Abstract
Clinical and preclinical in vivo immune cell imaging approaches have been used to study immune cell proliferation, apoptosis and interaction at the microscopic (intra-vital imaging) and macroscopic (whole-body imaging) level by use of ex vivo or in vivo labeling method. A series of imaging techniques ranging from non-radiation based techniques such as optical imaging, MRI, and ultrasound to radiation based CT/nuclear imaging can be used for in vivo immune cell tracking. These imaging modalities highlight the intrinsic behavior of different immune cell populations in physiological context. Fluorescent, radioactive or paramagnetic probes can be used in direct labeling protocols to monitor the specific cell population. Reporter genes can also be used for genetic, indirect labeling protocols to track the fate of a given cell subpopulation in vivo. In this review, we summarized several methods dealing with dendritic cell, macrophage, and T lymphocyte specifically labeled for different macroscopic wholebody imaging techniques both for the study of their physiological function and in the context of immunotherapy to exploit imaging-derived information and immune-based treatments.
Recent advances in molecular imaging allow us to visualize cellular and subcellular process within living subjects at the molecular level as well as at the anatomical level (1). In a narrow meaning, molecular imaging implies molecular-genetic imaging for visualizing cellular processes by combination of molecular biology and biomedical imaging. This marvelous technique provides research attention not only in molecular cell biology but also in related fields, such as genetics, pharmacology, chemistry, physics, engineering and medicine.
Especially, the development of controlled gene delivery and gene expression vector systems promotes to generate various types of reporter genes for visualization such as fluorescent and bioluminescent proteins (2-5). Conventionally, a recombinant plasmid, which contains a reporter gene, has been used to monitor cells by assaying reporter gene expression. By using constitutive promoters, it is possible to monitor cell survival and localization in vivo. Similarly, by using cell/tissue-specific promoter or inducible promoter, it is possible to visualize long term monitoring of cell fate such as differentiation. However, this method cannot be used directly in in vivo imaging because the insertion of the reporter gene demands genetic modification and the invariable light intensity from reporter proteins was not enough to be visualized for non-invasive whole animal imaging. For this reason, different methods are required for monitoring viable cells in vivo. Accumulation of specific imaging signal for amplifying its intensity makes it possible to visualize the localization, quantification, and repetitive determination of cells (6,7), but more effective strategies have been tried to answer the questions for monitoring in vivo by recruiting methods from radio-pharmaceutics and physics. Radiolabeled small compounds and paramagnetic probes were developed for imaging specific proteins and magnetic signals, accelerating noninvasive molecular imaging (8,9). In recent publications, these strategies have been reviewed by researchers for introducing the concept of molecular imaging (10-12).
The development of molecular imaging technologies has been facilitated by associated development of imaging instruments as well as imaging materials such as imaging enhancement agents, probes, ligands and reporter constructs. In clinical practice, in vivo imaging is based on the use of computed tomography (CT), positron emission tomography (PET), single photon emission computed tomography (SPECT), magnetic resonance imaging (MRI), and ultrasound (US) techniques.
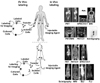
Small animal models have a great advantage in disease studies that are difficult, or impossible, to perform in humans. Repetitive observation is a virtue of non invasive small animal imaging, and provides information of a spatial and a temporal dimension in disease development and progression. Multiple imaging modalities including CT, PET, SPECT, MR, US, and various optical techniques using fluorescence imaging (FLI) and bioluminescence imaging (BLI) are also available for small animal imaging (13,14). Because each modality has its unique advantages and disadvantages (Table I), advances in imaging technology have been developed to generate combined imaging modalities, such as PET/CT, SPECT/ CT, and PET/MR (15). Using the newly developed instrumental merging techniques, more precise localization information of anatomical and molecular activity can be acquired in a single imaging session (16). The advantages of multimodal approaches to molecular imaging provide better images for visualizing cellular, functional, and morphological changes (Fig. 1).
The above techniques are mostly macroscopic whole body imaging. For microscopic imaging, fluorescence based intravital multiphoton microscopic imaging has been used to visualize cell morphology, distribution, and interaction with higher solution in living organism (17,18). Multiphoton microscopy utilizes pairs of photons with a higher wavelength more efficiently than classic confocal microscopy in the detection of inner fluorescent signals thanks to their high penetration through tissue. However, the tissue of interest in living animals was usually exposed to implanted transparent chambers on their skin for multiphoton microscopy. For this reason, this method is limited by a field of view and its procedure is very invasive. Therefore, this review will cover only macroscopic imaging.
Though significant challenges still remain to be overcome when we compare the image of a small animal model with a human, which comes from different size (20 g of body weight for a mouse vs. 70 kg for a human) and physiology (approximately 1.5 ml of total blood volume in a mouse vs. 5,000 ml in a human; 730/min for heart rate in a mouse vs. 75/min in a human) (19), but animal models still provide a unique source of in vivo information and the predictable link between in vitro studies and human patients. An important advantage of in vivo imaging is that it allows the same animals to be examined repeatedly at different time points, thereby decreasing the variability in the animal population and reducing the number of animals required.
Despite advances in both the diagnostic and therapeutic use of molecular imaging, the detection and the tracking of immune cells still constitute major challenges. In vivo immune cell imaging depends on the ability of the cells to generate a detectable signal and different cells have to be labeled with different kinds of marker using ex vivo or in vivo labeling methods (Fig. 2).
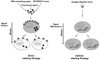
Labeling strategies can be divided into two classes: direct and indirect. Direct labeling involves the use of probes that are internalized by the immune cells, while indirect labeling requires genetic modification, which results in the expression of detectable reporter protein or enzymatic reaction for imaging (Fig. 3).
For direct labeling, immune cells are harvested, labeled ex vivo with probes such as fluorophores, radiotracers or paramagnetic nanoparticles and then reinfused, allowing them to be visualized by FLI, PET/SPECT and, MRI respectively. The great advantage of direct labeling is the simple procedure; labeling probes bind to a specific target or penetrate the cell membrane easily. Particularly, several labeling probes are already approved for clinical use such as paramagnetic nanoparticles (MNPs) for MRI. This strategy, however, has major drawbacks. First, the level of labeling depends on the label retaining ability of cells. Second, this technique is only useful for the in vivo imaging of terminally differentiated cells such as dendritic cells and macrophages, while it does not allow long-term monitoring of cell viability and proliferation in the body. This is because the label is lost as a result of cell death or the marker decay, and also because the label is diluted at each mitotic event. Third, this strategy has the possibility of affecting motility, viability, functionality, proliferation and differentiation potential of labeled cells depending on the specific feature of label such as generating oxidative stress, radiation damage, and photodynamic damage.
For optical imaging, quantum dots (QDs) and fluorescent molecules have been developed to monitor immune cell distribution. By conjugating a specific optical probe designed for ex vivo labeling, it is possible to study immune cell tracking in vivo. These probes can easily label cells by entering the cytoplasm or by binding to the membrane. For visualization with nuclear imaging (PET/SPECT), 18F-fluorodeoxyglucose (FDG) and 64Cu-pyruvaldehyde-bis (N4-methylthiosemicarbazone) (PTSM) for PET, 99mTc-hexamethyl propylene amine oxime (HMPAO), 111In-oxine for SPECT have been extensively used to label cells ex vivo. Various MNPs have been produced such as iron oxides nanoparticles (dark signal in T2- and T2*-weighted imaging), gadolinium chelates (bright signal in T1-weighted imaging) and manganese chelates (dark T2-weighted signal to bright T1 signal in lysosome) for monitoring the biodistribution of immune cells and their localization at the target organ. While phagocytic cells can easily internalize nanoparticles, non-phagocytic cells need positive charged carrier molecules, such as poly-L-lysine or protamine sulphate, to facilitate the nanoparticles engulfment. Table II shows the various direct imaging strategies for tracking immune cells.
On the other hand, indirect labeling requires genetic manipulation of cells by ex vivo transduction with a vector carrying a reporter gene such as fluorescence protein (GFP, RFP), luciferase, Sodium Iodide Symporter (NIS), transferrin receptor, etc. The reporter protein expression generates signals to track the viable cells in vivo. Various fluorescence reporter genes emitting at different wavelengths also have been developed, but only some of them can be used for in vivo imaging because of their low photon emission and tissue absorption (4). Recently, near-infrared (NIR) fluorescence reporters have been developed for in vivo application to overcome the limited spatial resolution and low tissue penetration. Luciferase genes, including several enzymes whose expression can be revealed by in vivo BLI, have been used to immune cells in order to monitor their activity (20-22).
A constitutive promoter provides the information of homing and localization of living cells in the target organ. Depending on the tissue specific promoter or the specific regulatory element used to create the construct, this strategy can be used to study a specific cellular process such as activation or differentiation of the labeled cells. The beauty of indirect labeling is that the reporter gene is passed on to the next generations without signal dilution (Fig. 3), making it possible to track the fate of a given cell population for a long term study. However, the insertion of the exogenous reporter gene requires genetic modification of the cells and thus this strategy is restricted in preclinical research.
Despite the pros and cons of direct/indirect cell labeling strategies, each strategy has its advantages to tracking immune cells. Direct cell labeling strategies are fast and easy for monitoring the short term fate of cells, while indirect labeling allows a long term evaluation of cells. Direct labeling using clinically approved molecules such as radionuclides and MNPs can be applied to translational research in a short period of time. Clearly, with all the issues related to genetic modification including insertion mutagenesis and changing replication/proliferation rate, indirect labeling strategies may not be used for human application. However, by use of reporter transgenic mouse (23), it is possible to gain a better understanding of immune cell movement escape from the disadvantage of indirect labeling at the preclinical level.
Since the maturation state, route of administration, migration ability and homing to the target site can influence the therapeutic efficacy of immunotherapy, cell labeling strategies have been developed for monitoring the fate of injected immune cells in space and time. Protocols have been developed based on the use of different cell populations, mainly dendritic cells (DCs), T lymphocytes and macrophages. For tumor immunotherapy, DCs need to reach the lymph node in order to evoke a consistent cyto-toxic immune response. For this reason, most of imaging studies focus on biodistribution and dynamic in vivo monitoring of lymph node migration of DCs to evaluate the therapeutic efficacy. 111In-Oxine and 99mTc-conjugated radionuclide compound have been used for nuclear imaging. 99mTc-HMPAO was not a good compound for DC homing visualization. Mixture of 111In-Oxine labeled DCs and SPIO-labeled DCs revealed higher number of lymph nodes by SPECT and MRI (24).
T-cell mediated treatments are primarily evaluated by infiltration of immune cells into tumor and their differentiation into effector, helper, memory, and cytotoxic cells. Because T lymphocytes are a proliferating population, direct labeling is not the first choice. In order to prevent signal dilution by clonal expansion, T cells are usually labeled in an indirect method. T cell biodistribution was easily monitored through BLI/PET by engineering luciferase/HSV1-TK-containg construct (25). T cells specific for human prostate specific membrane antigen (hPSMA) or human carcinoembryonic antigen (hCEA) were administrated into murine lung tumor model of prostate cancer, and imaged PET with 2'-18F-fluoro-2'-deoxy-1-beta-d-arabinofuranosyl-5'-ethyluracil (18F-FEAU). PET signal was observed only in hPSMA-specific T cell injected mouse not in hCEA-specific T cell injected mouse.
Macrophage plays pivotal roles in the development of immune response against pathogens and tumors. Recently, imaging tumor associated macrophages (TAMs) provide visualized location in the myeloid-rich region of the tumor stroma by using fluorescent molecular tomography (FMT) and MRI (26). AMTA680 which is constituted by SPIO labeled with fluorescent dye was used for multimodal imaging of macrophages. By T2-weighted MRI imaging due to the core iron and 3D reconstruction of FMT, both magnetic and fluorescence signals were successfully detected.
Here, we described both direct and indirect immune cell labeling strategies. Direct labeling is more clinically applicable, but restricted in the visualization of terminally differentiated, non-proliferating cells. In this case, the stability of labeling and the physical half-life of probe are more important than the survival of cells. On the other hand, indirect labeling does not lose the imaging marker with proliferation and marker expression is closely related to cell viability and function. This provides possible tools of monitoring long-term cell survival, proliferation and activation. However, random insertion of the reporter cassette and elevated level of the reporter expression could induce cell behavior by genetic perturbation. As well as the labeling procedure, technical features of imaging device should be considered for in vivo non invasive whole body imaging. Optical approaches are easy to use, relatively inexpensive, and highly sensitive, but limited in small animal imaging to this day. Nuclear imaging such as PET or SPECT has problems with the use of radionuclides but higher sensitivity, easier quantification and clinical application can be offered. Since MRI generates higher morphological resolution, PET/MR or SPECT/MR can be expected for generating better spatio-temporal resolution, sensitivity, and accuracy in non invasive immune cell imaging.
Figures and Tables
Table II
Immune cell tracking

HSV-TK, herpes simplex virus thymidine kinase; NIS, sodium iodide symporter; NET, norepinephrine transporter; TfR, transferrin recepter; FR, ferritin receptor; QDs, quantum dots; SPIO, superparamagnetic iron oxide; USPIO, ultra-small superparamagnetic iron oxide; CLIO, cross-linked iron oxide; MION, monocrystalline iron oxide; PFPE, perfluoropolyether; 99mTc-hexamethyl propylene amine oxime (HMPAO), 111In-oxine, 18F-fluorodeoxyglucose (FDG) and 64Cu-pyruvaldehyde-bis (N4-methylthiosemicarbazone) (PTSM).
ACKNOWLEDGEMENTS
This study was supported by a grant of the Korea Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (Grant No: A101446).
References
1. Blasberg RG, Gelovani-Tjuvajev J. In vivo molecular-genetic imaging. J Cell Biochem Suppl. 2002. 39:172–183.

2. Forss-Petter S, Danielson PE, Catsicas S, Battenberg E, Price J, Nerenberg M, Sutcliffe JG. Transgenic mice expressing beta-galactosidase in mature neurons under neuron-specific enolase promoter control. Neuron. 1990. 5:187–197.

3. Yu YA, Timiryasova T, Zhang Q, Beltz R, Szalay AA. Optical imaging: bacteria, viruses, and mammalian cells encoding light-emitting proteins reveal the locations of primary tumors and metastases in animals. Anal Bioanal Chem. 2003. 377:964–972.

4. Shaner NC, Steinbach PA, Tsien RY. A guide to choosing fluorescent proteins. Nat Methods. 2005. 2:905–909.

5. Tjuvajev JG, Stockhammer G, Desai R, Uehara H, Watanabe K, Gansbacher B, Blasberg RG. Imaging the expression of transfected genes in vivo. Cancer Res. 1995. 55:6126–6132.
6. Gambhir SS, Barrio JR, Phelps ME, Iyer M, Namavari M, Satyamurthy N, Wu L, Green LA, Bauer E, MacLaren DC, Nguyen K, Berk AJ, Cherry SR, Herschman HR. Imaging adenoviral-directed reporter gene expression in living animals with positron emission tomography. Proc Natl Acad Sci U S A. 1999. 96:2333–2338.

7. Brown RS, Leung JY, Fisher SJ, Frey KA, Ethier SP, Wahl RL. Intratumoral distribution of tritiated-FDG in breast carcinoma: correlation between Glut-1 expression and FDG uptake. J Nucl Med. 1996. 37:1042–1047.
8. Ichikawa T, Högemann D, Saeki Y, Tyminski E, Terada K, Weissleder R, Chiocca EA, Basilion JP. MRI of transgene expression: correlation to therapeutic gene expression. Neoplasia. 2002. 4:523–530.

9. Blasberg RG, Tjuvajev JG. Molecular-genetic imaging: current and future perspectives. J Clin Invest. 2003. 111:1620–1629.

10. Massoud TF, Gambhir SS. Molecular imaging in living subjects: seeing fundamental biological processes in a new light. Genes Dev. 2003. 17:545–580.

11. Serganova I, Mayer-Kukuck P, Huang R, Blasberg R. Molecular imaging: reporter gene imaging. Handb Exp Pharmacol. 2008. (185 Pt 2):167–223.

12. Hoffman JM, Gambhir SS. Molecular imaging: the vision and opportunity for radiology in the future. Radiology. 2007. 244:39–47.

13. Townsend DW, Carney JP, Yap JT, Hall NC. PET/CT today and tomorrow. J Nucl Med. 2004. 45:Suppl 1. 4S–14S.
14. Kang JH, Chung JK. Molecular-genetic imaging based on reporter gene expression. J Nucl Med. 2008. 49(Suppl 2):164S–179S.

15. Pichler BJ, Judenhofer MS, Catana C, Walton JH, Kneilling M, Nutt RE, Siegel SB, Claussen CD, Cherry SR. Performance test of an LSO-APD detector in a 7-T MRI scanner for simultaneous PET/MRI. J Nucl Med. 2006. 47:639–647.
16. Ottobrini L, Martelli C, Trabattoni DL, Clerici M, Lucignani G. In vivo imaging of immune cell trafficking in cancer. Eur J Nucl Med Mol Imaging. 2011. 38:949–968.

17. Dunn KW, Sutton TA. Functional studies in living animals using multiphoton microscopy. ILAR J. 2008. 49:66–77.

19. De Jong M, Maina T. Of mice and humans: are they the same?- Implications in cancer translational research. J Nucl Med. 2010. 51:501–504.

20. Pham W, Kobukai S, Hotta C, Gore JC. Dendritic cells; therapy and imaging. Expert Opin Biol Ther. 2009. 9:539–564.

21. Mandl S, Schimmelpfennig C, Edinger M, Negrin RS, Contag CH. Understanding immune cell trafficking patterns via in vivo bioluminescence imaging. J Cell Biochem Suppl. 2002. 39:239–248.

22. Negrin RS, Contag H. In vivo imaging using bioluminescence: a tool for probing graft-versus-host disease. Nat Rev Immunol. 2006. 6:484–490.

23. Song MG, Kang B, Jeon JY, Chang J, Lee S, Min CK, Youn H, Choi EY. In vivo imaging of differences in early donor cell proliferation in graft-versus-host disease hosts with different pre-conditioning doses. Mol Cells. 2012. 33:79–86.

24. de Vries IJ, Lesterhuis WJ, Barentsz JO, Verdijk P, van Krieken JH, Boerman OC, Oyen WJ, Bonenkamp JJ, Boezeman JB, Adema GJ, Bulte JW, Scheenen TW, Punt CJ, Heerschap A, Figdor CG. Magnetic resonance tracking of dendritic cells in melanoma patients for monitoring of cellular therapy. Nat Biotechnol. 2005. 23:1407–1413.

25. Dobrenkov K, Olszewska M, Likar Y, Shenker L, Gunset G, Cai S, Pillarsetty N, Hricak H, Sadelain M, Ponomarev V. Monitoring the efficacy of adoptively transferred prostate cancer-targeted human T lymphocytes with PET and bioluminescence imaging. J Nucl Med. 2008. 49:1162–1170.

26. Leimgruber A, Berger C, Cortez-Retamozo V, Etzrodt M, Newton AP, Waterman P, Figueiredo JL, Kohler RH, Elpek N, Mempel TR, Swirski FK, Nahrendorf M, Weissleder R, Pittet MJ. Behavior of endogenous tumor-associated macrophages assessed in vivo using a functionalized nanoparticle. Neoplasia. 2009. 11:459–468.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download