita A, et al. An alternative method of arterial reconstruction after hepatic arterial thrombosis following living-related liver transplantation. Transplantation. 2000; 69:1953–5.

15). Yang Y, Yan LN, Zhao JC, Ma YK, Huang B, Li B, et al. Microsurgical reconstruction of hepatic artery in A-A LDLT: 124 consecutive cases without HAT. World J Gastroenterol. 2010; 16:2682–8.

16). Yan S, Zhang QY, Yu YS, He JJ, Wang WL, Zhang M, et al. Microsurgical reconstruction of hepatic artery in living donor liver transplantation: experiences and lessons. Hepatobiliary Pancreat Dis Int. 2009; 8:575–80.
17). Okazaki M, Asato H, Takushima A, Nakatsuka T, Sarukawa S, Inoue K, et al. Hepatic artery reconstruction with double-needle microsuture in living-donor liver transplantation. Liver Transpl. 2006; 12:46–50.

18). Okochi M, Ueda K, Hirose T, Okochi H, Watanabe H, Suzuki Y, et al. A modified technique for hepatic artery reconstruction in living donor liver transplantation. Microsurgery. 2010; 30:541–4.

19). Mazzaferro V, Esquivel CO, Makowka L, Belle S, Kahn D, Koneru B, et al. Hepatic artery thrombosis after pediatric liver transplantation-medical or surgical event? Transplantation. 1989; 47:971–7.
20). Silva MA, Jambulingam PS, Gunson BK, Mayer D, Buckels JA, Mirza DF, et al. Hepatic artery thrombosis following orthotopic liver transplantation: a 10-year experience from a single centre in the United Kingdom. Liver Transpl. 2006; 12:146–51.

21). Matsuda H, Yagi T, Sadamori H, Matsukawa H, Shinoura S, Murata H, et al. Complications of arterial reconstruction in living donor liver transplantation: a singlecenter experience. Surg Today. 2006; 36:245–51.

22). Jiang XZ, Yan LN, Li B, Zhao JC, Wang WT, Li FG, et al. Arterial complications after living-related liver transplantation: singlecenter experience from West China. Transplant Proc. 2008; 40:1525–8.

23). Sheiner PA, Varma CV, Guarrera JV, Cooper J, Garatti M, Emre S, et al. Selective revascularization of hepatic artery thromboses after liver transplantation improves patient and graft survival. Transplantation. 1997; 64:1295–9.
Go to : 
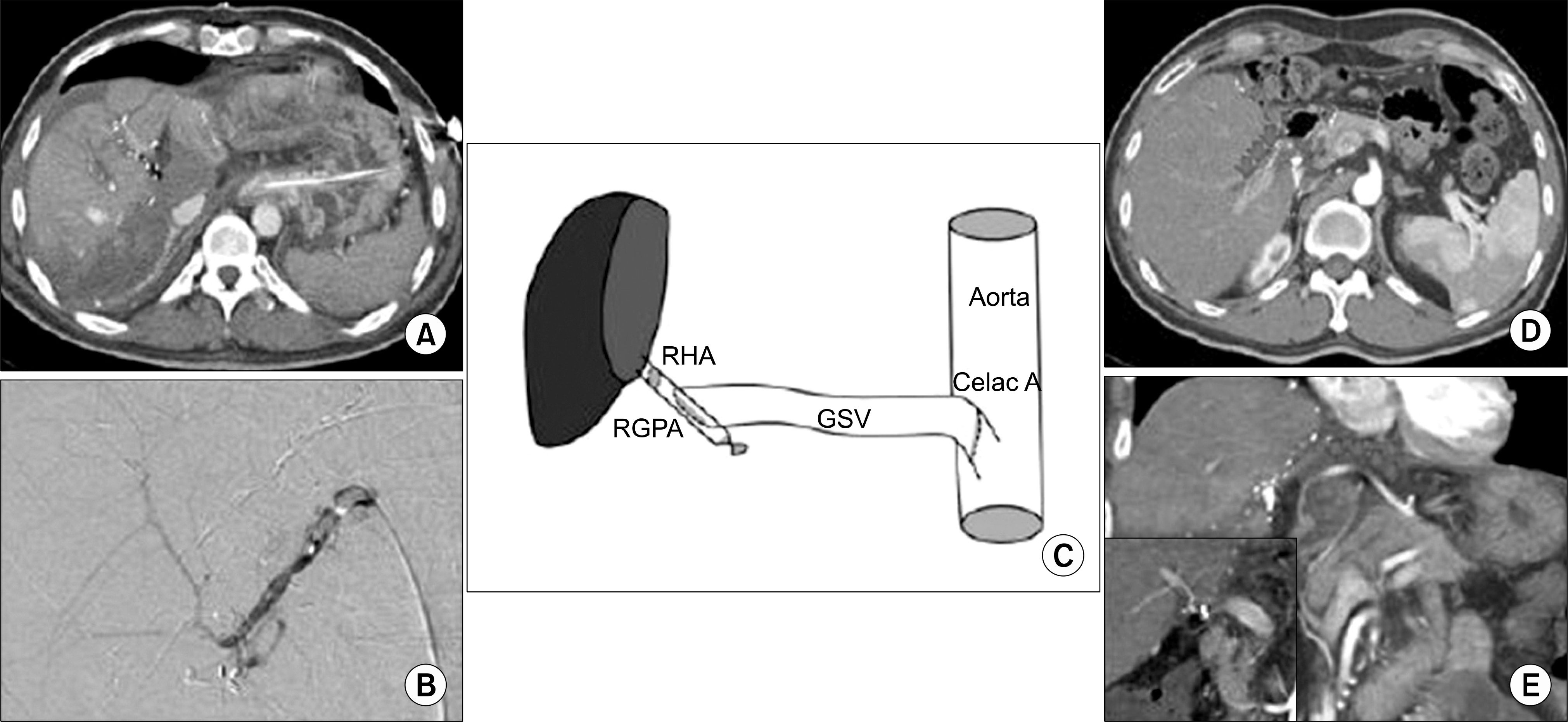 | Fig. 1.Ischemic necrosis followed by hepatic artery stenosis. CT shows ischemic necrosis followed by hepatic arterial thrombosis, which occurred at segment VI at 1st postoperation day. Hepatic angiography show arterial vasospam ans thrombus occurred at the right gastroepiploic artery (A, B). Hepatic flow can be preserved after jump graft by using great saphenous vein from supraceliac aorta to right gastroepiploic artery (C). The ischemic necrosis portion show relatively good parenchymal regeneration and duct dilatation in 7 months later (D, E). |
 | Fig. 2.Hepatic angiography showing arterial kicking and stenosis. Hepatic angiography show arterial kicking and stenosis at the anastomosis of CHA in CLT. The kicking of anastomosis site was revised by ballooning and stenting (A, B). Intimal dissection and stenosis in non-anastomotic site was relieved by ballooning and stenting (C, D). |
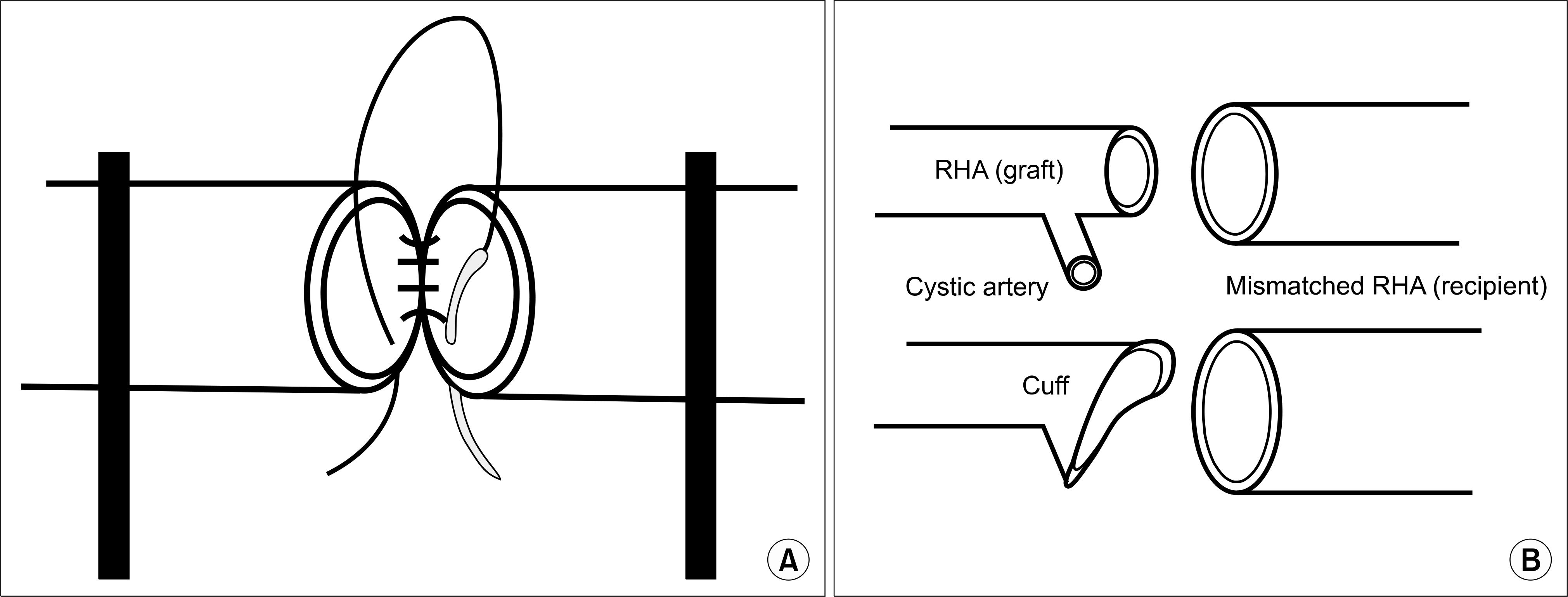 | Fig. 3.Schematic drawing of the microvascular anastomosis. Schematic drawings show the microvascular anastomosis. The recipient and graft artery are clamped without twisting. The first suture is placed at upper and posterior wall at most deep portion of two arteries (A), and hepatic artery cuff by using cystic attery to overcome size discrepancy (B). |
Table 1.
Anastomosed arterial feature and complication
| Type | Anastomoed artery | Size (mean, mm) | n (%) | SD | Complication |
|---|---|---|---|---|---|
| LDLT | RHA-RHA | 2.6 | 24 (68.5%) | 0.26 | 2/24 |
| RHA-PHA | 3.2 | 2 (5.7%) | 0.1 | – | |
| RHA-RAHA | 1.3 | 4 (11.4%) | 0.07 | – | |
| RHA-LHA | 2.4 | 2 (5.7%) | 0.1 | – | |
| RHA-aberrant RHA a | 2.5 | 3 (8.5%) | 0.08 | – | |
| CLT | CHA-CHA | 13 | 30 | 1.57 | 1/30 |
| CHA-RHA | 7 | 4 | 0.07 | – |
Table 2.
Clinical data from 3 patients with hepatic arterial complication after liver transplantation




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download