Abstract
Zika virus (ZIKV) was spread to both eastward and westward from Uganda where the virus was identified approximately in 1947 by a group of arbovirus researchers. In 2015, ZIKV reached Americas with major outbreaks in Brazil. Most countries with mosquito transmitted ZIKV infection are located in tropical and subtropical areas, where ZIKV is endemic with other flaviviruses, including JEV, dengue and yellow fever virus. Approximately 40 countries in Central and South Americas and territories in South Pacific Islands and South East Asia show autochthonous ZIKV endemics. American lineage of ZIKV is known significantly to be mutated in susceptibility to host and in pathogenicity from Asian and Asian lineages approximately since 2014. Early and specific identification of ZIKV infection is very important for the effective management of patients. First of all, optimal collection of specimens for the laboratory diagnosis is required for both nucleic acid testing (NAT) and serological tests. Specimens for NAT tests and serological tests should be determined by the available laboratory resources, work-flow in each laboratory and the geographic areas of specimen collected in addition to days after showing symptoms. Testing strategy for specific differentiation among flaviviruses will vary depending on the prevalence of viruses known to be circulating in the area where the patients were exposed. NAT will be employed for the patients presenting with onset of symptoms less than 7 days. Advanced diagnostic technologies should be continuously developed for the increase of specificity and sensitivity of ZIKV diagnosis.
Zika virus (ZIKV) was isolated and identified from a rhesus macaque by scientists working on yellow fever in Zika Forest, Uganda in 1947 (1). Subsequently, the virus was isolated from a human in Africa 5 years later. The monkey virus must have been converted to be virulent virus when the virus changed host to another species of monkey and human (2). Historically, ZIKV in early period after emergence was rarely associated with diseases in humans (http://www.who.int/emergencies/zika-virus/en/) (3). ZIKV has continued spreading to eastward and westward throughout the world for more than sixty years without particular attention until 2014 (45). Nearly 70 years later, the ZIKV found public consciousness because the virus was suspected of association with brain damage in the pregnant women in America including Brazil.
From early 2016, millions of persons with ZIKV infection were found with increases of infants with microcephaly in Braool, Brazil (6). As of mid-2016, ZIKV is spreading to the Americas, the Pacific and Southeast Asia and also affects several islands in the Pacific Islands (57). Approximately 50 countries or territories worldwide have shown the autochthonous epidemics of ZIKV (48). ZIKV has obtained public concerns since diseases like microcephaly and Guillain-Barre syndrome were increased in several countries in America, mainly in Brazil (9). Therefore, health authorities worldwide and WHO have had special attentions on the spread of Zika fever.
Naturally it is expected that more persons would be returned to their original countries after their infection in ZIKV epidemic areas, therefore, intensive prevention policies are required to be prepared for those persons infected with ZIKV and general public. Furthermore, increasing necessities for developments of advanced diagnostic reagents are present for rapid and sensitive detection of the persons with ZIKV infection or suspected symptoms. In the laboratory diagnosis of persons with ZIKV infections, it is very difficult to perform specific detection of infected persons in the areas with simultaneous flavivirus transmissions because of nonspecific reactions (2). For example, positive results of serological tests to ZIKV in Korea could be caused by past natural infection or vaccination of Japanese encephalitis virus (JEV), especially in case of old persons.
In addition to diagnostic reagents, developments of vaccine and therapeutics are required for the better control of the persons related (10). For the successful development of ZIKV vaccine, understanding on biology, epidemics and other aspects of ZIKV in addition to diagnostic technologies is very important. In this review, advances on ZIKV epidemics and diagnostic technologies are introduced.
As described above, ZIKV was accidently discovered from Macaca mulatta (an old monkey species) in Entebbe, Uganda (1). The ZIKV were isolated from mosquito populations (Aedes africanus) from the same locations in 1948 (1). The same viruses were also isolated from indigenous monkey species, namely different from Macaca mulatta species (135). The ZIKV is interpreted to be adapted to Asian monkey species from indigenous monkey species, which means translocation of host species. Three humans with ZIKV infection were reported for the first time in Nigeria in 1954 (211). Furthermore, first ZIKV infection in human in Asia was confirmed in Malaysia in 1969 (12). First patient with ZIKV was diagnosed in Java, Indonesia in 1977. Various evidences on serological and virological tests suggest wide spread of ZIKV in both Africa and Southeast Asia during the period of 1960-70s (1213). ZIKV reached French Polynesia in the Southeast Asia area with concomitant increase of patients with Guillain-Barre syndrome in this area in 2007 (47). However, the ZIKV did not show any significant virulence during the course of the transmission in Southeast areas (24). Directions and time of ZIKV transmission from original discovery area to the areas of large outbreaks are summarized in Fig. 1.
ZIKV infections in human and mosquitos were confirmed around 2007 when nearly 4,000 sera from patients of febrile diseases from Pacific Islands with dengue and chikungunya virus infection and two Aedes albopictus pools collected in the regions were tested (4). Up to 2013, many islands in South Pacific Ocean showed ZIKV prevalence (13) when tested using molecular and serological tests of ZIKV. Then, it is presumed that ZIKV transmitted to mainland America from one of islands with ZIKV epidemics (213). Also during this outbreak, there must be the evidence of risk that ZIKV could be also transmitted through blood banks, and ZIKVs were also detected in semen, saliva and urine around 2013 (1213).
From numerous patients with rash and mild fever and arthralgia, ZIKV infections were first detected in Northeastern Brazil and patient numbers increased during the first months of 2015 (691415). For theses outbreaks, ZIKV infections were announced to be confirmed as suspected etiology by Salvador city authority of Brazil. Several patients with dengue-like illness showed positive results to serum samples when tested using reverse transcription polymerase chain reaction (RT-PCR) methods (13). ZIKV rapidly reached nearly all districts of Brazil by the end of 2015. It is believed that ZIKV continued to spread to other neighboring countries in American continent around this period.
In addition, there was the unusual rise in the number of newborns with microcephaly in northeastern Brazil that began months after the increases of cases (6151617). The rapid increase of microcephaly and Guillain-Barré syndrome in Brazil and other neighboring areas alarmed World Health Organization (WHO) and many other governments for Public Health Emergency of International Concern (6). On the other hand, this emergency action by WHO is known to be lifted on the date of 22, November 2016 (18).
Data on the prevalence of ZIKV are updated in the home page sites of WHO, Pan American Health Organization (PAHO), Center for Disease Control and Prevention (CDC), European CDC (ECDC) and Korean CDC (KCDC) (61819). ZIKV prevalence is classified to several groups by countries and territories (Fig. 2). WHO reported 68 countries with present epidemics of ZIKV and 7 countries with past epidemics worldwide, as of October 28, 2016. First of all, the areas with the biggest ZIKV epidemics are in Brazil and neighboring countries. 47 countries and territories are reported presently to be autochthonous infections in Central and South America, while United States of America is only country with autochthonous transmission in North America (6181920). Officially, the Brazilian Ministry of Health estimated that number of persons with ZIKV infections are approximately 1,500,000 as of middle 2016 (615). In addition, Colombia which showed many autochthonous patients next to Brazil in America was also greatly affected by ZIKV epidemics (15). In the US, 4,115 persons who returned to the country after infection from abroad were reported as of November 2016, whereas 139 persons were reported in the areas of Florida, American Samoa and Puerto Rico and US Virgin Islands. Sexually transmitted cases are not reported for US territories with local transmission of ZIKV. Detailed epidemiological surveys in US are not possible to determine whether infections occurred due to mosquito-borne or sexual transmission. In Africa, four African countries have reported autochthonous epidemics of ZIKV. Cape Verde reported approximately 8,000 cases of ZIKV as of middle 2016. Twelve countries/or territories located in Oceania reported ZIKV epidemics, the countries include Samoa, Fiji and Cook Islands. Solomon Islands reported 302 confirmed cases of ZIKV infection. Eleven Asian countries are known to be ZIKV prevalent as of late October 2016. Singapore reported 435 confirmed autochthonous cases and Thailand is next to Singapore by reporting 392 autochthonous cases. Those countries are followed by 39 cases of Vietnam and 19 cases of Philippines. Largely, these reported numbers can be influenced by testing systems and strategies like epidemiological surveys, which means that the actual severity of prevalence cannot be directly compared with between territories and/or countries.
ZIKV is mainly transmitted by female mosquito bites. Major vector species of ZIKV transmission are Aedes aegypti and also rarely Aedes albopictus. Most of these Aedes mosquitoes are distribute mainly in tropical or subtropical areas of the world. Presently, natural host of ZIKV is not known in detail even in the areas with big epidemics. However, unlike other flaviviruses, e.g., JEV, ZIKV is known to be also transmitted by non-arboviruses routes, namely by transfusion and sexual activity through body fluids including urine. Viral loads of flaviviruses in human body fluids are recommended to be investigated for the elucidation of ZIKV role in the non-arboviral transmission. Furthermore, the possibility of vertical transmission is suggested due to the potential link between microcephaly and pregnant women with ZIKV infection (421).
No autochthonous infection of ZIKV has been reported so far in Korea. As of 24 September 2016, it was confirmed that 14 persons had been found infected with ZIKV in Korea. Latest person found with ZIKV infection in Korea is a 34-year-old man who recently traveled to Thailand and was surveyed to be infected with the mosquito-borne virus. All of the persons with ZIKV in Korea were epidemiologically surveyed to be infected by mosquito biting in foreign countries (Table 1). Of 14 Korean persons with ZIKV, the 10 persons traveled Southeast Asian countries including the Philippines, Vietnam and Thailand, while four others made trips to Central and South American countries such as Brazil. So far, no autochthonous ZIKV infected persons were found within Korea (Fig. 3). The possibility of autochthonous ZIKV epidemics in Korea is assessed to be very low in Korea because of low prevalence of Aedes albopictus species within the country.
Most countries throughout the world have followed WHO policies (22) for the effective control of Zika epidemics. Those policies largely include travel warnings for the people, advising their citizens to delay pregnancy and/or sexual activities for 1~6 months after staying in endemic areas and avoiding mosquito bite (23). The information on current Zika transmissions and guidance by WHO and government authorities are useful to evaluate the risk for people who have recently returned from or considering travel to countries with autochthonous prevalence.
Avoiding Aedes species mosquito biting in widespread ZIKV epidemic areas is recommended for the prevention of transmission (142425). The guideline of the CDC and WHO recommends wearing long sleeved clothes in the areas with autochthonous transmission and use of mosquito repellent agents during activity in fields. All the women from the areas of Zika infections who expect to be pregnant should delay pregnancy at least 2 months or longer. Furthermore, partners of women expecting pregnancies should not have sexual activity with their partners for 2 months. In addition, persons infected with ZIKV must avoid sexual contact at least for six months. Neither vaccine nor effective therapeutic drugs are available so far, even though some research reported the efficacy of interferons (16). In addition, no specific laboratory animal for vaccine or therapeutics except known natural host is presently available (1719). As of November 2016, ZIKV is lifted from quarantine diseases throughout the world so far (18).
FDA in the US established the “revised recommendations for reducing the risk of ZIKV transmission by blood and blood components (http://www.fda.gov/BiologicsBlood-Vaccines/GuidanceComplianceRegulatoryInformation/Guidances/Blood/)”. The main contents of the recommendations include the regulations that all the blood sources from the persons with ZIKV must be excluded from use of blood donations or blood components for securing the safety from the virus contaminations (1026). It is presumed that KFDA also follows the contents of the recommendations for the safety largely (18).
Flaviviruses are enveloped with single stranded positive RNA viruses and ZIKV is inferred to be about 50 nm in diameter, which is compatible with the observations performed for ZIKV (2728). It has been tested that ZIKV was most stable at pH of 6.8~7.4 and inactivated at under pH 6.2 and over pH 7.8. It is known that other flaviviruses, such as JEV, are very sensitive to narrow changes of pH (13). It might be interesting to investigate the sensitivity range of ZIKV to pH, considering highly narrow pH sensitivity of JEV. ZIKV can also be inactivated by many chemicals, including potassium permanganate, ether, and temperatures of 58℃ for 30 minutes as well (27). It is well known that the differences in ether sensitivity between JEV and polio virus were used even 1960s because JEV is inactivated, while polio virus is not when treated with ether. The difference between two small viruses were crudely used for preliminary diagnosis of patients with neurological diseases in 1950-1960s in Japanese and polio endemic areas.
It is elucidated that the ZIKV genome was 10,794 nucleotides in length (13). The genome contains a single open reading frame (ORF). The single ORF is sided by two untranslated regions (UTR) located at the 5' and 3' ends of the genome. These basic properties are same among ZIKV isolates even though ZIKV isolates are different in length and nucleotide sequences (13). There are differences on particular site even between ZIKV isolates, depending on different passage history, such as those of the prototypic strain ZIKV MR766, indicating that passage history influences glycosylation sites (81319272930).
Unlike other flaviviruses, which is difficult to find viruses in blood or body fluids soon after the infection, ZIKVs are present in various body fluids including semen, saliva (31), female genital secretions (32) and urine for extended period of time (2021273334). On the other hand, it has to be compared the period of remaining viremia of JEV and ZIKV in Korea using recent advanced molecular techniques, because it has been known that JEV detection period might be corrected by the use of more sensitive molecular technologies. For example, it is known that viremia in the persons with JEV infections disappears soon after showing symptoms.
In the screening against anti-ZIKV therapeutics, glutarimide antibiotics, e.g., lactimidomycin, are being tested against various RNA virus including ZIKV and dengue viruses (35). The antibiotic has shown certain level of anti-ZIKV activities, acting as a direct inhibitor of protein translation in ribosomes, in addition to antimicrobial activities (363738). However, no practical drugs and vaccine against ZIKV are available so far.
ZIKV is classified into the Flavivirus genus within the Flaviviride family which includes dengue virus, yellow fever virus and JEV (27). No details on natural host of ZIKV are known so far except natural vertebrate host range, generally including primates as its amplification and reservoir hosts. Thus, the known evolution of ZIKV has been associated mostly with sampling of strains from the enzootic cycle in Africa and rapidly expansion to Asia and Oceania and recently by its introduction into Central and South America including Brazil (30).
It is also presumed that ZIKV has been evolved by the global expansion of Aedes aegypti, the urbanization of human population and increase of international travels. The origin of known ZIKV strains is supposed to occur in early 1900s in western Africa (13). ZIKV is believed to have travelled westwards to Nigeria, in addition to eastwards to Eastern Africa. It is known by genome sequences that ZIKV was diverged into two major lineages of African and Asian/American after its origin. African strains are composed of two groups. First group's prototype, MRR 766 strain, originated from Senegal and neighboring countries during the period of 1947-2001, while the 2nd cluster is composed of strains isolated in Nigeria and Senegal during the period of 1968-1997 (13). Presently, it is supposed that more than two distinct lineages are circulating in Senegal.
On the other hand, Asian cluster is based on the proto type, P6-740 strain, isolated in Malaysia in 1966. Asian clusters include strains isolated in the areas of Southeast Asia and the islands of the South Pacific Islands. Within Asian clusters, a new lineage was emerged and was introduced to Western Hemisphere where it has circulated in Brazil and other South America presently. On the other hand, it was disclosed by virological analysis on isolated ZIKV entered to Brazil in 2013 even though ZIKV generally is supposedly to enter to the Brazil World Cup event in 2014 (13).
ZIKV is genetically and antigenically related to Spondweni virus which belongs to Flavivirus genus in Africa (13). Both viruses form a unique clade (clade X) within the mosquito-borne flavivirus cluster. The African lineage is consisted of the East African cluster group including the genetic variants of the prototype, MR766 strain, and second group of West African strains. ZIKV strains from Pacific Island are confirmed to be related to the Asian lineages which are presumed to be introduced to the Pacific island either by infected persons or mosquito. It was also disclosed that American strains are phylogenetically placed in a clade from the Asian lineage with a nearly complete identical sequence with a ZIKV isolate from South Pacific Islands. Furthermore, it was also elucidated that ZIKV strains collected in the same geographical region recently show minimal changes in their sequences (30). Human-to-human transmission of the Asian ZIKV strains has been associated with significant NS1 codon usage adaptation to human housekeeping genes. These adapted genes could facilitate viral replication and increase viral titers, compared with those of other flavivirus, e.g., dengue virus (19).
Furthermore, it was also disclosed that several ZIKV strains exhibited a 4 amino acids deletion corresponding to the glycosylation motifs in envelope protein 154, found in many flaviviruses. These kinds of mutations could make ZIKV different from other flaviviruses in virulence, transmission and algorithms of laboratory diagnosis (1319).
ZIKV is closely related to dengue virus, JEV, and other flaviviruses which could share same geographic distributions with ZIKV in disease prevalence (1327). In addition, ZIKV and dengue virus share similar symptoms and transmission cycles in the same areas (233940414243). On the other hand, it is supposed that ZIKV and JEV could show similar symptoms in Southeast Asia. Therefore, differential tests of ZIKV from other flaviviruses and characterization of ZIKV need to be considered for the diagnosis of suspected patients in these areas (4244). For example, it is known that ZIKV own considerably higher levels of viral loads for longer periods of time than other flaviviruses in various kinds of body fluids, such as serum, semen, and urine (21313234). Although serum is mainly used for the detection for antibody and RNA of ZIKV, importance of urine and other body fluids is in increasing trends for the RNA detection of ZIKV and final conclusion of the laboratory diagnosis. There are several reports that viral loads of ZIKV in urine are higher and last longer than in any other body fluids (34454647484950). Even though ZIKV detection guidelines recommend that urine can be used within up to 14 days, RNA in urine might is reported to be present up to 20 days, depending on patients (34).
In case of negative test results in molecular tests, immunoglobulin (Ig) M and neutralizing antibody testing should be employed for the obtaining final conclusion of the diagnosis. In the areas of ZIKV epidemic areas, cross reactions in serology between flaviviruses can make identification of the specific infecting virus difficult, especially when the tested person was infected with flaviviruses or vaccinated against related flaviviruses. This is important because the results of ZIKV and other flavivirus testing will guide clinical management and epidemiological control. Theoretically, using of monoclonal antibodies to ZIKV specific antigens in serological diagnosis can enhance specificity of tests between flaviviruses. Nonspecific test results could occur more often in the case of point of care medical testing in the field because of possible higher cross-contaminations among specimens (45). On the other hand, revised ZIKV diagnostic guidance of KCDC recommends that all tests related with ZIKV be performed in the laboratory of Biosafety Level 2 or above for the prevention of cross-contamination of specimens and safety of laboratory personnel (46).
As of middle 2016, most countries in the world are known to keep WHO guidance or modified guidance of WHO/CDC/ECDC (464748). Main path of those three guidelines is very similar in algorithms, employing test methods and interpretations of tests. RNA from ZIKV, dengue virus, and chikungunya virus is generally detectable in serum during the acute phase of infection in ZIKV epidemic areas. Particularly, one should notice that RNA from JEV could be detected in the patients from the areas of Southeast Asia.
Basically, KCDC is known to follow the WHO/CDC guidance and they revised the laboratory diagnosis guidance as of 10 November 2016. It describes all the details of contents on ZIKV laboratory diagnosis of suspected patients with arbovirus symptom. The contents of Korean guidance almost include those of WHO, except no description of other flavivirus tests on the patients from the area of multiple flaviviruses epidemics.
In performing ZIKV diagnosis, real-time RT-PCR (rRT-PCR), using serum within 7 days after symptom onset, is the most important test for the definite diagnosis. If the result of rRT-PCR is positive, it can be interpreted as ZIKV infection (51525354). In case of negative result, serum should be tested by various kinds of antibody detection methods. Serum for antibody detects should be collected during the periods of 2~14 weeks after symptom onset. If anti-IgM or equivalent tests show positive results, rRT-PCR and plaque reduction neutralization test (PRNT) should be followed. When those tests show positive results, the results should be interpreted as the virus infected as well. In special cases, rRT-PCR tests on ZIKV are performed on cerebrospinal fluid (CSF) and/or amniotic fluid specimens collected within 7 days after symptom onset.
It should be emphasized that most of persons, with ZIKV infections found in Korea, are from Southeast Asia where dengue virus and JEV are prevalent. Therefore, for the test of those returning persons with ZIKV infection or suspects from Southeast Asia, test for JEV also must be included, in addition to dengue, and/or other flaviviruses. Because JEV is more prevalent flavivirus than chikungunya in these areas, unlike in Central or South America. Therefore, the detailed protocol for ZIKV tests must be adjusted based on the possibility of co-infection with other flaviviruses.
In addition, other factors must be considered when results of IgM serological tests are interpreted. That is exact data on the duration of IgM antibody persistence following infection ZIKV are not disclosed with detail. For comparison, IgM antibodies against West Nile virus belonged to flavivirus have been known to be detected for at least 3 months after viremic blood donations. Neutralizing antibodies to ZIKV develop shortly after rising of IgM antibodies and are expected to persist for years. In addition, neutralizing antibodies are presumed to confer possibly lifelong immunity. In persons previously infected or vaccinated with a flavivirus or JEV can result in a rapid rise in neutralizing antibodies against multiple flaviviruses. When performing serologic testing, the presence of these neutralizing antibodies against multiple flaviviruses can preclude conclusive determination of which flavivirus was responsible for the recent infection. Presently, enzyme-linked immunosorbent assay (ELISA) is used to detect anti-ZIKV IgM antibodies in serum or cerebrospinal fluid.
Moreover, cross-reactions among flaviviruses in anti-ZIKV IgM ELISA tests could occur very commonly. In the Korean ZIKV diagnosis guidance, reduction neutralization test (PRNT) is described to be employed for the measures of specific neutralizing antibody titers and performed against various related flaviviruses to rule out such false-positive ELISA results. PRNT also can be used to identify the infecting virus in primary flavivirus infections. Because importance of appropriate clinical management of ZIKV and the risk for adverse pregnancy outcomes in women infected with ZIKV during pregnancy, other arbovirus infections should be considered as well in order to exclude the possibility of ZIKV infection.
On the other hand, the latest revised guidance of CDC (47) includes overview, specimens, qualification of laboratories, and biological safety with detail. Furthermore, it also includes methods of molecular testing and antibody detection in addition to algorithm and flow-chart of whole pipelines.
In United States, FDA is in a position that all the reagents for ZIKV tests are authorized or cleared reagents for Emergency Use Authorizations (EUA). As of July 2016, three kinds of commercial reagent are used for the test of ZIKV RNA only tests after authorization in the level of EUA (50). Therefore, many other countries also seem to be in similar positions in the use of ZIKV reagents. In Korea, it is known that commercial reagents of EUA level are used for molecular tests and most serological tests including PRNT are performed in ZIKV reference laboratory of KNIH only (48).
Present diagnostic system for final determination of ZIKV infection is very complex and takes long time for final diagnosis because of the possibilities of false diagnosis caused by simultaneous infections of flaviviruses in ZIKV epidemic/endemic areas. Particularly, anti-ZIKV IgM test can cause high level of nonspecific cross-reaction and neutralizing test using PRNT on cell culture can only be performed by advanced reference laboratories. Therefore, these complexities of diagnostic system must be simplified and easy to perform with the advent of more specific and easy diagnostic methods to perform. Most of emerging technologies which are being developed for the viral infection can be employed for the improvement of the present diagnostic methods. Among numerous research reports, several examples directly connected to ZIKV detection will be taken here. First of all, Priyamvada et al. reported the issue of highly cross-reactivity between dengue virus and ZIKV when testing dengue patients against ZIKV (43). Their data on these cross-reactivities between these two viruses required more specific ZIKV serology and more sensitive molecular tests. Developing both methods are presumed to help solving the problems in present ZIKV diagnosis.
On the other hands, simultaneous tests using NS1 antigen and IgM tests are reported to Increase specificity to 92.9%, which can be good example of technology Improvements (4950). In addition, Moulin et al. reported to employ more practical and easy differentiation methods among flaviviruses which he suggested more simplified algorithms (42).
Niemz et al. (40) reviewed the literatures relating to point-of-care nucleic acid testing for infectious diseases. Present nucleic acid testing is mainly PCR-based, and its use is limited in hospital or centralized reference laboratories which approaches could be difficult to access to developing countries worldwide. Point-of-care nucleic acid testing, mainly using isothermal nucleic acid amplification methods is expected to supplement many shortcomings of present methods. For example, it is supposed to shorten the time required for diagnosis using integrated platforms instead of the present 3 main steps, namely sample preparation, amplification and detection. Faye et al. is reported to develop one step PCR methods of ZIKV detection with sensitivity of 7.7 pfu/reaction in human serum and with high level of specificity (33). However, it is concluded that one-step RT-PCR for the detection of ZIKV requires further evaluation for the application of laboratory diagnosis (33).
In the laboratory diagnosis of ZIKV, body fluids other than serum and cerebrospinal fluid (CSF) is expected to supplement serum because of split test results between serum and other body fluids (31). Recently it is reported that saliva and urine often showed higher viral loads than serum specimens for longer periods of time after infection (31). In addition, urine and saliva have advantages over serum in specimen obtaining, handling and treatment for test over serum. Advances in molecular technologies using urine and saliva are supposed to enhance the value of urine and saliva specimens in virology diagnosis.
Future direction of viral tests like ZIKV will be in trends to shorten the time required for testing. For example, Tian et al. (52) reported loop-mediated isothermal amplification (LAMP) and AC susceptometry assay for rapid and highly sensitive quantitative ZIKV. The proposed detection system recognized ZIKV oligonucleotide just in 27 minutes with high specificity and sensitivity. Another example of new development is that Xu MY et al. (53) developed a SYBR Green based one-step real-time RT-PCR assay for rapid detection of ZIKV. In their study, ZIKV replication at different time points in infected cells could be rapidly monitored by the real-time RT-PCR assay. In this research it turned out that the developed real-time RT-PCR method showed acceptable level of performance in the RNA measurement of infectious virus. The sensitivity of the newly developed method was a titer of as low as 1 PFU/ml which is much higher than that of other reports. They insisted that the method would be useful tool for further virology surveillance and diagnosis of ZIKV.
In addition, future Zika viral diagnosis will cost less than the present methods using programmable biomolecule components by technological advances (54).
ZIKV has taken public attention recently, when the virus became pandemic in Central and South America including Brazil nearly 70 years after the first confirmation of its presence. Nowadays the epidemic influences nearly all the countries throughout the world, regardless of autochthonous infections because of possibilities of patients' outbreaks by intensive international travelling.
As of middle 2016, the areas with highest epidemics are surveyed to be distributed through many countries in Central and South America and Southeast Asia with different patterns in clinical symptoms. Health educations and public awareness on ZIKV are required for the people who travel or reside in countries with autochthonous areas. Depending on ZIKV epidemics, more persons with ZIKV infection can be expected to return to their own countries after infection. However, presently there is very low possibility of autochthonous epidemic of ZIKV in Korea even though 14 persons found with ZIKV overseas were confirmed infection. The reasons are that prevalence of Aedes species mosquito in Korea is lower than 3% in Korea and autochthonous cycle is not expected to form.
Future direction of ZIKV epidemics worldwide is not certain presently. Even though ZIKV belongs to Flavivirus genus like JEV, which is known to be only arbovirus in Korea, ZIKV shows differences both in viral loads and in some other viral properties from those of JEV. Detailed differences of ZIKV from JEV are recommended to be elucidated for the exact differential diagnosis of ZIKV and lead to valuable vaccine development. Algorithms and flow-chart of testing procedures for returning patients from Southeast Asia recommend to be differentiated from the patients from Americas where JEV is not prevalent. Furthermore, the presence of longer ZIKV viremia in the body fluids after infection than other flaviviruses must be kept in mind when employing molecular tests. In addition, the possibilities of nonspecific IgM results and cross-reactions among flaviviruses must be excluded in order to enhance the accuracy of the serological diagnosis.
The present guidances of WHO and CDC are mainly established for the diagnosis of patients in both Central and South America where chickungunya and/or dengue virus are mainly prevalent, unlike in Southeast Asia. Therefore, algorithm of ZIKV tests for patients from Southeast Asias should be differentiated from those of patients from America. Recent technological trends of ZIKV laboratory diagnosis are in forwards to the developments of easy diagnostic technologies with high accuracy using less time and cost.
Figures and Tables
Figure 1
Origins and transmission route of ZIKV transmission with years. The virus spread to Americas through South Asia and Pacific Islands. On the other hand, it spread to Eastern Africa afterwards without significant symptoms.
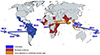
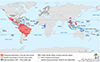
Figure 2
Epidemics of ZIKV worldwide by the levels of prevalence groups of ZIKV infections. As shown in the map, many countries in Central and South Americas and several countries in South Eastern Asia show relatively high numbers of autochthonous persons with ZIKV (from European Center for Disease Control, https://www.emma.ecdc.europa.eu).
References
1. Dick GW, Kitchen SF, Haddow AJ. Zika virus. I. Isolations and serological specificity. Trans R Soc Trop Med Hyg. 1952; 46:509–520.

2. Wikan N, Smith DR. Zika virus: history of a newly emerging arbovirus. Lancet Infect Dis. 2016; 16:e119–e126.

3. Lanciotti RS, Kosoy OL, Laven JJ, Velez JO, Lambert AJ, Johnson AJ, et al. Genetic and serologic properties of Zika virus associated with an epidemic, Yap State, Micronesia, 2007. Emerg Infect Dis. 2008; 14:1232–1239.

4. Chang C, Ortiz K, Ansari A, Gershwin ME. The Zika outbreak of the 21st century. J Autoimmun. 2016; 68:1–13.

5. Oduyebo T, Igbinosa I, Petersen EE, Polen KN, Pillai SK, Ailes EC, et al. Update: Interim Guidance for Health Care Providers Caring for Pregnant Women with Possible Zika Virus Exposure - United States, July 2016. MMWR Morb Mortal Wkly Rep. 2016; 65:739–744.

6. Epidemiological Update: Neurological syndrome, congenital anomalies and Zika virus infection. Washington, D.C.: PAHO/WHO;2016.
7. Jouannic JM, Friszer S, Leparc-Goffart I, Garel C, Eyrolle-Guignot D. Zika virus infection in French Polynesia. Lancet. 2016; 387:1051–1052.

9. Heymann DL, Hodgson A, Sall AA, Freedman DO, Staples JE, Althabe F, et al. Zika virus and microcephaly: why is this situation a PHEIC? Lancet. 2016; 387:719–721.

10. Dichtelmüller HO, Biesert L, Fabbrizzi F, Gajardo R, Gröner A, von Hoegen I, et al. Robustness of solvent/detergent treatment of plasma derivatives: a data collection from Plasma Protein Therapeutics Association member companies. Transfusion. 2009; 49:1931–1943.

11. Macnamara FN. Zika virus: a report on three cases of human infection during an epidemic of jaundice in Nigeria. Trans R Soc Trop Med Hyg. 1954; 48:139–145.

12. Marchette NJ, Garcia R, Rudnick A. Isolation of Zika virus from Aedes aegypti mosquitoes in Malaysia. Am J Trop Med Hyg. 1969; 18:411–415.
13. Weaver SC, Costa F, Garcia-Blanco MA, Ko AI, Ribeiro GS, Saade G, et al. Zika virus: History, emergence, biology, and prospects for control. Antiviral Res. 2016; 130:69–80.

14. Gardner LM, Chen N, Sarkar S. Global risk of Zika virus depends critically on vector status of Aedes albopictus. Lancet Infect Dis. 2016; 16:522–523.

15. Rapid risk assessment: Zika virus epidemic in the Americas: potential association with microcephaly and Guillain-Barré syndrome. Stockholm: ECDC;2015.
16. Baud D, Van Mieghem T, Musso D, Truttmann AC, Panchaud A, Vouga M. Clinical management of pregnant women exposed to Zika virus. Lancet Infect Dis. 2016; 16:523.

17. Kim K, Shresta S. Neuroteratogenic Viruses and Lessons for Zika Virus Models. Trends Microbiol. 2016; 24:622–636.

18. Park Y, Noh Y, Ryu B, Shin SH, Ra S. Preparedness plan and response to Zika virus infection in the Republic of Korea. Public Health Wkly Rep. 2016; 9:426–429.
19. Ramos da Silva S, Gao SJ. Zika virus: An update on epidemiology, pathology, molecular biology, and animal model. J Med Virol. 2016; 88:1291–1296.

20. Dupont-Rouzeyrol M, Biron A, O'Connor O, Huguon E, Descloux E. Infectious Zika viral particles in breastmilk. Lancet. 2016; 387:1051.

21. Mansuy JM, Dutertre M, Mengelle C, Fourcade C, Marchou B, Delobel P, et al. Zika virus: high infectious viral load in semen, a new sexually transmitted pathogen? Lancet Infect Dis. 2016; 16:405.

22. Kaddumukasa MA, Mutebi JP, Lutwama JJ, Masembe C, Akol AM. Mosquitoes of Zika Forest, Uganda: species composition and relative abundance. J Med Entomol. 2014; 51:104–113.

23. Terzian AC, Mondini A, Bronzoni RV, Drumond BP, Ferro BP, Cabrera EM, et al. Detection of Saint Louis encephalitis virus in dengue-suspected cases during a dengue 3 outbreak. Vector Borne Zoonotic Dis. 2011; 11:291–300.

24. Ayres CF. Identification of Zika virus vectors and implications for control. Lancet Infect Dis. 2016; 16:278–279.

26. World Health Organization on Regulations for Transport of Infectious Substances. 2015-2016. Geneva: WHO;2015.
27. Gubler D, Kuno G, Markoff L. Flaviviruses. In : Fields BN, Knipe DM, Howley PM, editors. Fields virology. 5th edition. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins;2007.
28. Sirohi D, Chen Z, Sun L, Klose T, Pierson TC, Rossmann MG, et al. The 3.8 A resolution cryo-EM structure of Zika virus. Science. 2016; 352:467–470.
29. Gadea G, Bos S, Krejbich-Trotot P, Clain E, Viranaicken W, El-Kalamouni C, et al. A robust method for the rapid generation of recombinant Zika virus expressing the GFP reporter gene. Virology. 2016; 497:157–162.

30. Giovanetti M, Faria NR, Nunes MR, de Vasconcelos JM, Lourenco J, Rodrigues SG, et al. Zika virus complete genome from Salvador, Bahia, Brazil. Infect Genet Evol. 2016; 41:142–145.

31. Musso D, Roche C, Nhan TX, Robin E, Teissier A, Cao-Lormeau VM. Detection of Zika virus in saliva. J Clin Virol. 2015; 68:53–55.

32. Prisant N, Bujan L, Benichou H, Hayot PH, Pavili L, Lurel S, et al. Zika virus in the female genital tract. Lancet Infect Dis. 2016; 16:1000–1001.

33. Faye O, Dupressoir A, Weidmann M, Ndiaye M, Alpha Sall A. One-step RT-PCR for detection of Zika virus. J Clin Virol. 2008; 43:96–101.

34. Campos Rde M, Cirne-Santos C, Meira GL, Santos LL, de Meneses MD, Friedrich J, et al. Prolonged detection of Zika virus RNA in urine samples during the ongoing Zika virus epidemic in Brazil. J Clin Virol. 2016; 77:69–70.

35. Carocci M, Yang PL. Lactimidomycin is a broad-spectrum inhibitor of dengue and other RNA viruses. Antiviral Res. 2016; 128:57–62.

36. Schneider-Poetsch T, Ju J, Eyler DE, Dang Y, Bhat S, Merrick WC, et al. Inhibition of eukaryotic translation elongation by cycloheximide and lactimidomycin. Nat Chem Biol. 2010; 6:209–217.

37. Micoine K, Persich P, Llaveria J, Lam MH, Maderna A, Loganzo F, et al. Total syntheses and biological reassessment of lactimidomycin, isomigrastatin and congener glutarimide antibiotics. Chemistry. 2013; 19:7370–7383.

38. Garreau de Loubresse N, Prokhorova I, Holtkamp W, Rodnina MV, Yusupova G, Yusupov M. Structural basis for the inhibition of the eukaryotic ribosome. Nature. 2014; 513:517–522.

39. Mackay IM, Arden KE, Nitsche A. Real-time PCR in virology. Nucleic Acids Res. 2002; 30:1292–1305.

40. Niemz A, Ferguson TM, Boyle DS. Point-of-care nucleic acid testing for infectious diseases. Trends Biotechnol. 2011; 29:240–250.

41. Fernanda Estofolete C, Terzian AC, Parreira R, Esteves A, Hardman L, Greque GV, et al. Clinical and laboratory profile of Zika virus infection in dengue suspected patients: A case series. J Clin Virol. 2016; 81:25–30.

42. Moulin E, Selby K, Cherpillod P, Kaiser L, Boillat-Blanco N. Simultaneous outbreaks of dengue, chikungunya and Zika virus infections: diagnosis challenge in a returning traveller with nonspecific febrile illness. New Microbes New Infect. 2016; 11:6–7.

43. Priyamvada L, Quicke KM, Hudson WH, Onlamoon N, Sewatanon J, Edupuganti S, et al. Human antibody responses after dengue virus infection are highly cross-reactive to Zika virus. Proc Natl Acad Sci U S A. 2016; 113:7852–7857.

44. Calvo EP, Sánchez-Quete F, Durán S, Sandoval I, Castellanos JE. Easy and inexpensive molecular detection of dengue, chikungunya and zika viruses in febrile patients. Acta Trop. 2016; 163:32–37.

45. Ehrmeyer SS, Laessig RH. Point-of-care testing, medical error, and patient safety: a 2007 assessment. Clin Chem Lab Med. 2007; 45:766–773.

46. World Health Organization. Laboratory biosafety manual. 3rd edition. Geneva: WHO;2004.
47. Chambers TJ, Monath TP. The Flaviviruses: Detection, Diagnosis and Vaccine Development. 1st edition. Academic Press;2003.
48. World Health Organization. Laboratory testing for Zika virus infection (Interim guidance). Geneva: WHO;2016.
49. World Health Organization. Consultation on the documentary evidence and independent performance evaluation requirement for the emergency use assessment and listing procedure for Zika virus in vitro diagnostics. Geneva: WHO;2016.
50. CDC. Guidance for U.S. Laboratories Testing for Zika Virus Infection. 2016.
51. Rabe IB, Staples JE, Villanueva J, Hummel KB, Johnson JA, Rose L, et al. Interim Guidance for Interpretation of Zika Virus Antibody Test Results. MMWR Morb Mortal Wkly Rep. 2016; 65:543–546.

52. Tian B, Qiu Z, Ma J, Zardán Gómez de la Torre T, Johansson C, Svedlindh P, et al. Attomolar Zika virus oligonucleotide detection based on loop-mediated isothermal amplification and AC susceptometry. Biosens Bioelectron. 2016; 86:420–425.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download