Abstract
All of the methicillin-resistant Staphylococcus aureus (MRSA) strains exhibit resistance to oxacillin by producing PBP2a encoded by mecA, whereas methicllin-susceptible Staphylococcus aureus (MSSA) strains do not. To investigate phenotypic differences other than oxacillin resistance level in responses to oxacillin between MSSA and MRSA, we compared alterations of viability and ultrastructure of MSSA by oxacillin treatment with those of MRSA. When MSSA and MRSA strains were exposed to oxacillin of their respective MICs, and then were assayed for viability and observed by transmission electron microscope, increase in thickness of cell wall was more prominent in MRSA strains than in MSSA strains, while decrease in number of surviving cells was more evident and change in morphology of growing cross wall was greater in MSSA strains than in MRSA strains. It is assumed that these different responses to oxacillin between MSSA and MRSA strains may be due to activation of some PBP2a unbound to oxacillin. In conclusion, MSSA and MRSA showed different functional and morphological responses to oxacillin, although they were treated with oxacillin of concentrations that respectively inhibit their proliferation.
Go to : 
REFERENCES
1). Brunmfitt W., Hamilton-Miller J. Methicillin-resistant Staphylococcus aureus. N Engl J Med. 1989. 320:1188–96.
2). Jevons MP., Rolinson GN., Knox R. Celbenin-resistant staphylococci. Br Med J. 1961. 1:124–6.
5). Hartman BJ., Tomasz A. Low-affinity penicillin-binding protein associated with β-lactam resistance in Staphylococcus aureus. J Bacteriol. 1984. 158:513–6.
6). Fontana R. Penicillin-binding proteins and the intrinsic resistance to β-lactams in gram-positive cocci. J Antimicrob Chemother. 1985. 16:412–6.
7). Reynolds PE., Fuller C. Methicilin-resistant strains of Staphylococcus aureus: presence of identical additional penicillin-binding proteins in all strains examined. FEMS Microbiol Lett. 1986. 33:251–4.
8). Ito T., Hiramatsu K. Acquisition of methicillin resistance and progression of multiantibiotic resistance in methicillin-resistant Staphylococcus aureus. Yonsei Med J. 1998. 39:526–33.
9). Berger-Bächi B., Strässle A., Gustafson JE., Kayser FH. Mapping and characterization of multiple chromosomal factors involved in methicillin-resistance in Staphylococcus aureus. Antimicrob Agents Chemother. 1992. 36:1367–73.
10). Maki H., Yamaguchi T., Murakami K. Cloning and characterization of a gene affecting the methicillin resistance level and the autolysis rate in Staphylococcus aureus. J Bacteriol. 1994. 176:4993–5000.
11). Komatsuzawa H., Sugai M., Ohta K., Fujiwara T., Nakashima S., Suzuki J., Lee CY., Suginaka H. Cloning and characterization of the fmt gene which affects the methicillin resistance level and autolysis in the presence of Triton X-100 in methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 1997. 41:2355–61.
12). Wyke AW. Isolation of five penicillin-binding proteins from Staphylococcus aureus. FEMS Microbiol Lett. 1984. 22:133–8.
13). Georgopapadakou NH., Liu FY. Binding of β-lactam antibiotics to penicillin-binding proteins of Staphylococcus aureus and Streptococcus faecalis: relation to antibacterial activity. Antimicrob Agents Chemother. 1980. 18:834–6.
14). Georgopapadakou NH., Dix BA., Mauriz YR. Possible physiological functions of penicillin-binding proteins in Staphylococcus aureus. Antimicrob Agents Chemother. 1986. 29:333–6.
15). Curtis NAC., Hayes MV., Wyke AW., Ward JB. A mutant of Staphylococcus aureus H lacking penicillin-binding protein 4 and transpeptidase activity in vitro. FEMS Microbiol Lett. 1980. 9:263–6.
16). Giesbrecht P., Kersten T., Maidhof H., Weche J. Staphylococcal cell wall: morphogenesis and fatal variations in the presence of penicillin. Microbiol Mol Biol Rev. 1998. 62:1371–414.

17). Duckworth GJ., Jordens JZ. Adherence and survival properties of an epidemic methicillin-resistant strain of Staphylococcus aureus compared with those of methicillin-sensitive strains. J Med Microbiol. 1990. 32:195–200.
18). Cree RG., Aleljung P., Paulsson M., Witte W., Noble WC., Ljungh A., Wadström T. Cell surface hydrophobicity and adherence to extra-cellular matrix proteins in two collections of methicillin-resistant Staphylococcus aureus. Epidemiol Infect. 1994. 112:307–14.
19). Vaudaux PE., Monzillo V., Francois P., Lew DP., Foster TJ., Berger-Bächi B. Introduction of the mec element (methicillin resistance) into Staphylococcus aureus alters in vitro functional activities of fibrinogen and fibronectin adhesins. Antimicrob Agents Chemother. 1998. 42:564–70.
20). Coia JE., Browning L., Haines L., Birkbeck TH., Platt DJ. Comparison of enterotoxins and haemolysins produced by methicillin-resistant (MRSA) and sensitive (MSSA) Staphylococcus aureus. J Med Microbiol. 1992. 36:164–71.
21). Schmitz FJ., Mackenzie CR., Geisel R., Wagner S., Idel H., Verhoef J., Hadding U., Heinz HP. Enterotoxin and toxic shock syndrome toxin-1 production of methicillin-resistant and methicillin-sensitive Staphylococcus aureus strains. Eur J Epidemiol. 1997. 13:699–708.
22). Robert J., Etienne J., Bertrand X. Methicillin-resistant Staphylococcus aureus producing Panton-Valentine leukocidin in a retrospective case series from 12 French hospital laboratories, 2000-2003. Clin Microbiol Infect. 2005. 11:585–7.
23). Chini V., Petinaki E., Foka A., Paratiras S., Dimitracopoulos G., Spiliopoulou I. Spread of Staphylococcus aureus clinical isolates carrying Panton-Valentine leukocidin genes during a 3-year period in Greece. Clin Microbiol Infect. 2006. 12:29–34.
Go to : 
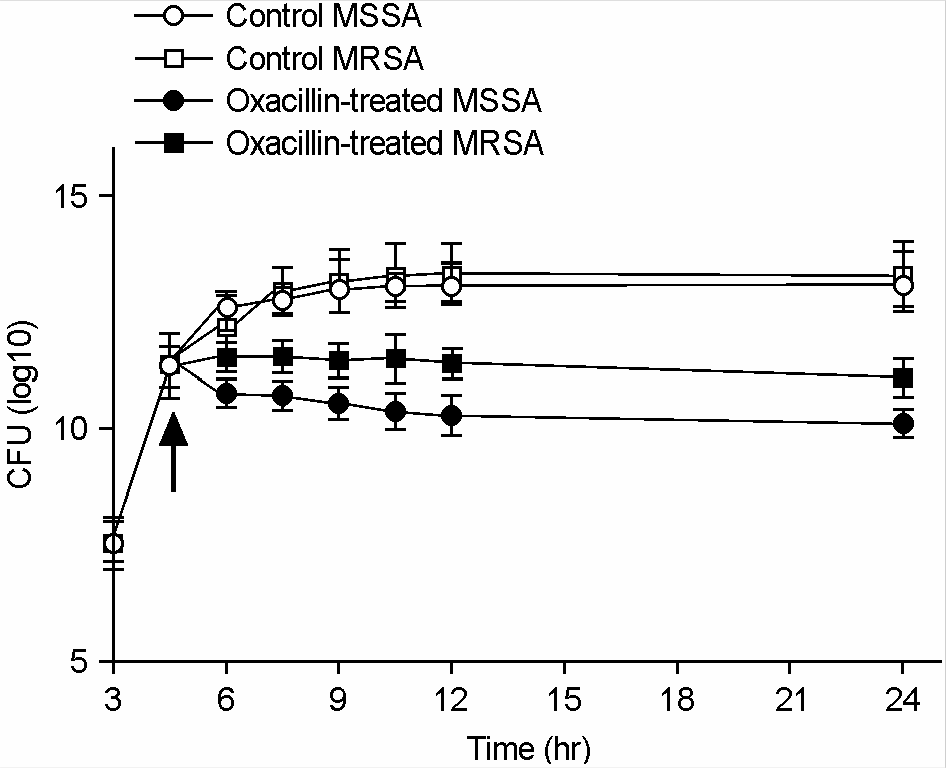 | Figure 2.Viability of MSSA and MRSA against oxacillin. Oxacillin of their respective MICs (MSSA, 0.5 μg/ml; MRSA, 1024 μg/ml) was added at 4.5 h (arrow). |
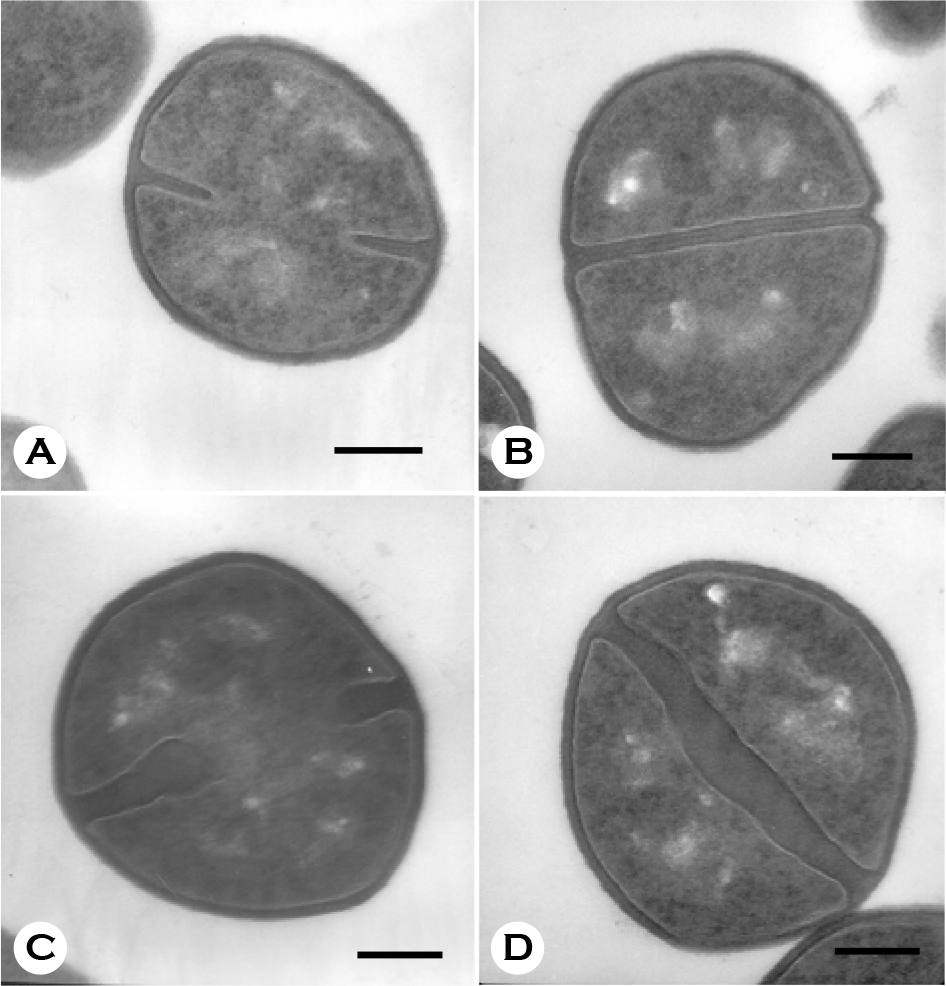 | Figure 3.Electron micrographs of MSSA grown in the absence (A, B) or presence (C, D) of oxacillin of its MIC (0.5 μg/ml) for 180 min. Bars = 260 nm. |
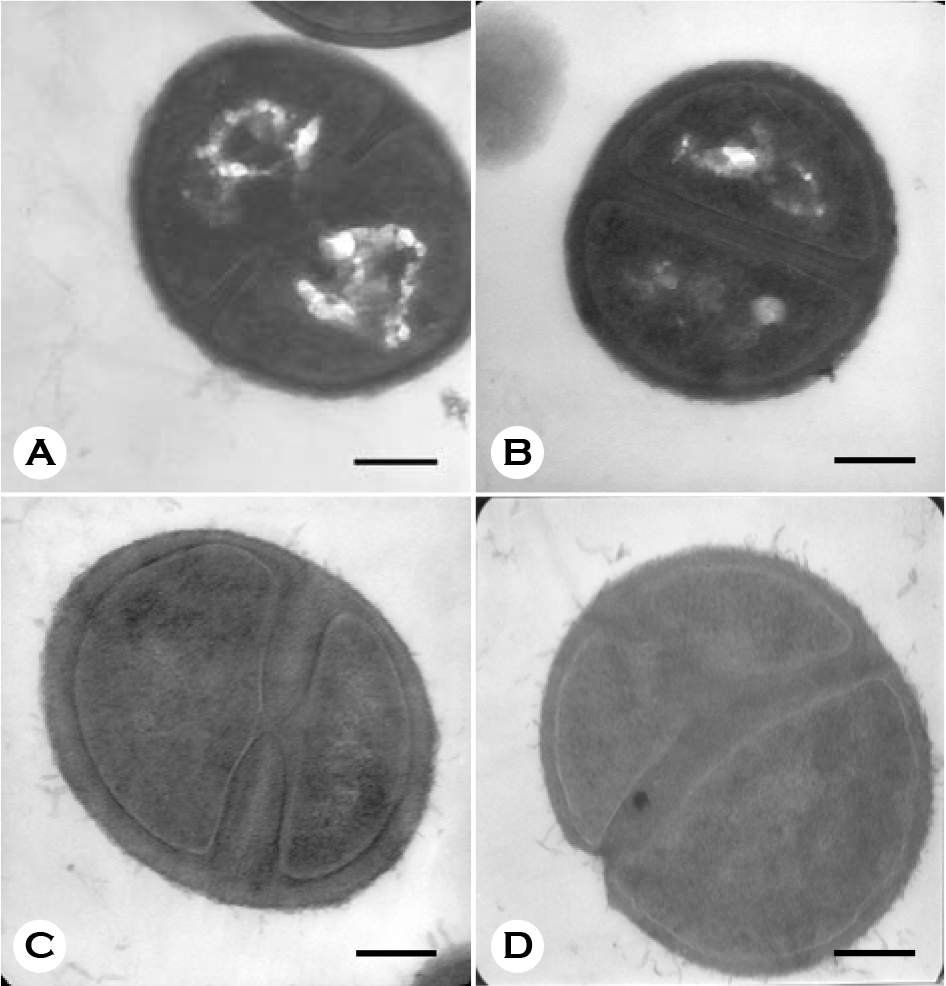 | Figure 4.Electron micrographs of MRSA grown in the absence (A, B) or presence (C, D) of oxacilln of its MIC (1024 μg/ml) for 180 min. Bars = 260 nm. |
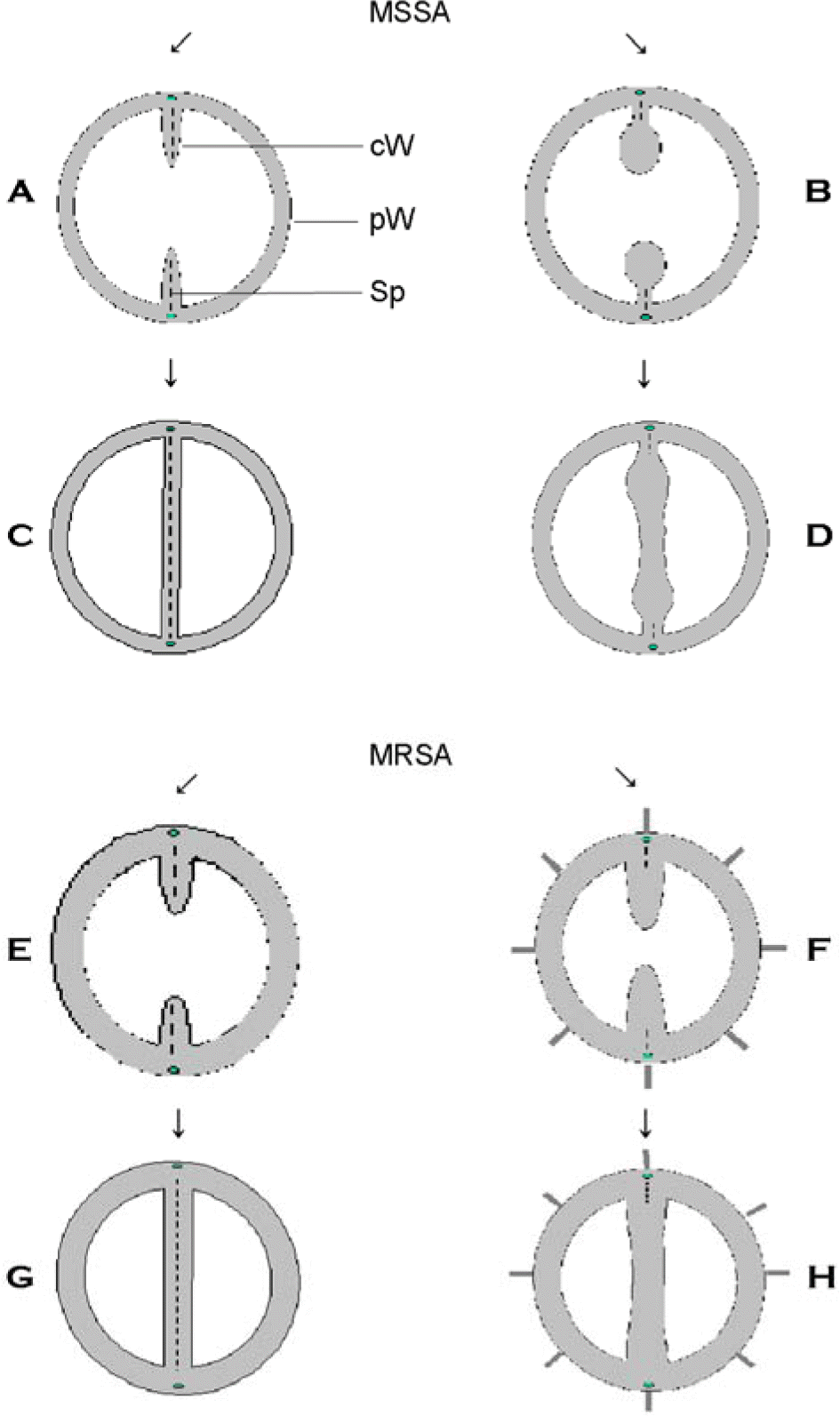 | Figure 5.Schematic diagram of alteration in cell wall and cross wall of MSSA and MRSA treated with oxacillin of their respective MICs. A, C, E, and G are control cells, and B, D, F and H are oxacillin-treated cells. cW, cross wall; pW, peripheral cell wall; Sp, splitting system. |
Table 1.
Comparison of MSSA and MRSA in ultrastructure observed by transmission electron microscope
| MSSA | MRSA | |||
|---|---|---|---|---|
| Control | Oxacillin-treated | Control | Oxacillin-treated | |
| Thickness of cell wall | 32.2±7.3 | 32.8±8.3 | 41.9±5.4 | 85.7±10.4∗ |
| Thickness of cross wall | 35.2±10.2 | 133.6±31.4∗ | 62.0±10.2 | 167.1±20.7∗ |
| Surface of cell wall | Smooth surface | Not changed | Rough surface | Rougher surface |
| Shape of cross wall | Thin and cone-shaped | Thick and club-shaped | Thin and cone-shaped | Thick and cone-shaped |
| Center of cross wall | Obvious splitting system | Obscure splitting system | Obvious splitting system | Obscure splitting system |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download