This article has been
cited by other articles in ScienceCentral.
Abstract
Background
Bortezomib has significant activity in treating multiple myeloma (MM). The risk of herpes zoster (HZ) has been reported to increase significantly with bortezomib treatment, but the predisposing factors for HZ are not clear. This study is a retrospective analysis of the relevant risk factors for HZ in Korean MM patients treated with bortezomib.
Methods
Sixty-six patients with refractory or relapsed MM who underwent chemotherapy with bortezomib were included in the study. Prophylactic antiviral drugs were not used for treatment. The following parameters were reviewed: age, gender, stage and type of MM, extent of previous treatment, history of HZ, duration from the time of diagnosis to the time of bortezomib treatment initiation, and absolute lymphocyte counts (ALC) at the time of bortezomib treatment initiation.
Results
The incidence of HZ was 16.7%. There were no intergroup differences between the HZ-positive and the HZ-negative groups with regard to a history of HZ, number of previous treatments, and exposure to steroids before bortezomib treatment. The median duration from the time of MM diagnosis to the time of bortezomib treatment initiation in the HZ-positive group was significantly shorter than that in the HZ-negative group. The median ALC at the time of bortezomib initiation in the HZ-positive group was significantly lower than that in the HZ-negative group.
Conclusion
Bortezomib itself might act as a risk factor for HZ by inhibiting cell-mediated immunity, and patients with low ALC at the time of bortezomib treatment initiation were at greater risk of HZ during bortezomib treatment.
Go to :

Keywords: Multiple myeloma, Protease inhibitors, Herpes zoster
INTRODUCTION
Multiple myeloma (MM) is characterized by the malignant proliferation of a plasma cell originating from a single clone and is associated with innate impairment of humoral immunity rather than cell-mediated immunity. In addition, in patients with MM, treatment-associated immunodeficiency increases the susceptibility to various infections.
Bortezomib, introduced in the late 1990s, has significant activity against MM and is one of the most widely used drugs in the treatment of MM. As a proteosome inhibitor, bortezomib directly induces apoptosis of myeloma cells and acts in the bone marrow microenvironment, inhibits myeloma cells binding to bone marrow stromal cells, alters cytokine secretion, and restricts angiogenesis [
1-
3]. Notably, the risk of HZ was significantly higher in patients treated with bortezomib. In the Assessment of Proteasome Inhibition for Extending Remissions APEX study, the incidence of HZ after bortezomib treatment was significantly higher than that after dexamethasone treatment (13% vs. 5%,
P<0.001) [
4]. Other studies have reported the incidence of bortezomib-treatment associated HZ to be as high as 10-22.3% [
5-
11].
The predisposing factors to HZ during bortezomib treatment are not clear. History of HZ, performance status (PS), age, sex, disease stage, baseline β2-microglobulin levels, Hb, platelets, ALC, absolute neutrophil count (ANC), the frequency of prior therapies, and disease duration prior to initiation of bortezomib treatment did not show any associations with HZ development [
4,
8].
Therefore, we aimed to perform a retrospective analysis investigating the incidence and the relevant risk factors for HZ development in Korean MM patients undergoing bortezomib treatment.
Go to :

MATERIALS AND METHODS
From 3 centers located in the Southern part of the Republic of Korea, 73 patients previously diagnosed with MM (July 1998-December 2009) and treated with bortezomib-containing chemotherapy were selected for the study. Seven patients were excluded due to the absence of follow-up (n=1) or initial (n=6) data, and the latter 6 did not develop HZ. A total of 66 patients were included in the study.
Bortezomib was used as a single agent or combined with other drugs such as dexamethasone, doxorubicin, cyclophosphamide, or thalidomide. The patients all had refractory or relapsed MM, and were given bortezomib as a salvage therapy. Prophylactic antiviral drugs were not used. Patients who developed HZ during the bortezomib treatment were identified.
At the time of MM diagnosis, the following parameters were recorded: age, gender, the Eastern Cooperative Oncology Group (ECOG) PS, the international staging system, type and level of heavy chain and light chains, serum β2-microglobulin, albumin, creatinine, Hb, WBC, ANC, ALC, and platelet counts. The potential risk factors that may predispose patients to HZ were also recorded at the initiation of bortezomib treatment: HZ history, frequency of prior treatments, past steroid exposure, median duration from MM diagnosis to bortezomib initiation, serum β2-microglobulin, albumin, creatinine, Hb, WBC, ANC, ALC, and any other drugs combined with bortezomib.
The clinical and laboratory characteristics of HZ-positive and HZ-negative groups were compared using chi-square test and Wilcoxon rank sum test. Statistical significance was defined as P<0.05. All statistical analyses were performed using SPSS software (version 15.0; SPSS Inc., Chicago, IL, USA).
Go to :

RESULTS
1. HZ incidence and characteristics during bortezomib treatment
The incidence of HZ during bortezomib treatment was 16.7% (12/66 patients). The median duration from bortezomib treatment initiation to HZ onset was 2 months (range, 1-13 months). In 1 patient, HZ took longer than 6 months to develop (13 months), whereas in the remaining 11 patients, HZ developed within 6 months of the initial bortezomib administration. The patient with the 13-month interval time received 10 cycles of bortezomib in combination with dexamethasone for 28 months until disease progression developed. Nine of the 12 patients (75%) resumed bortezomib after receiving HZ treatment. The remaining 3 patients (25%) discontinued bortezomib treatment for reasons of disease progression, self-refusal, and poor PS, respectively.
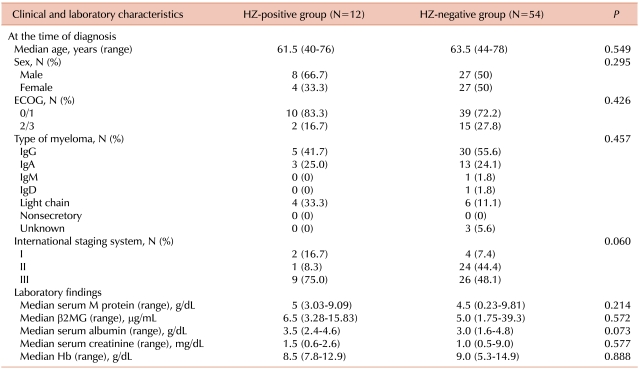
2. Patient characteristics at MM diagnosis
The median age, gender, ECOG PS, disease type, and stage of disease did not account for significant differences between the HZ-positive and HZ-negative groups (
Table 1). Laboratory parameters such as the median serum M protein, β2-microglobulin, albumin, creatinine, and Hb were similar between the 2 groups.
Table 1
Comparison of patient characteristics between HZ-positive group and HZ-negative group at the time of diagnosis of multiple myeloma.

3. Patient characteristics at bortezomib treatment initiation
A history of HZ prior to bortezomib treatment in the HZ-positive group occurred no more frequently than in the HZ-negative group (8.3% vs. 14.9%,
P=0.554) (
Table 2). Thus, a history of HZ at the initiation of bortezomib treatment may have no influence on HZ occurrence during bortezomib treatment.
Table 2
Comparison of patient characteristics between HZ-positive group and HZ-negative group at the initiation of bortezomib treatment.
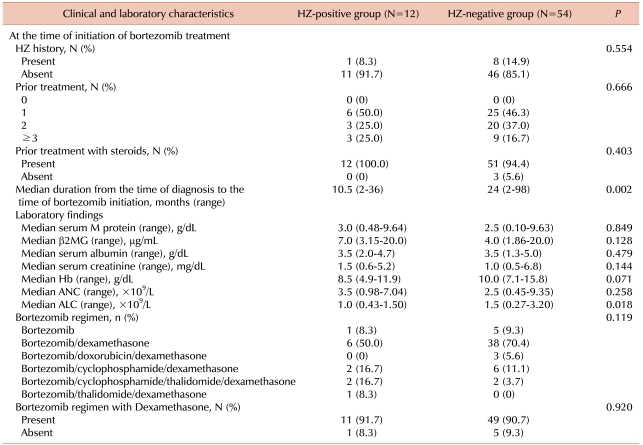

Although all patients previously received more than 1 treatment, the number of treatments prior to bortezomib initiation did not significantly associate with HZ incidence (
P=0.119). The rate of steroid exposure did not differ significantly between the two groups. However, in the HZ-positive group, the median duration from MM diagnosis to the initiation of bortezomib treatment was significantly shorter when compared to the HZ-negative group (10.5 vs. 24 months,
P=0.002) (
Fig. 1).
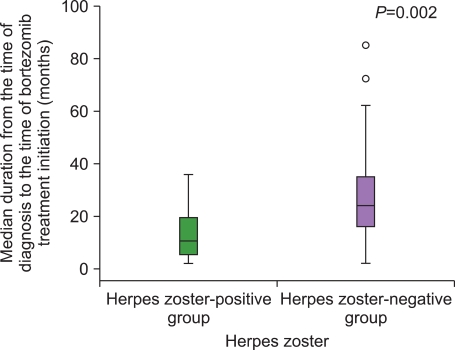 | Fig. 1Median duration from the time of diagnosis to the time of bortezomib treatment initiation (months) in the herpes zoster-positive and herpes zoster-negative groups. The interval between the time of diagnosis and the time of bortezomib treatment initiation in the herpes zoster-positive group was significantly shorter than that in the herpes zoster-negative group (P=0.002). 
|
No significant differences were detected in laboratory parameters (e.g., serum M protein level, β2-microglobulin, albumin, creatinine, Hb, and ANC) at the initiation of bortezomib treatment. However, the median ALC at bortezomib treatment initiation was significantly lower in the HZ-positive group (1.12×10
9/L vs. 1.57×10
9/L,
P=0.018) (
Fig. 2).
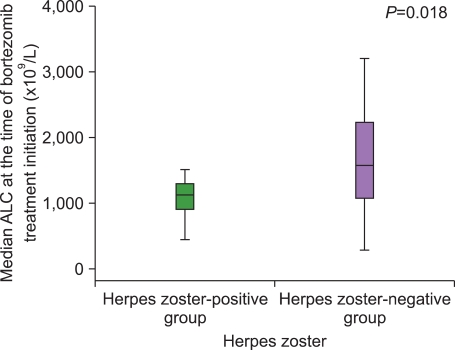 | Fig. 2Median ALC at the time of bortezomib treatment initiation in the herpes zoster-positive and herpes zoster-negative groups. The median absolute lymphocyte counts (ALC) at the time of bortezomib initiation in the herpes zoster-positive group was significantly lower than that in the herpes zoster-negative group (P=0.018). 
|
In both groups, more than half of the patients were given bortezomib in combination with dexamethasone. Neither dexamethasone nor other drugs used in combination with bortezomib were significantly associated with HZ occurrence during treatment.
Go to :

DISCUSSION
HZ commonly occurs in elderly and immunocompromised individuals, and is associated with decreased cell-mediated immunity [
12]. In immunocompromised individuals, particularly in those with impaired cell-mediated immunity, HZ can disseminate and cause more serious organ involvement, including hepatitis, pneumonia, and encephalitis [
12]. However, impaired innate immunity in myeloma primarily affects humoral immunity rather than cell-mediated immunity; therefore, patients with MM were not at increased risk for HZ [
13].
Bortezomib is one of the most widely used drugs for treating MM, and it is effective as one of the first-line regimen in previously untreated myeloma patients [
7] as well as the second-line regimen in relapsed or refractory patients [
14-
16].
Several studies have indicated increased HZ risk during bortezomib treatment. In a phase III APEX trial, the incidence of HZ significantly increased in the bortezomib group compared to the high-dose dexamethasone group in relapsed MM patients [
4]. Other studies identified an association between bortezomib and increased HZ incidence in the range of 10-22.3% in relapsed MM [
6,
8,
10,
11]. The overall HZ incidence in the present study was 16.7% in patients with refractory or relapsed MM, which is comparable to previous studies.
In addition to myeloma-related immunodeficiency and its complications, cumulative immunosuppression by salvage therapy may also increase susceptibility to infection. However, the present study showed a significantly shorter duration from MM diagnosis to bortezomib treatment initiation in the HZ-positive group compared to the HZ-negative group, identical to data reported in the previous study [
8], suggesting that disease duration is not a risk factor for zoster development. This result is supported by several recent studies on bortezomib-associated HZ in newly diagnosed myeloma patients. The incidence of HZ during bortezomib treatment in newly diagnosed patients is similar to HZ incidence in relapsed or refractory cases, in the range of 13-20% [
5,
7,
9,
17].
Several experimental studies have shown that bortezomib decreases the number and function of CD56+ NK cells and CD8+ cytotoxic T cells, with no definite decrease in CD4+ T cells [
18], and it inhibits the function of dendritic cells [
19]. Bortezomib targets the 26S proteasome and thereby prevents the activation of nuclear factor (NF)-κB, which then leads to inhibition of T cell activation as well as anti-myeloma effects [
20]. Although mechanisms are not clear, this may lead to impaired cell-mediated immunity such as above. Thus, the use of bortezomib might itself act as a predisposing factor to HZ. In the present study, the median ALC at bortezomib treatment initiation in the HZ-positive group was significantly lower than in the HZ-negative group. Although no lymphocyte subset or other immune cell data has been acquired and the sample size is small, the decreased ALC at bortezomib treatment initiation may reflect increased risk for the development of HZ during bortezomib treatment.
Although untreated MM patients were not at increased risk for HZ [
13], HZ was more likely to occur following dexamethasone-based regimens due to decreased cell-mediated immunity [
17]. However, as shown in the phase III APEX trial, the incidence of HZ was significantly higher in the bortezomib group despite the comparable ALC baseline levels detected in bortezomib-treated patients with HZ and dexamethasone-treated patients with HZ [
4]. The incidence of
Candida and other fungal infections was significantly higher in the dexamethasone group, and the overall incidence of infection was similar between the two groups [
4]. These results suggest that bortezomib and dexamethasone have different immunosuppressive properties, and the associated HZ development may involve more than the degree of lymphocytopenia.
In conclusion, the use of bortezomib itself is a risk factor for HZ because it inhibits T cell activation and impairs cell-mediated immunity. Clinically, patients who presented decreased ALC at the initiation of bortezomib treatment were associated with HZ development. Future studies will focus on serial changes of the specific lymphocyte subsets and other immune cells that affect zoster development, and on the strategy of prophylaxis.
Go to :







 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download