Abstract
Three QIAG93 strains, QIAG9301, QIAG9302 and QIAG9303 that have been identified as Getah virus (GETV) are analyzed in this study. The morphological features of three virus isolates were observed by using electron microscopy, suggesting that the QIAG9301, QIAG9302 and QIAG9303 isolate can be classified as tentative member of Alphavirus species in the Semliki Forest complex. The full length of the structural polyprotein gene of each QIAG93 isolate (QIAG9301, QIAG9302 and QIAG9303) was determined that are identical in size, comprising 3759 nucleotides that encoded 1253 amino acids. The sequence analysis of the structural polyprotein gene, including the C, E3, E1, 6K and E2 domain, showed that each QIAG93 isolate shares >98.9% sequence identity. The phylogenetic analysis and evolutionary distance (ED) estimation based on the structural polyprotein gene sequence showed that the QIAG9301 isolate is closely related to GETV South Korea strain (99.9% sequence identity and ED value 0.001) and Chinese GETV YN0540 strain (99.3% sequence identity ED value 0.007) than other Alphavirus species analyzed in this study. Both QIAG9032 and QIAG9303 isolate exhibited genetically close relationship with Mongolian GETV LEIV17741MPR strain (at least 99.3% sequence identity and mean ED value 0.0065). Therefore, our findings will be valuable for molecular epidemiological analyses of GETV in Korea and contribute to a further study on pathogenicity of three QIAG93 isolates in animals.
Go to : 
REFERENCES
1). Berge TO. International catalogue of arboviruses, including certain other viruses of vertebrates. Washington D. C.: US Department of Health, Education and Welfare, Public Health Service;1975. p. 278.
2). Go YY, Balasuriya UB, Lee CK. Zoonotic encephalitides caused by arboviruses: transmission and epidemiology of alphaviruses and flaviviruses. Clin Exp Vaccine Res. 2014; 3:58–77.

3). Elisvberg BL, Buescher EL. Getah (MM 2021). Arbovirus catalogue card No.47, Arbovirus Information Exchange. 1963.
4). Doherty RL, Gorman BM, Whitehead RH, Carley JG. Studies of arthropod-borne virus infections in Queensland. V. Survey of antibodies to Group A arboviruses in man and other animals. Aust J Expt Biol Sci. 1966; 44:365–77.
5). Brown CM, Timoney PJ. Getah virus infection of Indian horses. Trop Anim Health Prod. 1988; 30:241–52.
6). Kamada M, Ando Y, Fukunaga Y, Kumanomido T, Imagawa H, Wada R, et al. Equine Getah virus infection: Isolation of the virus from racehorses during a enzootic in Japan. Am J Trop Med Hyg. 1980; 29:984–8.
7). Sentsui H, Kono Y. An epidemic of Getah virus infection among racehorses: isolation of the virus. Res Vet Sci. 1980; 29:157–61.

8). Timoney PJ. Getah virus infection. Infectious Diseases of Livestock. Coetzer JAW and Tustin RC, editor. 2nd ed.Cape Town: Oxford University Press;2004. p. 1023–26.
9). Kumanomido T, Wada R, Kanemaru T, Kamada M, Hirasawa K, Akiyama Y. Clinical and virological observations on swine experimentally infected with Getah virus. Vet Microbiol. 1988; 16:295–301.

10). Matsuyama T, Nakamura T, Isahai K, Oya A, Kobayashi M. Haruna virus, a group A arbovirus isolated from swine in Japan. Gunma J Med Sci. 1967; 16:131–4.

11). Marchette NJ, Rudnick A, Garcia R. Alphaviruses in Peninsular Malaysia: II. Serological evidence of human infection. Southeast Asian J Trop Med Public Health. 1980; 11:14–23.
12). Calisher CH, Karabatsos N. Arbovirus serogroups: definition and geographic distribution. The Arboviruses: Epidemiology and Ecology. Monath TP, editor. I. Boca Raton, FL: CRC Press;1988. p. 19–57.

13). Weaver SC, Dalgarno L, Frey TK, Huang HV, Kinney RM, Rice CM, et al. Family Togaviridae. Virus Taxonomy: Seventh Report of the International Committee on Taxonomy of Viruses. van Regenmortel MV, Fauguet CM, Bishop DHI, Carstens EB, Estes MK, Lemon SM, editors. San Diego: Academic Press;2000. p. 879–89.
14). Calisher CH, Shope RE, Brandt W, Casals J, Karabatsos N, Murphy FA, et al. Proposed antigenic classification of registered arboviruses I. Togaviridae, Alphavirus. Intervirology. 1980; 14:229–32.
15). Powers AM, Brault AC, Shirako Y, Strauss EG, Kang W, Strauss JH, et al. Evolutionary relationships and systematics of the alphaviruses. J Virol. 2001; 75:10118–31.

16). Bryant JE, Crabtree MB, Nam VS, Yen NT, Duc HM, Miller BR. Isolation of arboviruses from mosquitoes collected in northern Vietnam. Am J Trop Med Hyg. 2005; 73:470–3.

17). Chang CY, Huang CC, Huang TS, Deng MC, Jong MH, Wang FI. Isolation and characterization of a Sagiyama virus from domestic pigs. J Vet Diagn Invest. 2006; 18:156–61.

18). Kanamitsu M, Taniguchi K, Urasawa S, Ogata T, Wada Y, Wada Y, et al. Geographic distribution of arbovirus antibodies in indigenous human populations in the Indo-Australian archipelago. Am J Trop Med Hyg. 1979; 28:351–63.
19). Norder H, Lundström JO, Kozuch O, Magnius LO. Genetic relatedness of Sindbis virus strains from Europe, Middle East, and Africa. Virology. 1996; 222:440–5.

20). Turell MJ, O'Guinn ML, Wasieloski LP Jr, Dohm DJ, Lee WJ, Cho HW, et al. Isolation of Japanese encephalitis and Getah viruses from mosquitoes (Diptera: Culicidae) collected near Camp Greaves, Gyonggi Province, Republic of Korea, 2000. J Med Entomol. 2003; 40:580–4.
21). Tajima S, Kotaki A, Yagasaki K, Taniwaki T, Moi ML, Nakayama E, et al. Identification and amplification of Japanese encephalitis virus and Getah virus propagated from a single porcine serum sample: a case of coinfection. Arch Virol. 2014; 159:2969–75.

22). Pfeffer M, Kinney RM, Kaaden OR. The Alphavirus 3′-nontranslated region: size heterogeneity and arrangement of repeated sequence elements. Virology. 1998; 240:100–8.
23). Griffin DE. Alphaviruses. Knipe DM, Howley PM, editors. Fields' Virology. 4th ed.New York: Lippincott, Williams and Wilkins;2001. p. 917–62.
24). Calisher CH, Walton TE. Getah virus infections. Virus Infections of Equines. Studdert MK, editor. Amsterdam: Elsevier;1996. p. 157–65.
25). Fukunaga Y, Kumanomido T, Kamada M. Getah virus as an equine pathogen. Vet Clin North Am Equine Pract. 2000; 16:605–17.

26). Jo HY, Yang DK, Kim HH, Choi SS, Kang KS, Yang SJ, et al. Sero-surveillance of Getah virus among Thoroughbred horses in Korea. J Bacteriol Virol. 2015; 45:235–41.

27). Colombet J, Sime-Ngando T. Use of PEG, Polyethylene glycol, to characterize the diversity of environmental viruses. Microb Ecol. 2012; 58:728–36.
28). Vajda BP. Concentration and purification of viruses and bacteriophages with polyethylene glycol. Folia Microbiol (Praha). 1978; 23:88–96.

29). Jeanmougin F, Thompson JD, Gouy M, Higgins DG, Gibson TJ. Multiple sequence alignment with Clustal X. Trends Biochem Sci. 1998; 23:403–5.

30). Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG. The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997; 25:4876–82.

31). Hyypiä T, Hovi T, Knowles NJ, Stanway G. Classifiproperties. J Gen Virol. 1997; 78:1–11.
32). Acheson NH, Tamm I. Replication of Semliki Forest virus: an electron microscopic study. Virology. 1967; 32:128–43.

33). Grimley PM, Berezesky IK, Friedman RM. Cytoplasmic structures associated with an arbovirus infection: loci of viral ribonucleic acid synthesis. J Virol. 1968; 2:1326–38.

35). Dietzgen RG, Kuzmin IV. Taxonomy of rhabdoviruses. Rhabdoviruses: Molecular Taxonomy, Evolution, Genomics, Ecology, Host-Vector Interactions, Cytopathology and Control. Dietzgen RG, Kuzmin IV, editors. Norfolk: Caister Academic Press;2012. p. 13–22.
Go to : 
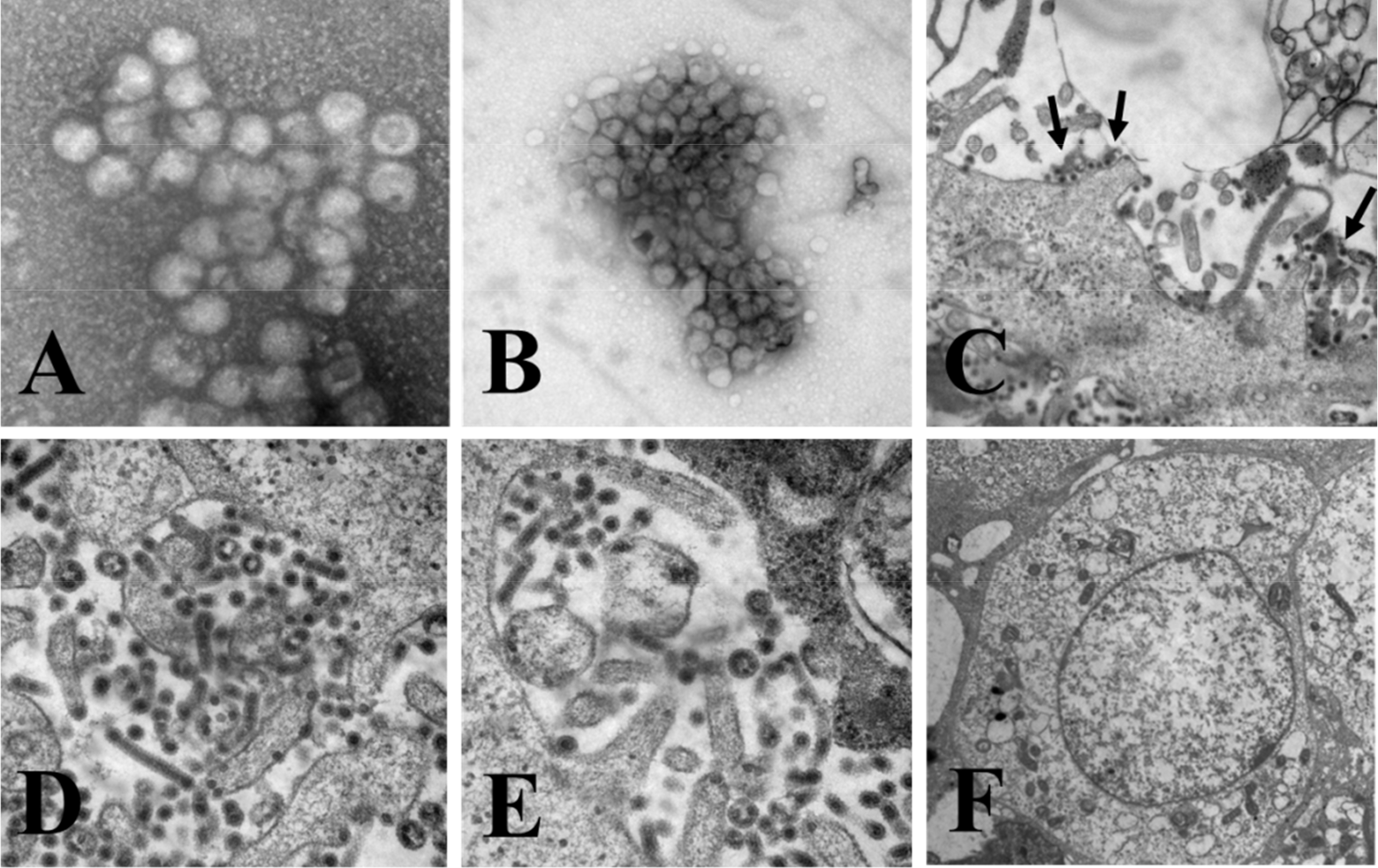 | Figure 1.Electron microscopy of QIAG93 isolates. QIAG9301 was used as a representative of the three isolates. Virus-like particles in specimens prepared by direct (A) or sucrose gradient centrifugation (B). General morphological characteristics of viruses and cytopathic changes in Vero cells after virus infection for 72 h (C, D and E). Mock-infected cells were used for comparison (F). Inset C, black solid arrows indicate virus-like particles. |
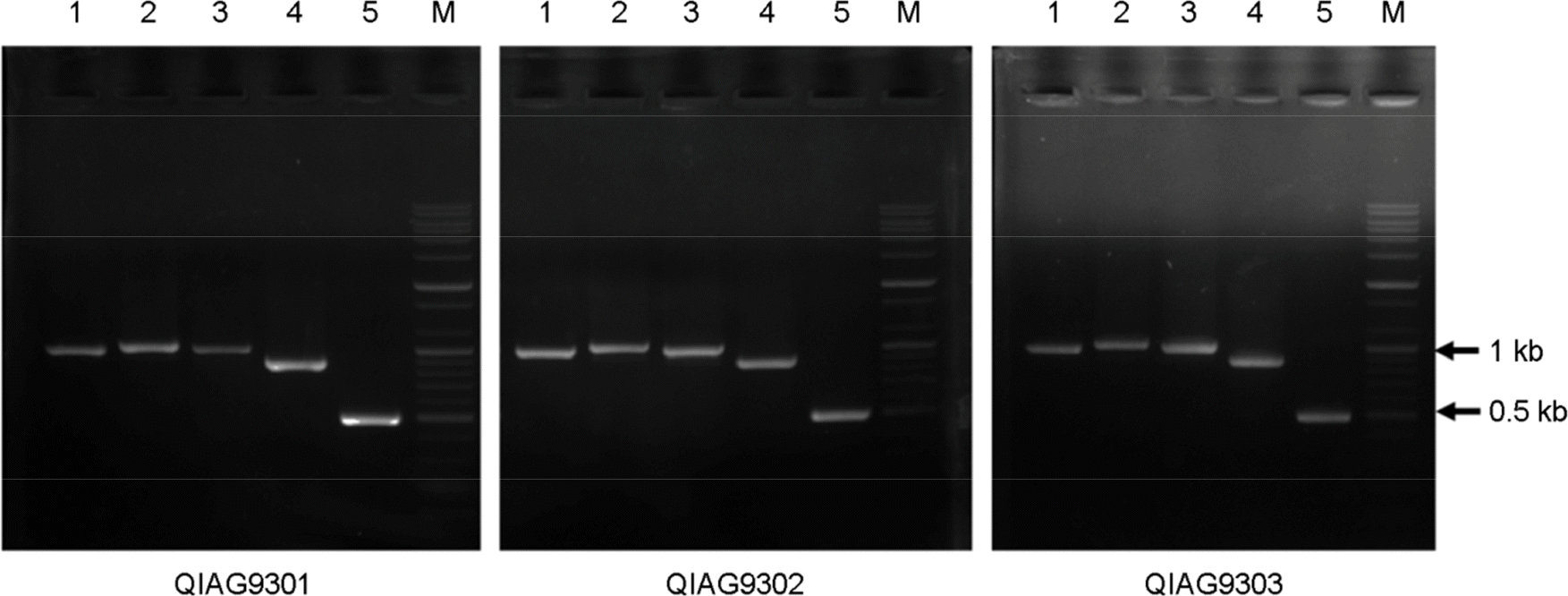 | Figure 2.Amplification of the structural polyprotein gene of the GETV QIAG93 isolate with a specific primer set (see Table 1). Lanes 1 to 5, PCR products amplified using the respective GE1, GE2, GE3, GE4, and GE5 primer sets. Lane M, 100 bp DNA ladder. Use of the GE1 to GE5 primer sets resulted in amplification of products with sizes of 990, 1,030, 990, 880 and 490 bp, respectively. |
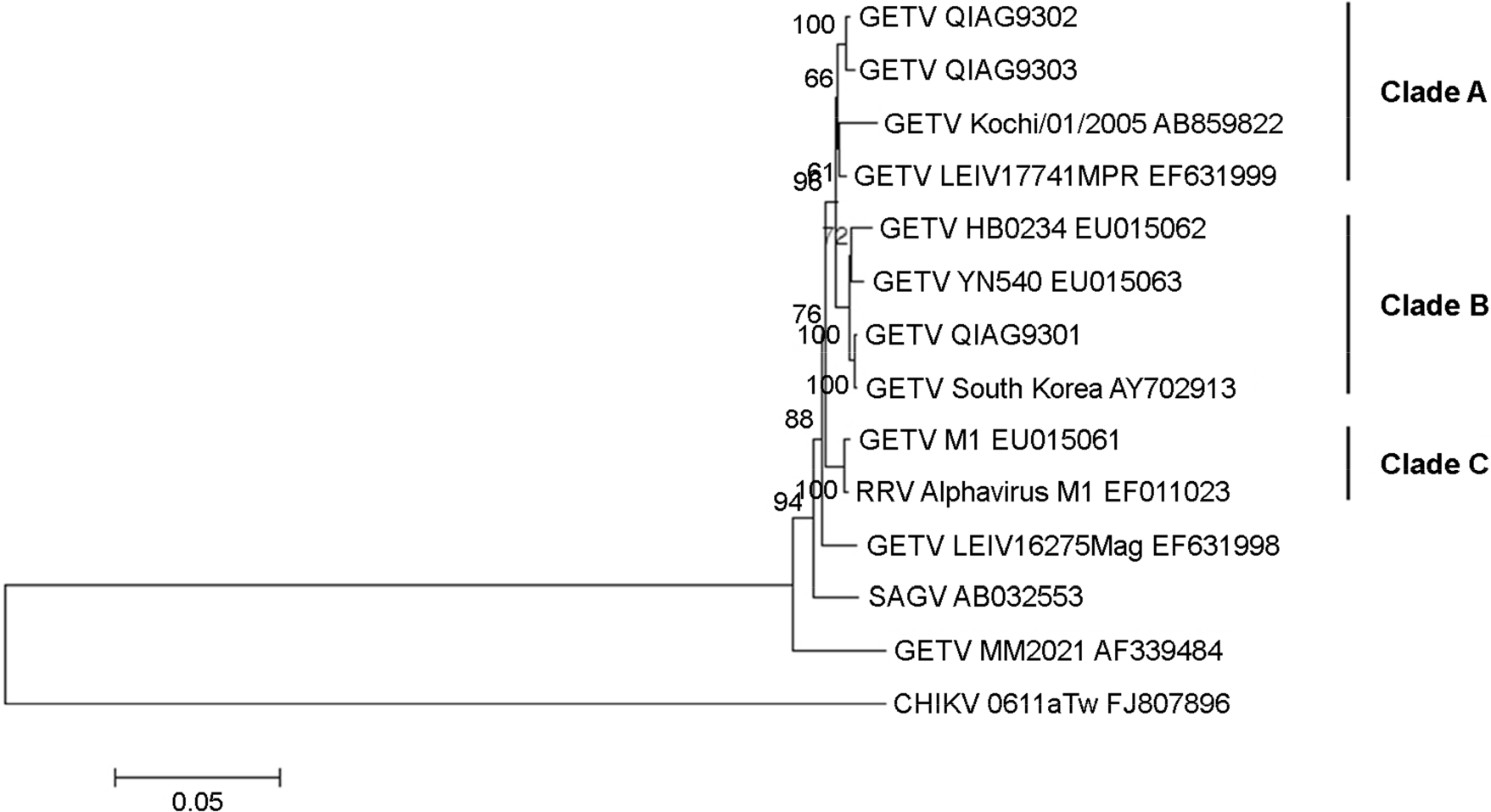 | Figure 3.Phylogenetic relationships between QIAG93 isolates and other Alphaviruses within the Semliki Forest antigenic complex. Phylogenies were assessed on the structural polyprotein gene using neighbor-joining methods. A bootstrap method with 1,000 replicates was applied in this analysis to evaluate the reliability of the inferred phylogenetic tree. In the neighbor-joining tree, the monophyletic sister groups that had bootstrap values > 70% were chosen. The GETV strains were classified into three sister groups rooted at GETV MM2021. |
Table 1.
Oligonucleotide primers used for RT-PCR of GETV, designed from the complete genome sequences of GETV South Korea (GenBank Accession No. AY702913)
Table 2.
Detailed information of the Alphaviruses analyzed in the present study
Table 3.
Nucleotide sequence comparison (above the diagonal) and estimation of evolutionary distance (below the diagonal) based on comparisons of the structural polyprotein gene of QIAG93 isolates with that of other Alphaviruses belonging to the Semliki Forest complex




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download