Abstract
The expression pattern of immunoproteasomes in human thymus has not been analyzed but may have important consequences during thymic selection. Here we examined the expression patterns of immunoproteasome subunits in fetal and adult thymic tissues by immunohistochemistry and found that all three subunits are expressed in both cortical and medullary stromal cells. These data suggest that thymic selection in human can be affected by peptide repertoires generated by immunoproteasomes.
The TCR (T Cell Receptor) repertoire of peripheral T cells is generated in the thymus by the interaction of the αβ TCR with peptide-MHC (major histocompatibility complex) on stromal cells. Immature thymocytes are positively and negatively selected for recognition of self-peptides presented by MHC molecules (1,2). While positive selection of T cells takes place in the thymic cortex and is mediated by cortical epithelial cells, medullary epithelial cells as well as hematopoietic stromal cells such as dendritic cells and macrophages mediate negative selection (3,4). These two different processes shape the self-MHC restricted and self-peptide tolerant repertoire of peripheral T cells.
MHC class I molecules are displayed on all nucleated cells, including specialized antigen presenting cells such as thymic epithelial cells and dendritic cells. Cell-surface MHC class I in the thymus presents self-peptides to the outside world so that they can be recognized and then induce selection of immature thymocytes. This sophisticated system depends upon the presentation of antigenic peptides into the groove of MHC molecules. The generation of these peptides requires the degradation of proteins into peptide fragments by proteasomes (5).
The constitutive proteasome is composed of 28 subunits arranged in four stacked rings, each made up of seven subunits, two outer α and two inner β rings. Three (β1, β2 and β5) of the seven β-subunits are proteolytically active (5). While these subunits are constitutively expressed, when cells are incubated with interferon-γ, they are replaced by newly-assembled proteins, large multifunctional peptidase 2 (LMP2), LMP7, and LMP10, respectively, leading to the replacement of constitutive proteasome with immunoproteasomes. Immunoproteasomes generate a spectrum of antigenic peptides different from those produced by constitutive proteasomes as they generate peptides with increased affinity for MHC class I (6-8). A recent report showed that expression of immunoproteasome subunits is differentially regulated in mouse thymic cortical and medullary stromal cells (9). In addition, studies with LMP2 and LMP7 deficient mice suggested that defects in immunoproteasome subunits affect the CD8+ T cell repertoire (10,11). These results suggest that immunoproteasomes are involved in the selection process in thymic development.
Here, we examined the expression pattern of immunoproteasome subunits in human thymus using immunohistochemical assay. Interestingly, all three of the immunoproteasome subunits (LMP2, 7, and 10) were detected in both cortical and medullary stromal cells.
Thirty paraffin-embedded thymic tissues (1 from myasthenia gravis, 2 from thymoma, and 27 from fetal thymi) were obtained from the files of the Department of Pathology Seoul National University Hospital, from the period of 1997 to 2004. Immunohistochemical staining of formalin-fixed paraffin-embedded tissue sections were performed with primary antibodies for human immunoproteasome subunits (LMP2, 7 and 10 respectively, provided by S. Ferrone, Roswell Park Center Institute, (12)) and Envision-polymer-HRP kit (DAKO, Glostrup, Denmark).
The characteristics of patients and expressions of immunoproteasome subunits are summarized in Table I. In both fetal and adult thymic tissues, the stromal cells such as epithelial cells and macrophages in the cortex and medulla were immunopositive for LMP2, 7 and 10. However, immature thymocytes did not show immunoreactivity for all three different immunoproteasome subunits. Representative staining examples are shown in Fig. 1.
The proteasomal system has been optimized by evolving alternative proteolytically active subunits whose expression depends on interferon-γ produced during immune response. By comparing the amino acid sequences of active sites of constitutive and immuno-proteasome subunits it predicted that the immunoproteasome specializes to generate peptides with hydrophobic and basic carboxyl termini, which can be preferred by MHC class I. Therefore, the immunoproteasome seems to generate a specialized peptide repertoire that is likely to be enriched for immunocompetent peptides (5,13). Substantial differences between peptide repertoires of constitutive and immuno-proteasomes would mean that following secretion of interferon-γ during infection, interferon-γ exposed cells would display peptides that were not presented in the absence of infection. This suggests that a more diverse TCR repertoire is needed to deal with many kinds of pathogens under inflammatory conditions.
In this study we demonstrated that three subunits of immunoproteasome are expressed in thymic stromal cells such as epithelial cells, not thymocytes. Self-peptides and MHC molecules in thymic cortical epithelial cells are indispensable in thymic positive selection (1,14). In addition it was reported that a diversity of peptides presented by MHC in the thymus is important in shaping a diverse repertoire of mature T cells (15). Together, including our data, it is conceivable that the imunoproteasomes in cortical epithelial cells of fetal thymi may contribute to the generation of a peptide repertoire large enough for positive selection of a fully diverse TCR repertoire.
We also reported that immunoproteasomes are expressed in cortical as well as medually epithelial cells and macrophages. During the course of infection, interferon-γ exposed cells display self-peptides generated by immunoproteasomes. This clearly necessitates clonal deletions of TCRs recognizing self peptides generated under condition of infection from the T cell repertoire and assumes that immunoproteasomes should be expressed in medullary epithelial cells and macrophages of the thymus.
In contrast to our human data, a recent paper showed more complicated expression patterns of immunoproteasomes in mouse thymus. In mouse thymus, although immunoporteasomes were expressed highly in medullary epithelial cells or dendritic cells, they were not expressed in cortical epithelial cells in absence of interferon-γ treatment or Listeria infection (9). The differences in expression patterns can be explained in many ways. First of all, the above paper utilized different detection methods. We used immunohistochemistry and did not manipulate the tissue except for fixation and staining. However, the above authors isolated cells and then analyzed the expression pattern by RT-PCR and immunoblotting (9). Therefore, it could not exclude that the manipulating step such as the isolation procedure affected the immunoproteasome expression. Second, we should consider differences in species. It can be speculated that although a peptide repertoire generated in the absence of immunoproteasomes could select TCR repertoires diverse enough to survive during a short mouse life span, immunoproteasomes and more diverse peptides would be needed to shape complete TCR repertoire in the case of humans (with a much longer life span).
In this study we did not detect possible functional consequences of differential immunoproteasome expression in thymic stroma. Further functional studies using in vitro systems such as thymic organ cultures and knock-down systems may shed light on the roles of immunoproteasomes in thymic selection in humans.
Figures and Tables
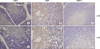 | Figure 1Thymic sections from human adults and fetuses were examined by immunohistochemistry using antibodies for three immunoproteasome subunits (LMP2, 7, 10) at maginification of ×200 (top row: A, B, C) or 400 (bottom row: D, E, F). (A, D) LMP2; (B, E) LMP7; (C, F) LMP10. Cells that appear brown are positive for immmunoreactivity. Three subunits are expressed exclusively in stromal cells such as epithelial cells and macrophages of both the cortex and medulla, but not thymocytes. Staining antibodies and magnification folds are indicated. |
ACKNOWLEDGEMENTS
We thank Dr. S. Ferrone (Roswell Park Cancer Center Institute, Buffalo, NY, USA) for providing antibodies.
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (2009-0065250).
References
1. Starr TK, Jameson SC, Hogquist KA. Positive and negative selection of T cells. Annu Rev Immunol. 2003. 21:139–176.

2. Siggs OM, Makaroff LE, Liston A. The why and how of thymocyte negative selection. Curr Opin Immunol. 2006. 18:175–183.

3. Cosgrove D, Chan SH, Waltzinger C, Benoist C, Mathis D. The thymic compartment responsible for positive selection of CD4+ T cells. Int Immunol. 1992. 4:707–710.

4. Anderson G, Jenkinson EJ. Lymphostromal interactions in thymic development and function. Nat Rev Immunol. 2001. 1:31–40.

5. Kloetzel PM, Ossendorp F. Proteasome and peptidase function in MHC-class-I-mediated antigen presentation. Curr Opin Immunol. 2004. 16:76–81.

6. Früh K, Gossen M, Wang K, Bujard H, Peterson PA, Yang Y. Displacement of housekeeping proteasome subunits by MHC-encoded LMPs: a newly discovered mechanism for modulating the multicatalytic proteinase complex. Embo J. 1994. 13:3236–3244.

7. Gaczynska M, Rock KL, Spies T, Goldberg AL. Peptidase activities of proteasomes are differentially regulated by the major histocompatibility complex-encoded genes for LMP2 and LMP7. Proc Natl Acad Sci U S A. 1994. 91:9213–9217.

8. Tanaka K, Kasahara M. The MHC class I ligand-generating system: roles of immunoproteasomes and the interferon-gamma-inducible proteasome activator PA28. Immunol Rev. 1998. 163:161–176.

9. Nil A, Firat E, Sobek V, Eichmann K, Niedermann G. Expression of housekeeping and immunoproteasome subunit genes is differentially regulated in positively and negatively selecting thymic stroma subsets. Eur J Immunol. 2004. 34:2681–2689.

10. Chen W, Norbury CC, Cho Y, Yewdell JW, Bennink JR. Immunoproteasomes shape immunodominance hierarchies of antiviral CD8(+) T cells at the levels of T cell repertoire and presentation of viral antigens. J Exp Med. 2001. 193:1319–1326.

11. Toes RE, Nussbaum AK, Degermann S, Schirle M, Emmerich NP, Kraft M, Laplace C, Zwinderman A, Dick TP, Müller J, Schönfisch B, Schmid C, Fehling HJ, Stevanovic S, Rammensee HG, Schild H. Discrete cleavage motifs of constitutive and immunoproteasomes revealed by quantitative analysis of cleavage products. J Exp Med. 2001. 194:1–12.

12. Bandoh N, Ogino T, Cho HS, Hur SY, Shen J, Wang X, Kato S, Miyokawa N, Harabuchi Y, Ferrone S. Development and characterization of human constitutive proteasome and immunoproteasome subunit-specific monoclonal antibodies. Tissue Antigens. 2005. 66:185–194.

13. Niedermann G. Immunological functions of the proteasome. Curr Top Microbiol Immunol. 2002. 268:91–136.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download