Abstract
Purpose of this study was to evaluate the influence of a lotion on the bacterial community in the human forearm skin. The chemical- and natural-based lotions were applied on the left and right inner forearm skins, respectively, of 14 participants, who cleansed forearm skin using sterilized cotton swabs. The germs on cotton swabs were analyzed using libraries of PCR amplicons. The genetic diversity of the bacterial communities detected on the natural-based lotion-applied skin (NLS) was significantly higher than that of the bacterial communities on the chemical-based lotion-applied skin (CLS) in all participants, except two. The diversity was estimated based on operational taxonomic unit (OTU), Chao1, Shannon, and Simpson indices. Bacterial communities obtained from the CLS and NLS were phylogenetically separated into 5 and 3 monophyletic groups, respectively, based on lotion types. The taxonomic distribution of the bacterial communities, which were composed of 198 genera in 14 phyla in the CLS and NLS, respectively, was irregularly and biasedly separated into 2 groups based on the lotion types. Among the 14 phyla, Proteobacteria, Firmicutes, Bacteroidetes, and Actinobacteria were found to be relatively dominant, and 15 of the 198 genera, including Methylobacterium, Propionibacterium, Pseudomonas, Staphylococcus, Streptococcus, and Bacillus were relatively dominant (>0.5%). The taxonomic distribution of dominant bacterial communities from CLS and NLS was irregularly and biasedly separated without relation to the lotion types. In conclusion, the chemical- and natural-based lotions were responsible for changing or influencing the genetic diversity, phylogenetic separation, and taxonomic distribution of skin bacterial communities.
Go to : 
REFERENCES
1). Segre JA. Epidermal barrier formation and recovery in skin disorders. J Clin Invest. 2006; 116:1150–8.

2). Grice EA, Kong HH, Conlan S, Deming CB, Davis J, Young AC. Topographical and temporal diversity of the human skin microbiome. Science. 2009; 324:1190–2.

5). Cogen AL, Nizet V, Gallo RL. Skin microbiota: A source of disease or defence? Br J Dermatol. 2008; 158:442–55.

6). Till AE, Goulden V, Cunliffe WJ, Holland KT. The cutaneous microflora of adolescent, persistent and late-onset acne patients does not differ. Br J Dermatol. 2000; 142:885–92.
8). Yarwood JM, Bartels DJ, Volper EM, Greenberg EP. Quorum sensing in Staphylococcus aureus biofilms. J Bacteriol. 2004; 186:1838–50.
9). Pesci EC, Pearson JP, Seed PC, Iglewski BH. Regulation of las and rhl quorum sensing in Pseudomonas aeruginosa. J Bacteriol. 1997; 179:3127–32.

10). Fuqua C, Winans SC, Greenberg EP. Census and consensus in bacterial ecosystems: the LuxR-LuxI family of quorum-sensing transcriptional regulators. Annu Rev Microbiol. 1996; 50:727–51.

11). Latifi A, Foglino M, Tanaka K, Williams P, Lazdunski A. A hierarchical quorum sensing cascade in Pseudomonas aeruginosa links the transcriptional activators LasR and R hIR (VsmR) to expression of the stationary-phase sigma factor Rpos. Mol Microbiol. 1996; 21:1137–46.
12). de Almeida e Borges LF, Silva BL, Gontijo Filho PP. Hand washing: Changes in the skin flora. Am J Infect Control. 2007; 35:417–20.
13). Wibowo C, Ng KM. Product-centered processing: manufacture of chemical-based consumer products. Am Inst Chem Eng J. 2002; 48:1212–30.

14). Gfatter R, Hackl P, Braun F. Effects of soap and detergents on skin surface pH, stratum corneum hydration and fat content in infants. Dermatology. 1997; 195:258–62.

15). Chen Q. Evaluate the effectiveness of the natural cosmetic product compared to chemical-based products. Int J Chem. 2009; 1:57–9.

16). Hoolnad KT, Bojar RA. Cosmetics: what is their influence on the skin microflora? Am J Clin Dermatol. 2002; 3:445–9.
17). Boireau-Adamezyk E, Baillet-Guffroy A, Stamatas GN. Age-dependent changes in stratum corneum barrier function. Skin Res Technol. 2014; 20:409–15.

18). Wick G, Grunbeck-Loebenstein B. The aging immune system: primary and secondary alterations of immune reactivity in the elderly. Exp Gerontol. 1997; 32:401–13.

19). Proksch E, Fölster-Holst R, Jensen JM. Skin barrier function, epidermal proliferation and differentiation in eczema. J Dermatol Sci. 2006; 43:159–69.

20). Harding CR, Watkinson A, Rawlings AV, Scott IR. Dry skin, moisturization and corneodesmolysis. Int J Cosmet Sci. 2000; 22:21–52.

21). Gomez-Alvarez V, Teal TK, Schmidt TM. Systematic artifacts in metagenomes from complex microbial communities. ISME J. 2009; 3:1314–7.

22). Chou HH, Holmes MH. DNA sequence quality trimming and vector removal. Bioinformatics. 2001; 17:1093–104.

23). Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol. 2011; 28:2731–9.

24). Fredricks DN. Microbial ecology of human skin in health and disease. J Investig Dermatol Symp Proc. 2001; 6:167–9.

25). Holland KT, Bojar RA. Cosmetics: what is their influence on the skin microflora? Am J Clin Dermatol. 2002; 3:445–9.
26). Gao Z, Tseng CH, Pei Z, Blaser MJ. Molecular analysis of human forearm superficial skin bacterial biota. Proc Natl Acad Sci U S A. 2007; 104:2927–32.

27). Grice EA, Kong HH, Renaud G, Young AC, Bouffard GG, Blakesley RW, et al. A diversity profile of the human skin microbiota. Genome Res. 2008; 18:1043–50.

28). Eloe-Fadrosh EA, Rasko DA. The human microbiome: from symbiosis to pathogenesis. Annu Rev Med. 2013; 64:145–63.

29). Leeming JP, Holland KT, Cunliffe WJ. The microbial ecology of pilosebaceous units isolated from human skin. J Gen Microbiol. 1984; 130:803–7.

30). de Almeida e Borges LF, Silva BL, Gontijo Filho PP. Hand washing changes in the skin flora. Am J Infect Control. 2007; 35:417–20.
31). Kampf G, Kramer A. Epidemiologic background of hand hygiene and evaluation of the most important agents for scrubs and rubs. Clin Microbiol Rev. 2004; 17:863–93.

32). Bowe WP, Puqliese S. Cosmetic benefits of natural ingredients. J Drugs Dermatol. 2014; 13:1021–5.
33). You JY, Park SH, Hwang IA, Jo SJ, Huh CH, Youn SW, et al. A clinical study on the effect of a cream containing ramulus mori extract and tea tree oil on acne vulgaris and aerobic skin flora. Korean J Dermatol. 2003; 41:1136–41.
34). Blanpain C, Fuchs E. Epidermal homeostasis: a balancing act of stem cells in the skin. Nat Rev Mol Cell Biol. 2009; 10:207–17.

35). Zoetendal EG, Vaughan EE, de Vos WM. A microbial world within us. Mol Microbiol. 2006; 59:1639–50.

Go to : 
 | Figure 1.A gathering method of cotton swabs for protection or minimization of contamination from the participants' (samplers') hands |
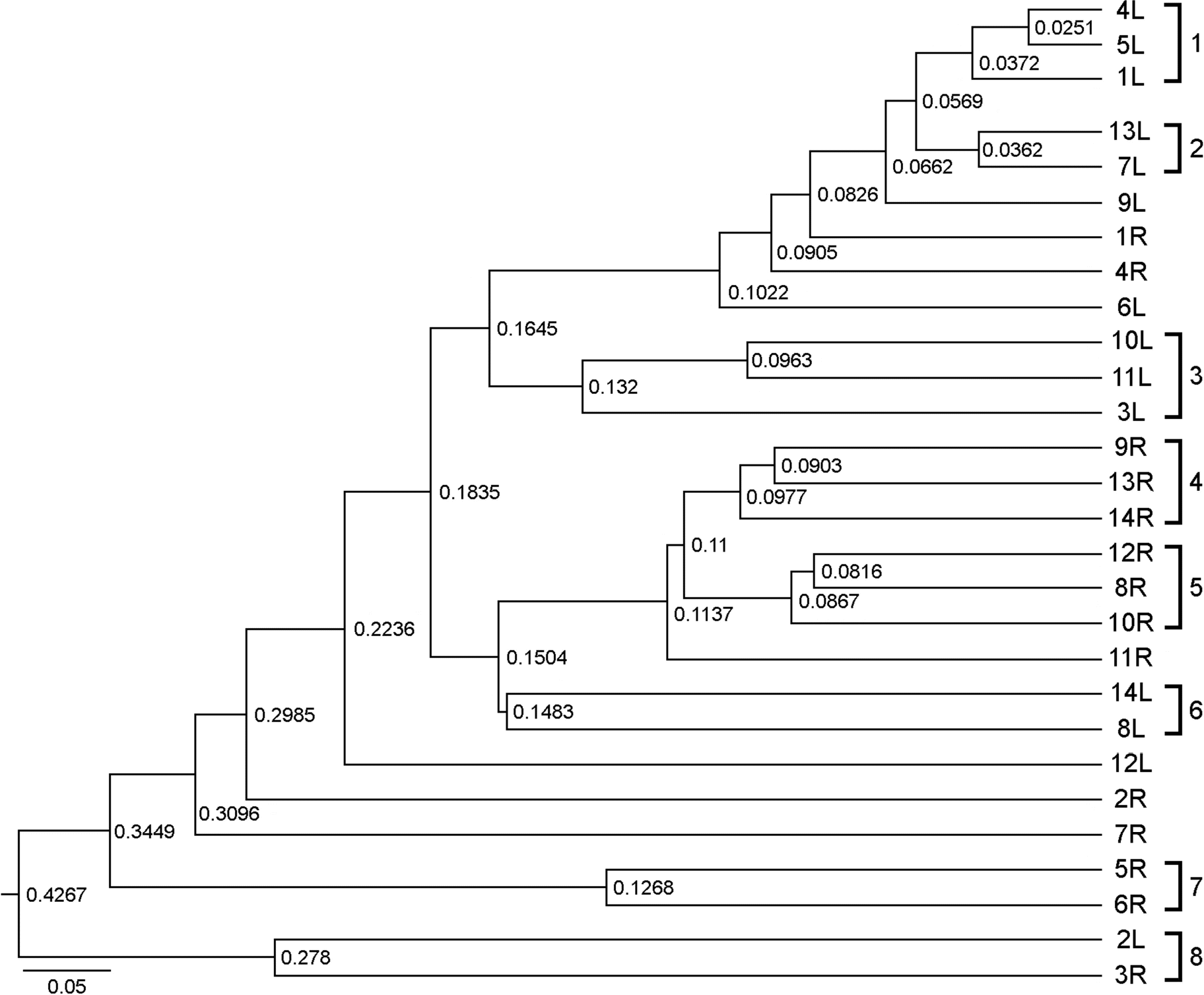 | Figure 2.Neighbor-joining phylogenetic tree (dendrogram) of bacterial communities originated from the chemical-based lotion (1L~14L) and natural-based lotion (1R~14R)-applied skin of 14 participants based on pairwise Bray-Curtis similarity. Branch length in the tree is proportional to the numbers of nucleotide substitutions as measured by the scale bar (5% dissimilarity). |
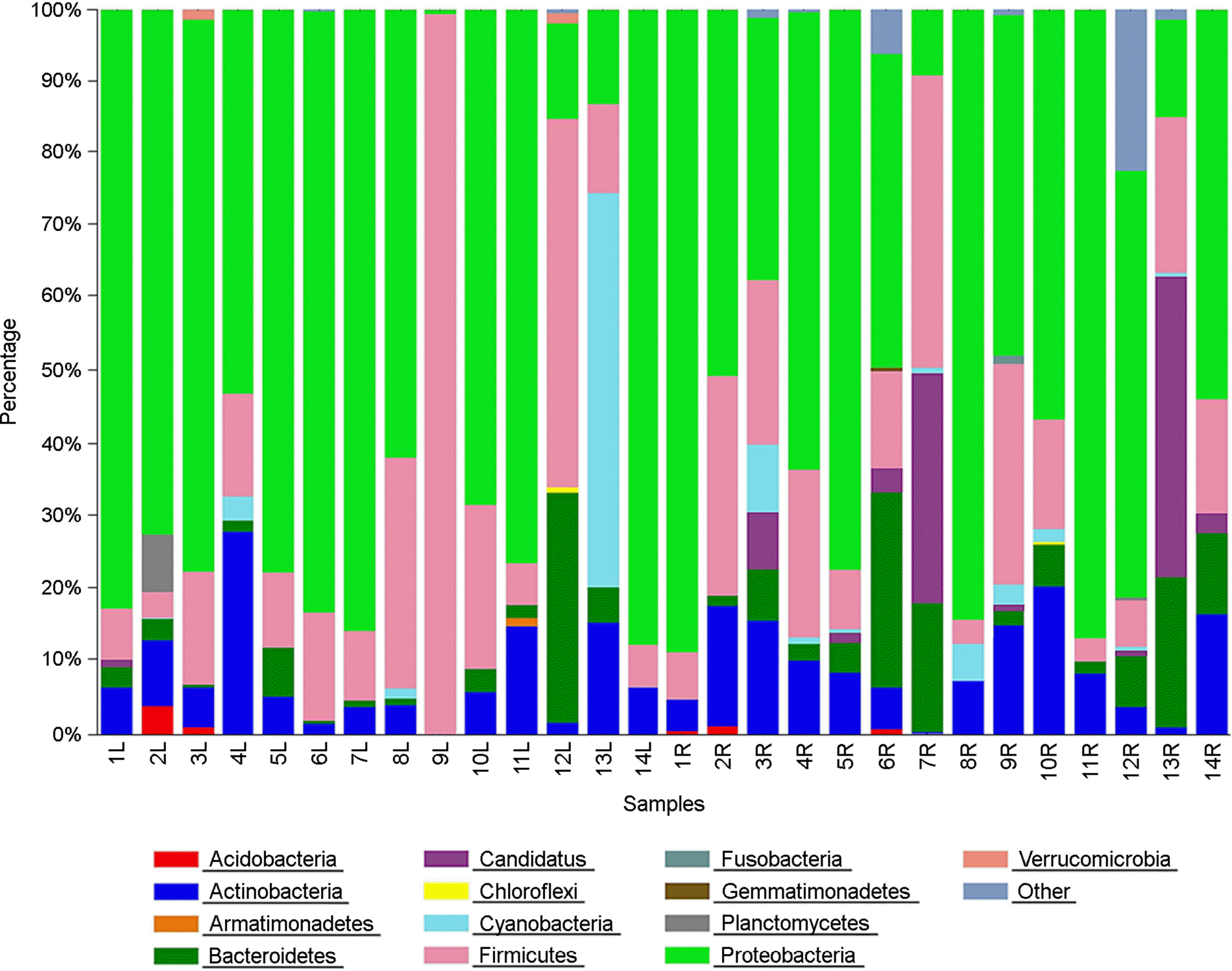 | Figure 3.Distribution of the 16S rRNA gene sequences across bacterial phyla in 28 skin bacterial communities originated from chemical-based lotion (1L~14L) and natural-based lotion (1R~14R)-applied skin of 14 participants. |
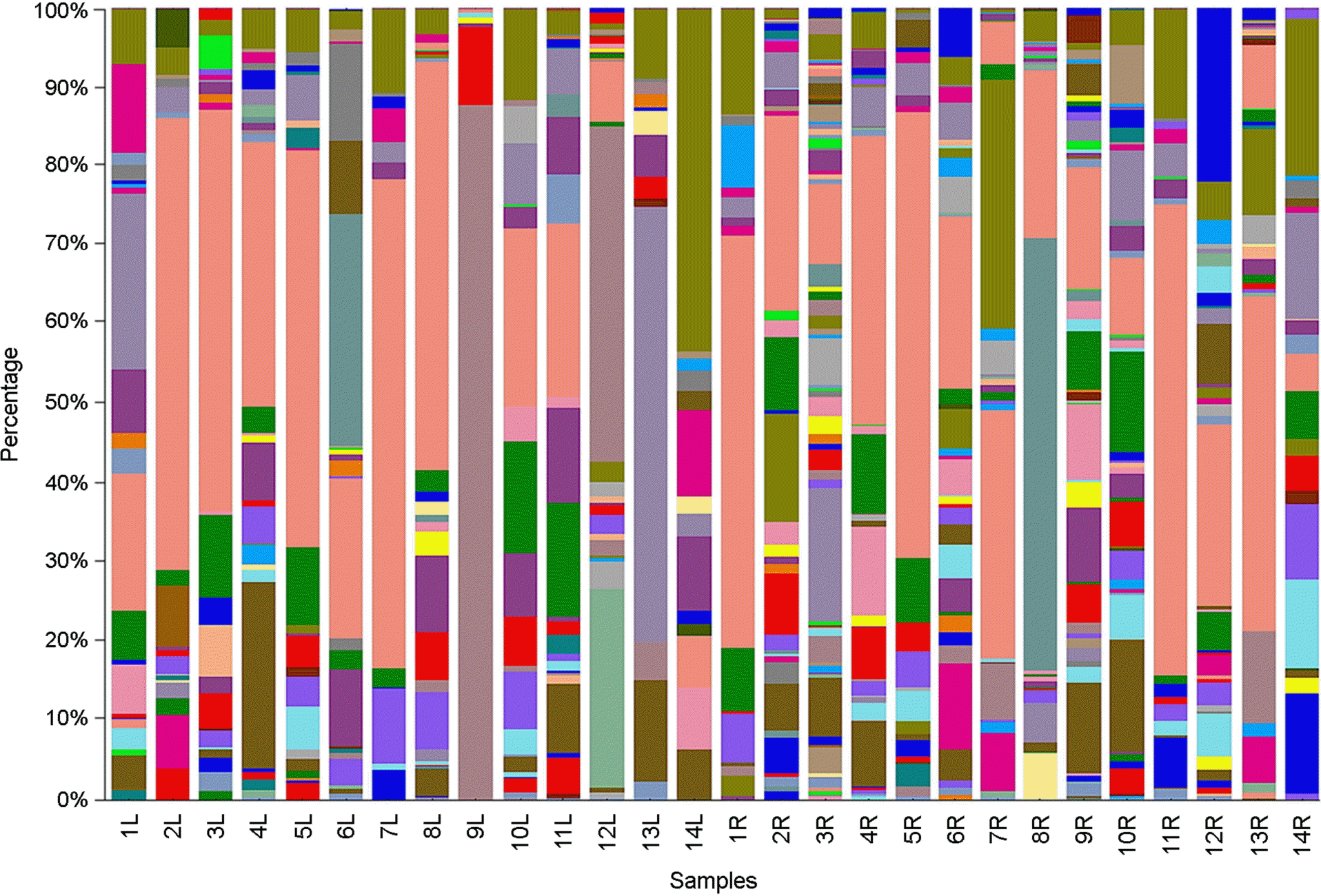 | Figure 4.Distribution of the 16S rRNA gene sequences across bacterial genera in 28 skin bacterial communities originated from chemical-based lotion (1L~14L) and natural-based lotion (1R~14R)-applied skin of 14 participants. |
 | Figure 5.Taxonomic diversity of bacterial genera (1 to 198) originated from chemical-based lotion (1L~14L) and natural-based lotion (1R~14R)-applied skin of 14 participants. Color bands are legends and genus names are explanation for taxonomic distribution in Figure 4. The average occupation rates of the relatively dominant genera (>0.5%) were quantitatively represented in parentheses. |
Table 1.
Ingredients of chemical-based and natural-based lotions sold in cosmetic shops
Table 2.
Diversity indices calculated for skin bacterial communities originated from the chemical-based lotion (L) and natural-based lotion (R)-applied skin of 14 participants




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download