Abstract
Tuberculosis, which is caused by Mycobacterium tuberculosis (M. tb), is one of the most important infectious diseases in the world. Although many functional studies have been conducted on M. tb proteins in the post-genomic era, little is known about the function of many proteins expressed specifically during latency. Previously, we reported that Rv2041c from M. tb H37Rv is highly expressed under conditions of low pH and hypoxia, which represent the in vitro mimicry of latent tuberculosis. In the present study, increased expression levels of Rv2041c under hypoxia and low pH in vitro culture was confirmed by RT-PCR. Interestingly, Rv2041c showed significantly increased expression among genes of the same operon and genes belonging to the same functional group. Finally, the immune responses elicited by the recombinant (r) Rv2041c protein were investigated using ex vivo and in vivo models of M. tb infection. A significantly high level of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-12p40 was detected in a dose-dependent manner by treatment of murine bone marrow-derived macrophages with rRv2041c protein. In addition, IFN-γ and TNF-α secretion increased after stimulation with purified Rv2041c protein to lymphocytes from latent and active TB mice in a modified Cornell model. In conclusion, our findings suggest that Rv2041c is a new T-cell antigen and could be a potential vaccine candidate against M. tb infection by inducing a strong cellular immune response.
REFERENCES
1). Harries AD., Dye C. Tuberculosis. Ann Trop Med Parasitol. 2006. 100:415–31.
3). Young DB., Perkins MD., Duncan K., Barry CE 3rd. Confronting the scientific obstacles to global control of tuberculosis. J Clin Invest. 2008. 118:1255–65.

4). Dietrich J., Doherty TM. Interaction of Mycobacterium tuberculosis with the host: consequences for vaccine development. APMIS. 2009. 117:440–57.
5). Leung CC., Tam CM., Chan SL., Chan-Yeung M., Chan CK., Chang KC. Efficacy of the BCG revaccination programme in a cohort given BCG vaccination at birth in Hong Kong. Int J Tuberc Lung Dis. 2001. 5:717–23.
6). Rodrigues LC., Pereira SM., Cunha SS., Genser B., Ichihara MY., de Brito SC., Hijjar MA., Dourado I., Cruz AA., Sant'Anna C., Bierrenbach AL., Barreto ML. Effect of BCG revaccination on incidence of tuberculosis in school-aged children in Brazil: the BCG-REVAC cluster-randomised trial. Lancet. 2005. 366:1290–5.

7). Brandt L., Feino Cunha J., Weinreich Olsen A., Chilima B., Hirsch P., Appelberg R., Andersen P. Failure of the Mycobacterium bovis BCG vaccine: some species of environmental mycobacteria block multiplication of BCG and induction of protective immunity to tuberculosis. Infect Immun. 2002. 70:672–8.
8). Sable SB., Plikaytis BB., Shinnick TM. Tuberculosis subunit vaccine development: impact of physico-chemical properties of mycobacterial test antigens. Vaccine. 2007. 25:1553–66.

9). Fontan P., Aris V., Ghanny S., Soteropoulos P., Smith I. Global transcriptional profile of Mycobacterium tuberculosis during THP-1 human macrophage infection. Infect Immun. 2008. 76:717–25.
10). Monahan IM., Betts J., Banerjee DK., Butcher PD. Differential expression of mycobacterial proteins following phagocytosis by macrophages. Microbiology. 2001. 147:459–71.

11). Voskuil MI., Visconti KC., Schoolnik GK. Mycobacterium tuberculosis gene expression during adaptation to stationary phase and low-oxygen dormancy. Tuberculosis (Edinb). 2004. 84:218–27.
12). Kim SY., Lee BS., Shin SJ., Kim HJ., Park JK. Differentially expressed genes in Mycobacterium tuberculosis H37Rv under mild acidic and hypoxic conditions. J Med Microbiol. 2008. 57:1473–80.
13). Braibant M., Gilot P., Content J. The ATP binding cassette (ABC) transport systems of Mycobacterium tuberculosis. FEMS Microbiol Rev. 2000. 24:449–67.
14). Sutcliffe IC., Harrington DJ. Lipoproteins of Mycobacterium tuberculosis: an abundant and functionally diverse class of cell envelope components. FEMS Microbiol Rev. 2004. 28:645–59.
15). Sampaio EP., Elloumi HZ., Zelazny A., Ding L., Paulson ML., Sher A., Bafica AL., Shea YR., Holland SM. Mycobacterium abscessus and M. avium trigger Tolllike receptor 2 and distinct cytokine response in human cells. Am J Respir Cell Mol Biol. 2008. 39:431–9.
16). Beggs WH., Jenne JW. Mechanism for the pyridoxal neutralization of isoniazid action of Mycobacterium tuberculosis. J Bacteriol. 1967. 94:793–7.
17). Payton M., Pinter K. A rapid novel method for the extraction of RNA from wild-type and genetically modified kanamycin resistant mycobacteria. FEMS Microbiol Lett. 1999. 180:141–6.

18). Kaushik RS., Uzonna JE., Zhang Y., Gordon JR., Tabel H. Innate resistance to experimental African trypanosomiasis: differences in cytokine (TNF-alpha, IL-6, IL-10 and IL-12) production by bone marrow-derived macrophages from resistant and susceptible mice. Cytokine. 2000. 12:1024–34.
19). Yang CS., Lee DS., Song CH., An SJ., Li S., Kim JM., Kim CS., Yoo DG., Jeon BH., Yang HY., Lee TH., Lee ZW., El-Benna J., Yu DY., Jo EK. Roles of peroxiredoxin II in the regulation of proinflammatory responses to LPS and protection against endotoxin-induced lethal shock. J Exp Med. 2007. 204:583–94.

20). Ha SJ., Jeon BY., Kim SC., Kim DJ., Song MK., Sung YC., Cho SN. Therapeutic effect of DNA vaccines combined with chemotherapy in a latent infection model after aerosol infection of mice with Mycobacterium tuberculosis. Gene Ther. 2003. 10:1592–9.
21). Ha SJ., Jeon BY., Youn JI., Kim SC., Cho SN., Sung YC. Protective effect of DNA vaccine during chemotherapy on reactivation and reinfection of Mycobacterium tuberculosis. Gene Ther. 2005. 12:634–8.
22). Dannenberg AM Jr. Delayed-type hypersensitivity and cell-mediated immunity in the pathogenesis of tuberculosis. Immunol Today. 1991. 12:228–33.

23). Flynn JL., Goldstein MM., Chan J., Triebold KJ., Pfeffer K., Lowenstein CJ., Schreiber R., Mak TW., Bloom BR. Tumor necrosis factor-alpha is required in the protective immune response against Mycobacterium tuberculosis in mice. Immunity. 1995. 2:561–72.
24). Trinchieri G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat Rev Immunol. 2003. 3:133–46.

25). Flynn JL., Chan J., Triebold KJ., Dalton DK., Stewart TA., Bloom BR. An essential role for interferon gamma in resistance to Mycobacterium tuberculosis infection. J Exp Med. 1993. 178:2249–54.
26). Rezwan M., Grau T., Tschumi A., Sander P. Lipoprotein synthesis in mycobacteria. Microbiology. 2007. 153:652–8.

27). Jo EK., Yang CS., Choi CH., Harding CV. Intracellular signalling cascades regulating innate immune responses to Mycobacteria: branching out from Toll-like receptors. Cell Microbiol. 2007. 9:1087–98.

28). Jung SB., Yang CS., Lee JS., Shin AR., Jung SS., Son JW., Harding CV., Kim HJ., Park JK., Paik TH., Song CH., Jo EK. The mycobacterial 38-kilodalton glycolipoprotein antigen activates the mitogen-activated protein kinase pathway and release of proinflammatory cytokines through Toll-like receptors 2 and 4 in human monocytes. Infect Immun. 2006. 74:2686–96.

29). Ellertsen LK., Storla DG., Diep LM., Brokstad KA., Wiker HG., Hetland G. Allergic sensitisation in tuberculosis patients at the time of diagnosis and following chemotherapy. BMC Infect Dis. 2009. 9:100.

30). Chakravarty SD., Zhu G., Tsai MC., Mohan VP., Marino S., Kirschner DE., Huang L., Flynn J., Chan J. Tumor necrosis factor blockade in chronic murine tuberculosis enhances granulomatous inflammation and disorganizes granulomas in the lungs. Infect Immun. 2008. 76:916–26.

31). Sutherland JS., Adetifa IM., Hill PC., Adegbola RA., Ota MO. Pattern and diversity of cytokine production differentiates between Mycobacterium tuberculosis infection and disease. Eur J Immunol. 2009. 39:723–9.
Figure 1.
Experimental schedules for the animal models of latent and active TB. Mice were exposed to M. tb H37Rv in the inhalation chamber of an airborne infection apparatus. Bacteria were counted 2 weeks after exposure. Mice were then treated with INH and PZA for 3 months, starting 2 weeks after aerosol infection. Bacterial counts were obtained from lung and spleen of mice at 2 and 16 weeks after the completion of 3 months of chemotherapy. The active TB mice in the Cornell model were obtained by natural reactivation.
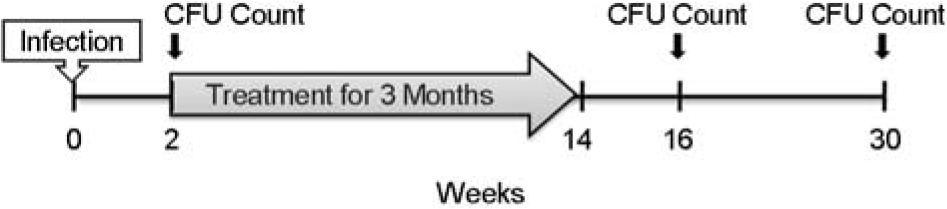
Figure 2.
SDS-PAGE analysis of dialyzed Rv2041c protein. Purified Rv2041c fusion protein was dialyzed against PBS and then resolved by 12% SDS-PAGE. The protein bands were visualized by staining with Coomassie brilliant blue. Lane 1 is a molecular weight marker (kDa). Lanes 2, 3, and 4 represent 2, 4, and 6 μg dialyzed recombinant Rv2041c protein, respectively.
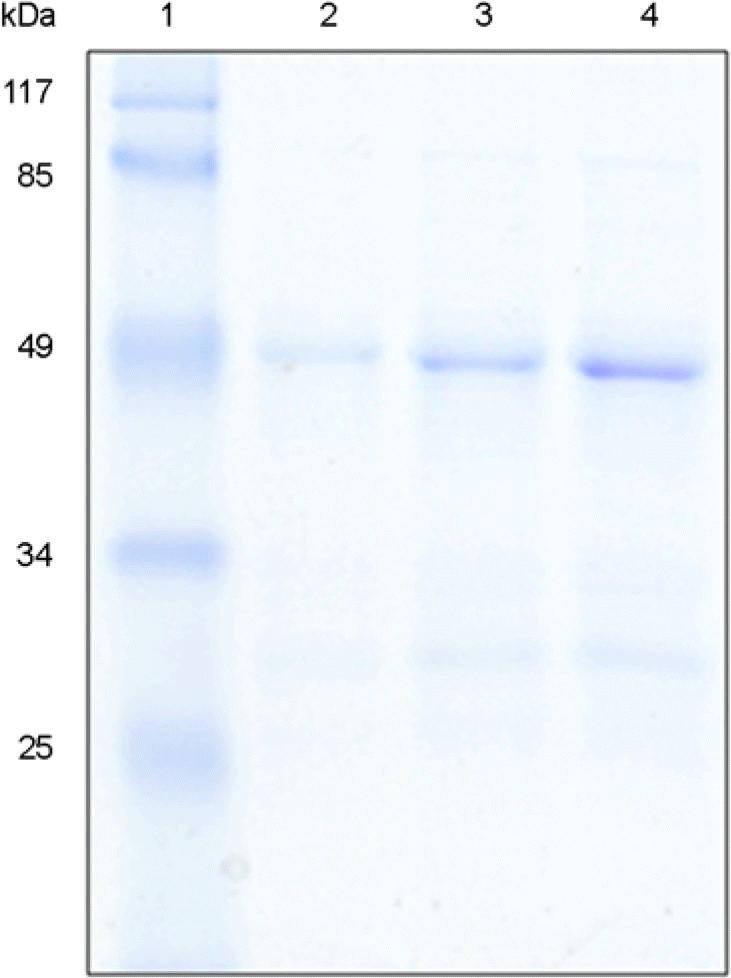
Figure 3.
RT-PCR analysis of Rv2038c, Rv2039c, Rv2040c, Rv2041c, lpqY, ugpB, and uspC in M. tb H37Rv cultured under various conditions. The transcription level of each gene from M. tb cultured at normal (pH 7.2) or acidic (pH 6.0) with hypoxic or anoxic conditions was measured by semi-quantitative RT-PCR (A). Based on the intensity of the bands, the results are expressed as fold-induction relative to the value for normal culture conditions (B). Lane 1, normal conditions (pH 7.2 and 21% oxygen tension); lane 2, hypoxic condition (pH 7.2 and 13% oxygen tension); lane 3, anoxic condition (pH 7.2 and 0% oxygen tension); lane 4, mildly acidic and hypoxic condition (pH 6.0 and 13% oxygen tension); lane 5, mildly acidic and anoxic condition (pH 6.0 and 0% oxygen tension).
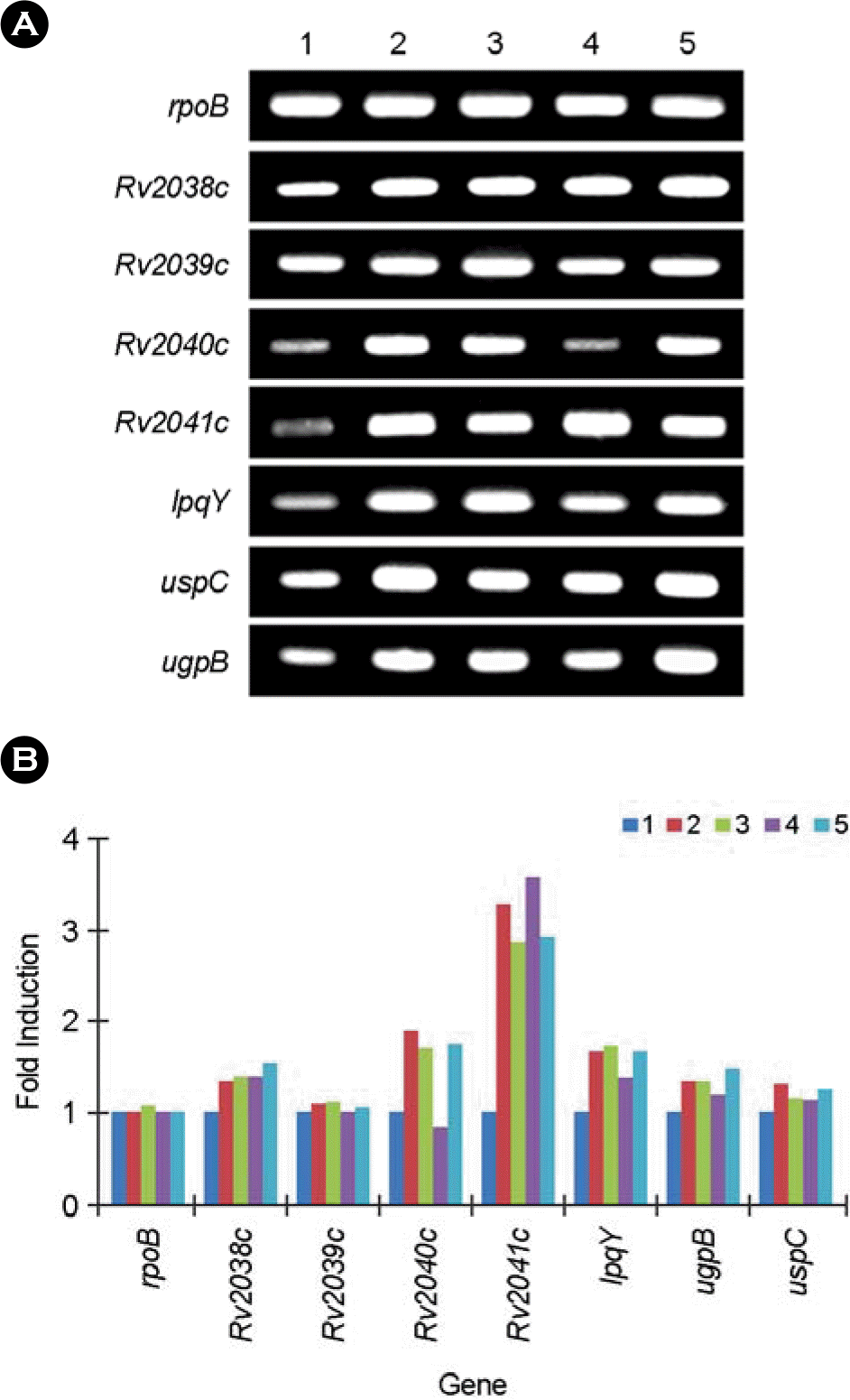
Figure 4.
IL-6, IL-12p40, and TNF-α production in response to purified Rv2041c recombinant protein in BMDMs from mice. BMDMs were treated with recombinant Rv2041c protein at concentrations ranging from 0.5 to 5 μg/ml. The supernatants were harvested after 18 h to assess cytokines by ELISA. Lane 1, non-treated (negative control); lane 2, LPS (positive control, 1 μg/ml); lane 3, rRv2041c (0.5 μg/ml); lane 4, rRv2041c (1 μg/ml); lane 5, rRv2041c (5 μg/ml). Values represent the mean ± SD of triplicate samples. ∗p < 0.05, ∗∗ p < 0.01; compared to LPS antigen (Student's t-test).
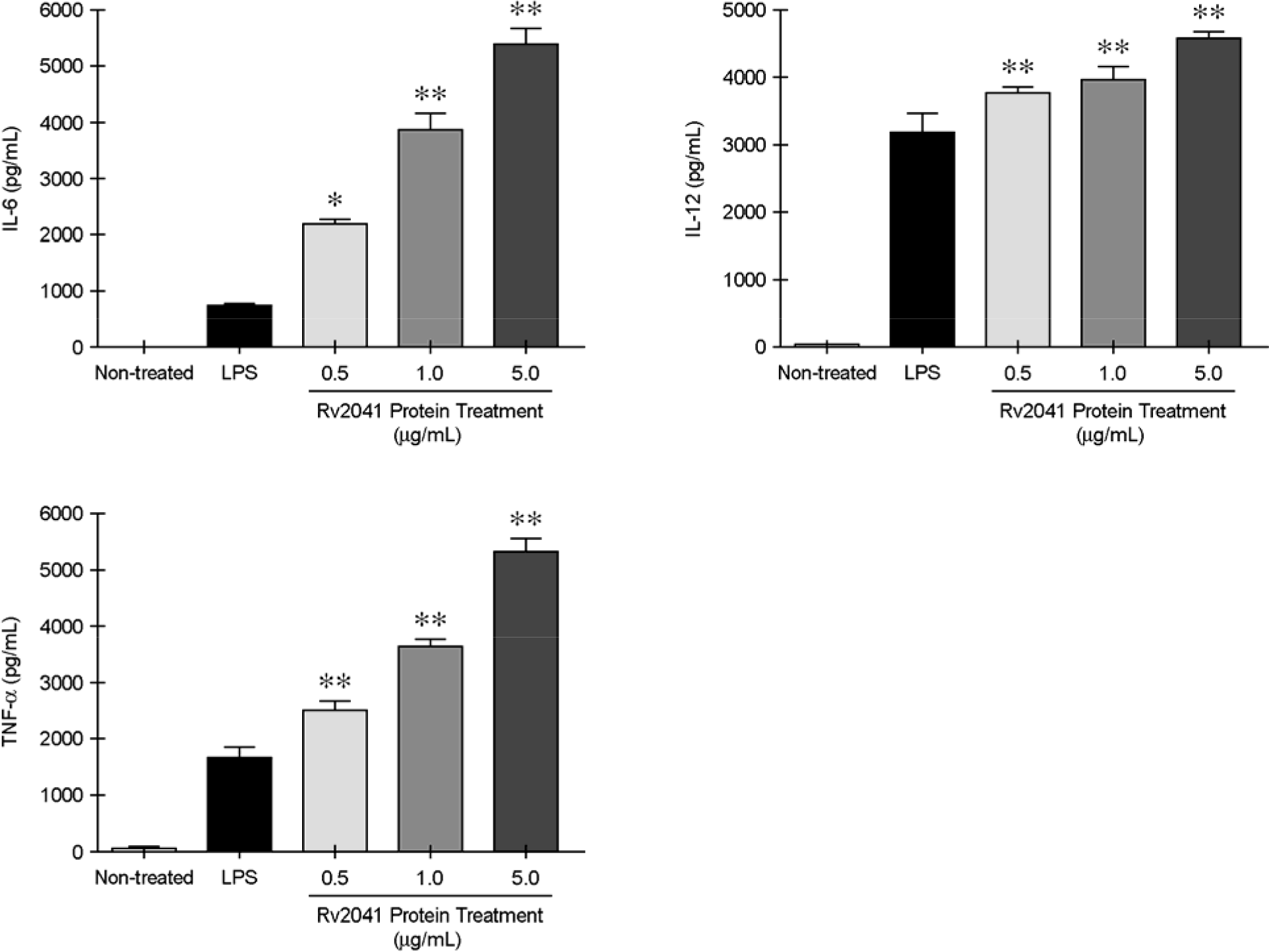
Figure 5.
IFN-γ and TNF-α production in response to purified Rv2041c recombinant protein in a latent and active TB model. The recombinant Rv2041c protein or Con A was used to treat lymphocytes of spleen from latent (A) and active (B) TB mice at a concentration of 10 μg/ml. The supernatants were harvested after 1 (gray bar) or 4 days (black bar) to assess cytokines by ELISA. Non-treated and Con A-treated cells were used as negative and positive controls, respectively. Values represent the mean ± SD of triplicate samples. ∗ p <0.05, ∗∗p < 0.01; compared to the Con A antigen (Student's t-test).
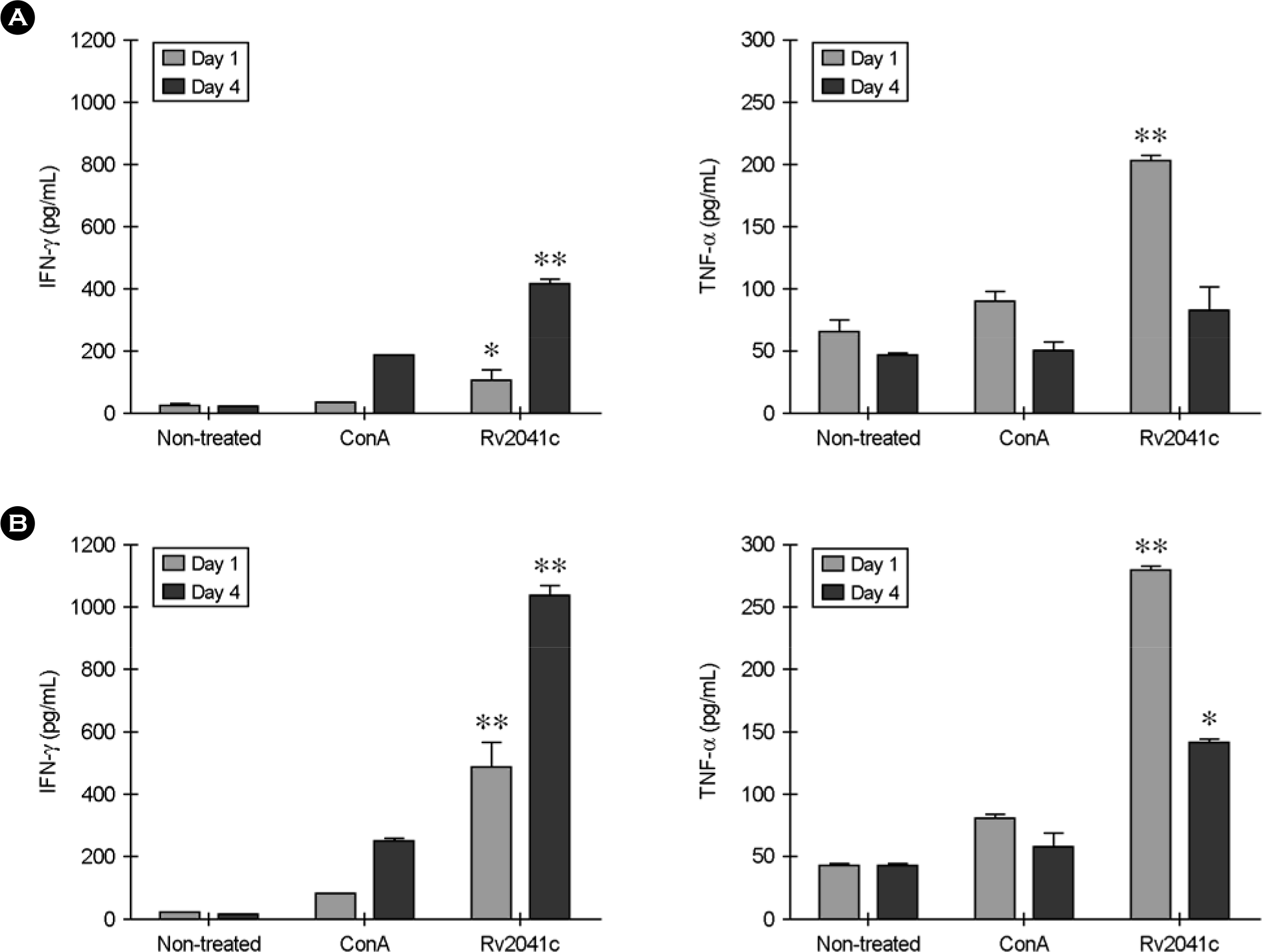
Table 1.
Primers used in RT-PCR




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download