Abstract
Figures and Tables
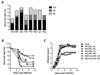 | Figure 1Gln and Leu, but not their combination, provide protection against mucosal infection of HSV-1. (A) In vivo anti-herpes activity of several amino acids. BALB/c mice (n=8) were infected with HSV-1 McKrae strain, and were then administered 4% solution of the indicated amino acid (200 µl/mouse) daily via intra-peritoneal injection. The proportion of survived mice was examined at day 6, 9, and 12 days post-infection. (B) Survival proportion of Gln and Leu-treated mice against HSV-1 mucosal infection. BALB/c mice were infected with HSV-1 McKrae strain, and were then administered a 4% solution of Gln, Leu, or their combined mixture (200 µl/mouse) daily via intra-peritoneal injection. The challenged mice were examined daily for any signs of inflammation, illness, and death until 15 days post-challenge. (C) The clinical severity of Gln and Leu-treated mice following HSV-1 mucosal infection. The clinical severity of Gln and Leu-treated mice were scored daily after HSV-1 mucosal infection. The data represent the average of clinical severity derived from each group. |
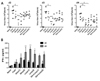 | Figure 2Virus burden and intravaginal tract IFN-γ levels of Gln and Leu-treated mice following HSV-1 mucosal infection. (A) Virus titer in vaginal lavages of Gln and Leu-treated mice following HSV-1 mucosal infection. BALB/c mice were infected with HSV-1 McKrae strain, and were then administered a 4% solution of Gln, Leu, or their combined mixture (200 µl/mouse) via intra-peritoneal injection. The vaginal lavages collected at 2nd, 3rd, and 4th day post-infection were used for plaque assay to determine virus titer. Dots on the graph represent the individual levels of virus and the bold bar shows the median level of each group. (B) IFN-γ levels in vaginal lavages of Gln and Leu-treated mice following HSV-1 mucosal infection. The levels of IFN-γ in vaginal lavages collected at 2nd and 4th day post-infection were determined by ELISA. The data represent the average±SD of levels derived from each group. Statistical significance calculated using Student's t-test. *p<0.05; **p<0.01 compared with the levels of control group. |
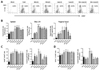 | Figure 3NK cell activity of Gln and Leu-treated mice following HSV-1 mucosal infection. (A) Frequency of NK cells (CD3-DX5+) in vaginal tract cells of Gln and Leu-treated mice. The vaginal tract cells were prepared from Gln and Leu-treated mice 2 days post-infection and were used for analysis of NK cell frequency. The values on dot-plots represent the average of NK cell frequencies derived from each group (n=4). (B) The absolute number of NK cells in spleen, iliac LN, and vaginal tract of Gln and Leu-treated mice. The absolute number of NK cells in spleen, iliac LN, and vaginal tract was determined by flow cytometric analysis 2 days post-infection. (C and D) The proportion and absolute number of granzyme B and IFN-γ-producing NK cells in spleen of Gln and Leu-treated mice. The proportion and absolute number of granzyme B (C) and IFN-γ producing cells (D) in NK cell population were determined by intracellular staining following PMA and ionomycin stimulation 2 days post-infection. The data represent the average±SD of levels derived from each group. Statistical significance was calculated using Student's t-test. *p<0.05; **p<0.01; ***p<0.001 compared with the levels of control group. |
 | Figure 4HSV-1 antigen-specific CD4+ T cell responses in Gln and Leu-treated mice following mucosal infection. HSV-1 antigen-specific CD4+ T cells were detected by combined intracellular staining of CD154, IFN-γ, and TNF-α in response to stimulation of UV- inactivated HSV-1 viral antigen 5 days after HSV-1 infection. The absolute number of IFN-γ+, TNF-α+, and IFN-γ+TNF-α+CD4+ T cells in spleen (A), iliac LN (B), and vaginal tract (C) were calculated after gated on CD4+CD154+ T cells. The data show the average±SD of levels derived from each group (n=4). Statistical significance was calculated using the Student's t-test. *p<0.05; **p<0.01; ***p<0.001 as compared with the levels of control group. |
Table I

*BALB/c mice (n=4) given the indicated amino acid were infected with HSV-1 McKrae strain (1.0×106 pfu/mouse) via intravaginal route. Splenocytes were prepared 5 days p.i., stained with indicated Abs, and analyzed by flow cytometry. The results are expressed as the average±SD of positive cells (CD69+, CD62Llow, CD44high or CD80+) in a given cell subset (CD4+, CD8+ or CD19+). †p<0.05; ‡p<0.01; §p<0.001; Student's t-test, calculated between control group infected with HSV-1 and the indicated group.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download