Abstract
According to World Health Organization, more than 200 million people suffer with chronic hepatitis caused by Hepatitis B virus (HBV) infection worldwide. Chronic hepatitis B causes various complications including liver cirrhosis and hepatocellular carcinoma and approximately 0.5~4.2 million deaths occur annually due to HBV infection. Current therapies such as antivirals and vaccine are often hampered by drug intolerance, side effects, and long-time medication, therefore, the development of powerful anti-HBV drugs is demanded. Recently, sodium taurocholate co-transporting polypeptide (NTCP) receptor was revealed to play a pivotal role in HBV entry into hepatocytes. Cell lines transfected with NTCP receptor enables to analyze HBV life cycle by inducing HBV infection stably, but in vivo models still have some limitations such as high costs, restrictive differentiation, and unveiled cofactors related to human NTCP. Therefore, it requires well-established in vivo models to develop and evaluate novel therapeutic agents targeting NTCP receptor, and viral entry inhibitors that inhibit the early step of viral infection are potent sufficient to substitute for existing antivirals.
Go to : 
REFERENCES
1). Global Burden of Disease Study 2010 outlines present and future health priorities, both nationally and internationally. Br J Hosp Med (Lond). 2013; 74:71.
2). Glebe D, Urban S, Knoop EV, Cag N, Krass P, Grun S, et al. Mapping of the hepatitis B virus attachment site by use of infection-inhibiting preS1 lipopeptides and tupaia hepatocytes. Gastroenterol. 2005; 129:234–45.

3). Watashi K, Urban S, Li W, Wakita T. NTCP and beyond: opening the door to unveil hepatitis B virus entry. Int J Mol Sci. 2014; 15:2892–905.

5). Iwamoto M, Watashi K, Tsukuda S, Aly HH, Fukasawa M, Fujimoto A, et al. Evaluation and identification of hepatitis B virus entry inhibitors using HepG2 cells overexpressing a membrane transporter NTCP. Biochem Biophys Res Commun. 2014; 443:808–13.

6). Wieland S, Thimme R, Purcell RH, Chisari FV. Genomic analysis of the host response to hepatitis B virus infection. Proc Natl Acad Sci U S A. 2004; 101:6669–74.

7). Urban S, Bartenschlager R, Kubitz R, Zoulim F. Strategies to inhibit entry of HBV and HDV into hepatocytes. Gastroenterol. 2014; 147:48–64.

8). Zoulim F, Locarnini S. Optimal management of chronic hepatitis B patients with treatment failure and antiviral drug resistance. Liver Int. 2013; 33(Suppl 1):116–24.

9). Zoulim F, Perrillo R. Hepatitis B: reflections on the current approach to antiviral therapy. J Hepatol. 2008; 48(Suppl 1):S2–19.

10). Neurath AR, Kent SB, Strick N, Parker K. Identification and chemical synthesis of a host cell receptor binding site on hepatitis B virus. Cell. 1986; 46:429–36.

11). Ni Y, Lempp FA, Mehrle S, Nkongolo S, Kaufman C, Falth M, et al. Hepatitis B and D viruses exploit sodium taurocholate co-transporting polypeptide for species-specific entry into hepatocytes. Gastroenterology. 2014; 146:1070–83.

13). Yan H, Peng B, He W, Zhong G, Qi Y, Ren B, et al. Molecular determinants of hepatitis B and D virus entry restriction in mouse sodium taurocholate cotransporting polypeptide. J Virol. 2013; 87:7977–91.

14). Hagenbuch B, Meier PJ. Molecular cloning, chromosomal localization, and functional characterization of a human liver Na+/bile acid cotransporter. J Clin Invest. 1994; 93:1326–31.

15). Meier PJ, Stieger B. Bile salt transporters. Annu Rev Physiol. 2002; 64:635–61.
16). Anwer MS, Stieger B. Sodium-dependent bile salt transporters of the SLC10A transporter family: more than solute transporters. Pflugers Arch. 2014; 466:77–89.

17). Yan H, Peng B, Liu Y, Xu G, He W, Ren B, et al. Viral entry of hepatitis B and D viruses and bile salts transportation share common molecular determinants on sodium taurocholate cotransporting polypeptide. J Virol. 2014; 88:3273–84.

18). Glebe D, Urban S. Viral and cellular determinants involved in hepadnaviral entry. World J Gastroenterol. 2007; 13:22–38.

19). Yan H, Zhong G, Xu G, He W, Jing Z, Gao Z, et al. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus. Elife. 2012; 1:e00049.

20). Gripon P, Diot C, Corlu A, Guguen-Guillouzo C. Regulation by dimethylsulfoxide, insulin, and corticosteroids of hepatitis B virus replication in a transfected human hepatoma cell line. J Med Virol. 1989; 28:193–9.

21). Verrier ER, Colpitts CC, Bach C, Heydmann L, Weiss A, Renaud M, et al. A targeted functional RNA interference screen uncovers glypican 5 as an entry factor for hepatitis B and D viruses. Hepatol. 2016; 63:35–48.

22). Huang LR, Wu HL, Chen PJ, Chen DS. An immunocompetent mouse model for the tolerance of human chronic hepatitis B virus infection. Proc Natl Acad Sci U S A. 2006; 103:17862–7.

23). Yang PL, Althage A, Chung J, Chisari FV. Hydrodynamic injection of viral DNA: a mouse model of acute hepatitis B virus infection. Proc Natl Acad Sci U S A. 2002; 99:13825–30.

24). Tateno C, Yoshizane Y, Saito N, Kataoka M, Utoh R, Yamasaki C, et al. Near completely humanized liver in mice shows human-type metabolic responses to drugs. Am J Pathol. 2004; 165:901–12.

25). Volz T, Allweiss L, Ben MM, Warlich M, Lohse AW, Pollok JM, et al. The entry inhibitor Myrcludex-B efficiently blocks intrahepatic virus spreading in humanized mice previously infected with hepatitis B virus. J Hepatol. 2013; 58:861–7.

26). Raney AK, Kline EF, Tang H, McLachlan A. Transcription and replication of a natural hepatitis B virus nucleocapsid promoter variant is regulated in vivo by peroxisome proliferators. Virol. 2001; 289:239–51.
27). Li N, Zhang P, Yang C, Zhu Q, Li Z, Li F, et al. Association of genetic variation of sodium taurocholate cotransporting polypeptide with chronic hepatitis B virus infection. Genet Test Mol Biomarkers. 2014; 18:425–9.

28). Warner N, Locarnini S. The new front-line in hepatitis B/D research: identification and blocking of a functional receptor. Hepatol. 2013; 58:9–12.

Go to : 
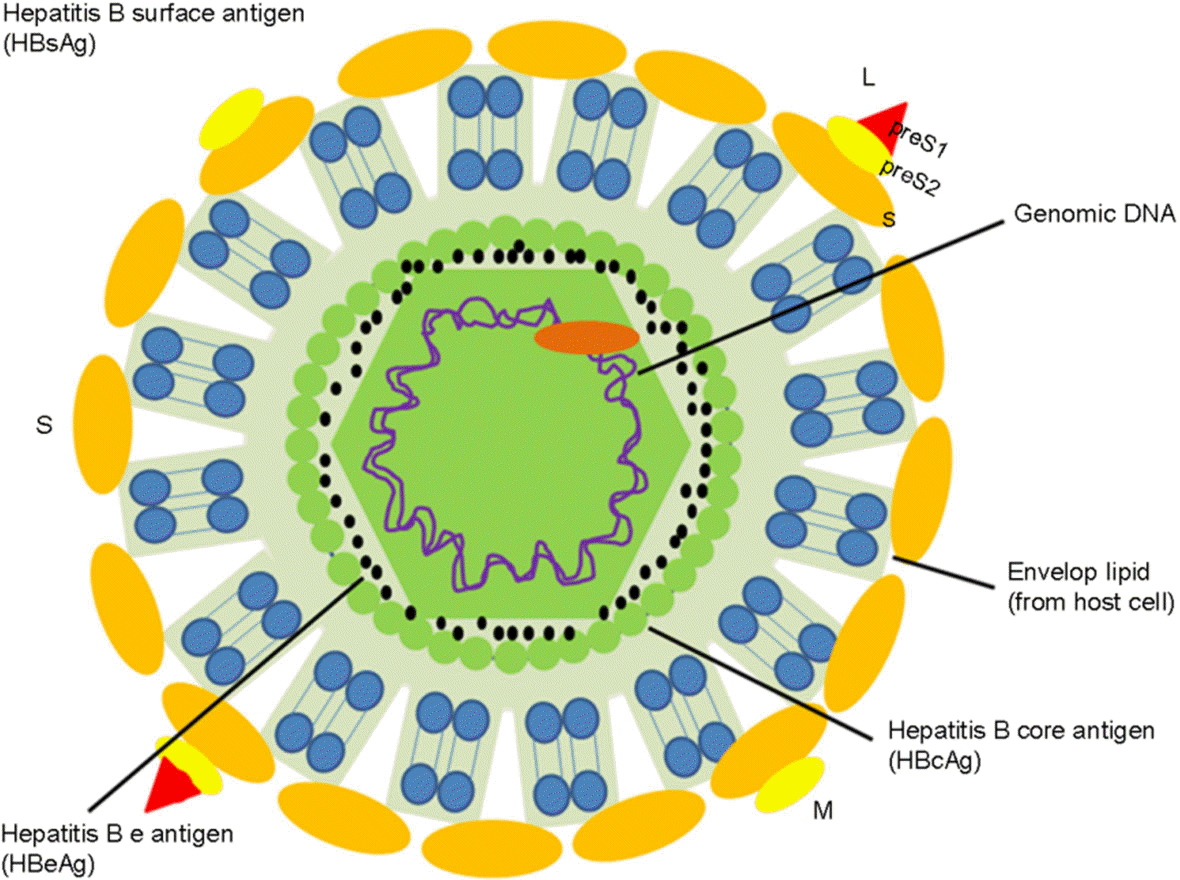 | Figure 1.Structure of Hepatitis B virus. HBV consists of an outer envelope and inner nucleocapsid. The envelop contains HBsAg which com-prises three proteins; the small (S), middle (S+ preS2), large (S+preS2+preS1) surface protein. Among them, only the preS1 domain has an infectious capacity. The capsid encloses genomic DNA, DNA polymerase, HBeAg, and HBcAg. |
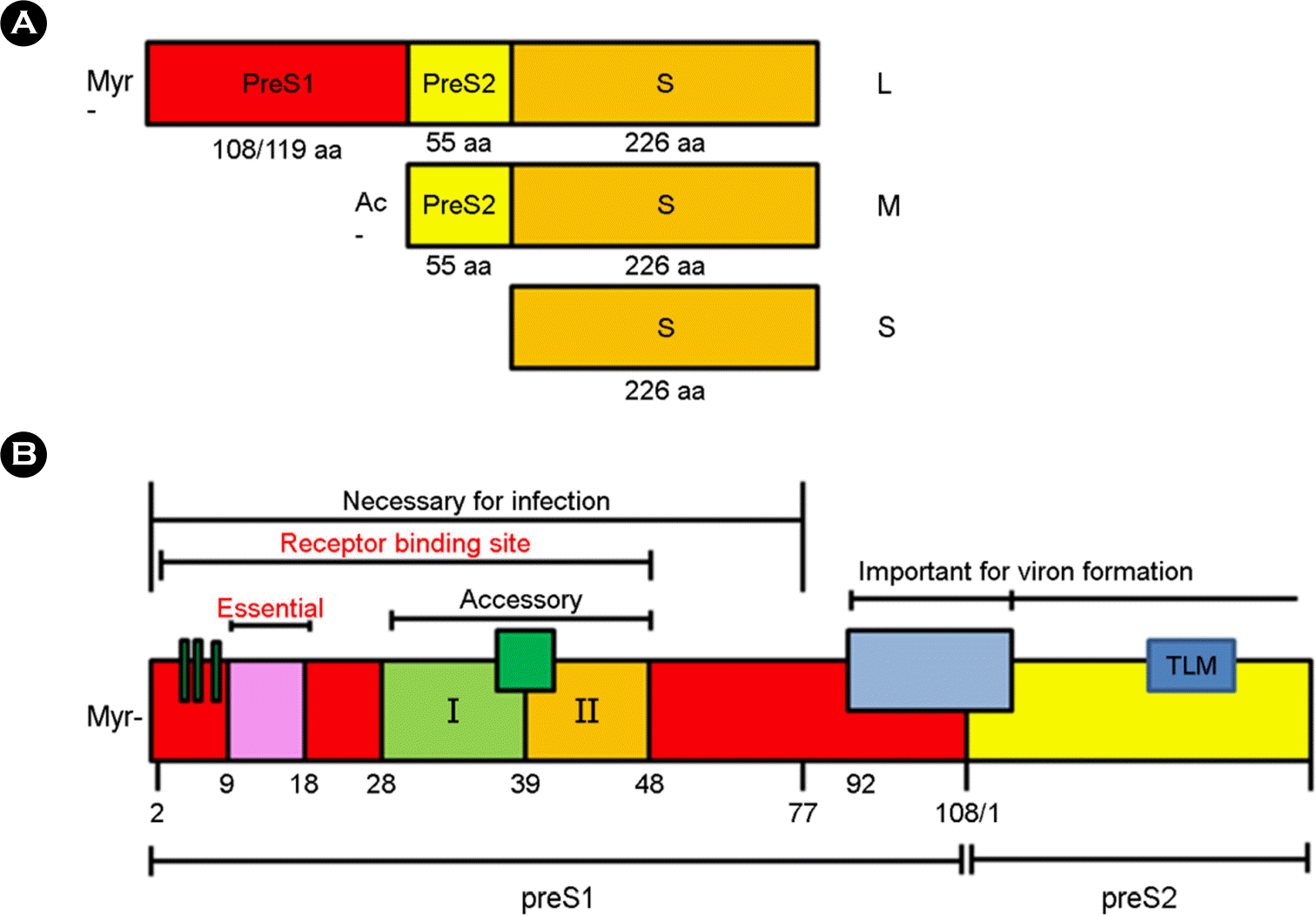 | Figure 2.Hepatitis B virus S protein structure. (A) HBsAg consists of three surface proteins. The small S antigen has only S domain and the middle S antigen harbors preS2 and S domains. The large S antigen contains preS1, preS2, and S domains. (B) Schematic structure of preS1-S2 domains. preS1 domain amino acids 2–77 are necessary for viral infection. Among amino acid 2–48, amino acid 9–18 are essential binding sites of HBsAg for NTCP receptor of human hepatocyte, and amino acid 28–48 are accessory parts. Both preS1 and preS2 domains are important for virion formation. |
 | Figure 3.NTCP amino acid sequence homology between human and other species. (A) Identities and Homologies come from protein sequence Homo sapiens against Crab eating monkey, Rattus norvegicus and Mus musculus. (B) Schematic drawing of HBs domain of Homo sapiens, macaca fascicularis and mus musculus. Amino acid full sequence of Homo sapiens is in discord with that of macaca fascicularis and mus musculus. Amino acid 157–165 of macaca facicularis and amino acid 84–87 of mus musculus are different from Homo sapiens amino acids. |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download