Abstract
Background:
Curcumin is a naturally occurring biologically active compound, and it has been shown to possess potent anti-inflammatory, anti-tumor and anti-oxidative properties. It is known for its anti-proliferative and proapoptotic effects in several cancer cells. Curcumin's effects on the mechanisms of cell survival and the expression of various cytokines were investigated in U266 cells and the in vivo effects of curcumin were examined using an animal model.
Methods:
Cell proliferation assay and flow cytometry were used to examine cell proliferation, along with cell cycle analysis. The protein expressions were analyzed by Western blotting and the expressed levels of cytokines were analyzed by the ELISA method.
Results:
Curcumin inhibited U266 cell growth in a dose-dependent and time-dependent manner. Cell cycle analysis showed an increased sub-G1 phase, a down regulated cyclinD1 expression and an induced p21 expression. Apoptosis induced a down regulated procaspase 3 expression and it induced cleaved PARP. Curcumin inhibited the IL (interleukin)-6 induced cell signal pathway via decreasing the STAT1 an 3, Erk cyclinD1 and c-myc expressions. Also, administration of 25mg/kg curcumin to a U266 animal model inhibited cancer cell engraftment in the bone marrow and it decreased the IL-6, sIL-6R and IL-8 expression levels.
REFERENCES
1). Hess U. The plasma cell myeloma—molecular pathogenesis and target therapies. Ther Umsch. 2006. 63:233–6.
2). Oyeyinka GO., Ofei V., Maddy SQ, et al. Homogeneous immunoglobulins in Ghanaians living in Accra, Ghana. Afr J Med Med Sci. 2004. 33:311–6.
3). Umeda M., Adachi Y., Tomiyama J, et al. Bone lesions in elderly multiple myeloma. Nippon Ronen Igakkai Zasshi. 2002. 39:631–8.

4). Shionoiri A., Horiuchi T., Onouchi T, et al. Hypercalcemia induced with the plasma levels of parathyroid hormone-related peptide in multiple myeloma. Intern Med. 2000. 39:810–3.

5). Tucci M., Grinello D., Cafforio P., Silvestris F., Dammacco F. Anemia in multiple myeloma: role of deregulated plasma cell apoptosis. Leuk Lymphoma. 2002. 43:1527–33.

6). Bharti AC., Shishodia S., Reuben JM, et al. Nuclear factor-kappaB and STAT3 are constitutively active in CD138+ cells derived from multiple myeloma patients, and suppression of these transcription factors leads to apoptosis. Blood. 2004. 103:3175–84.
7). Anderson KC., Lust JA. Role of cytokines in multiple myeloma. Semin Hematol. 1999. 36(1 Suppl 3):14–20.
8). Costes V., Portier M., Lu ZY., Rossi JF., Bataille R., Klein B. Interleukin-1 in multiple myeloma: producer cells and their role in the control of IL-6 production. Br J Haematol. 1998. 103:1152–60.

9). Lee JW., Chung HY., Ehrlich LA, et al. IL-3 expression by myeloma cells increases both osteoclast formation and growth of myeloma cells. Blood. 2004. 103:2308–15.

10). Treon SP., Anderson KC. Interleukin-6 in multiple myeloma and related plasma cell dyscrasias. Curr Opin Hematol. 1998. 5:42–8.

11). Jourdan M., Tarte K., Legouffe E., Brochier J., Rossi JF., Klein B. Tumor necrosis factor is a survival and proliferation factor for human myeloma cells. Eur Cytokine Netw. 1999. 10:65–70.
12). Qiang YW., Kopantzev E., Rudikoff S. Insulinlike growth factor-I signaling in multiple myeloma: downstream elements, functional correlates, and pathway cross-talk. Blood. 2002. 99:4138–46.

13). Nishino H., Tokuda H., Satomi Y, et al. Cancer prevention by antioxidants. Biofactors. 2004. 22:57–61.

14). Liao S., Lin J., Dang MT, et al. Growth suppression of hamster flank organs by topical application of catechins, alizarin, curcumin, and myristoleic acid. Arch Dermatol Res. 2001. 293:200–5.

15). Ammon HP., Wahl MA. Pharmacology of Curcuma longa. Planta Med. 1991. 57:1–7.
16). Koo JY., Kim HJ., Jung KO., Park KY. Curcumin inhibits the growth of AGS human gastric carcinoma cells in vitro and shows synergism with 5-fluorouracil. J Med Food. 2004. 7:117–21.
17). Sun M., Yang Y., Li H, et al. The effect of curcumin on bladder cancer cell line EJ in vitro. Zhong Yao Cai. 2004. 27:848–50.
18). Li H., Che Y., Tang W. Effects of curcumin on proliferation and apoptosis in human hepatic cells. Zhonghua Gan Zang Bing Za Zhi. 2002. 10:449–51.
19). Kuo ML., Huang TS., Lin JK. Curcumin, an antioxidant and anti-tumor promoter, induces apoptosis in human leukemia cells. Biochim Biophys Acta. 1996. 1317:95–100.

20). Duvoix A., Morceau F., Schnekenburger M, et al. Curcumin-induced cell death in two leukemia cell lines: K562 and Jurkat. Ann N Y Acad Sci. 2003. 1010:389–92.

21). Puthier D., Bataille R., Amiot M. IL-6 up-regulates mcl-1 in human myeloma cells through JAK/STAT rather than ras/MAP kinase pathway. Eur J Immunol. 1999. 29:3945–50.

22). Tobe M., Isobe Y., Tomizawa H., Nagasaki T., Takahashi H., Hayashi H. A novel structural class of potent inhibitors of NF-kappa B activation: structure-activity relationships and biological effects of 6-aminoquinazoline derivatives. Bioorg Med Chem. 2003. 11:3869–78.
23). Mitsiades N., Mitsiades CS., Richardson PG, et al. The proteasome inhibitor PS-341 potentiates sensitivity of multiple myeloma cells to conventional chemotherapeutic agents: therapeutic applications. Blood. 2003. 101:2377–80.

24). Schenkein D. Proteasome inhibitors in the treatment of B-cell malignancies. Clin Lymphoma. 2002. 3:49–55.

25). Salem M., Elbaz O., Zahran M, et al. Malignancy: identification of predictors of disease status and progression in patients with myeloma (MM). Hematology. 2000. 5:41–5.
Fig. 1
The growth of U266 cells was inhibited and activated apoptotic related protein by curcumin treatment. Curcumin treated with at various concentrations and times and curcumin (50∼75μM) reduced cell viability by 40∼60% for 72 hrs (A). U266 cell treated with indicated concentration of curcumin for 24 hrs. Apoptosis induced by exposure of 25 and 50μM concentration of curcumin (B).
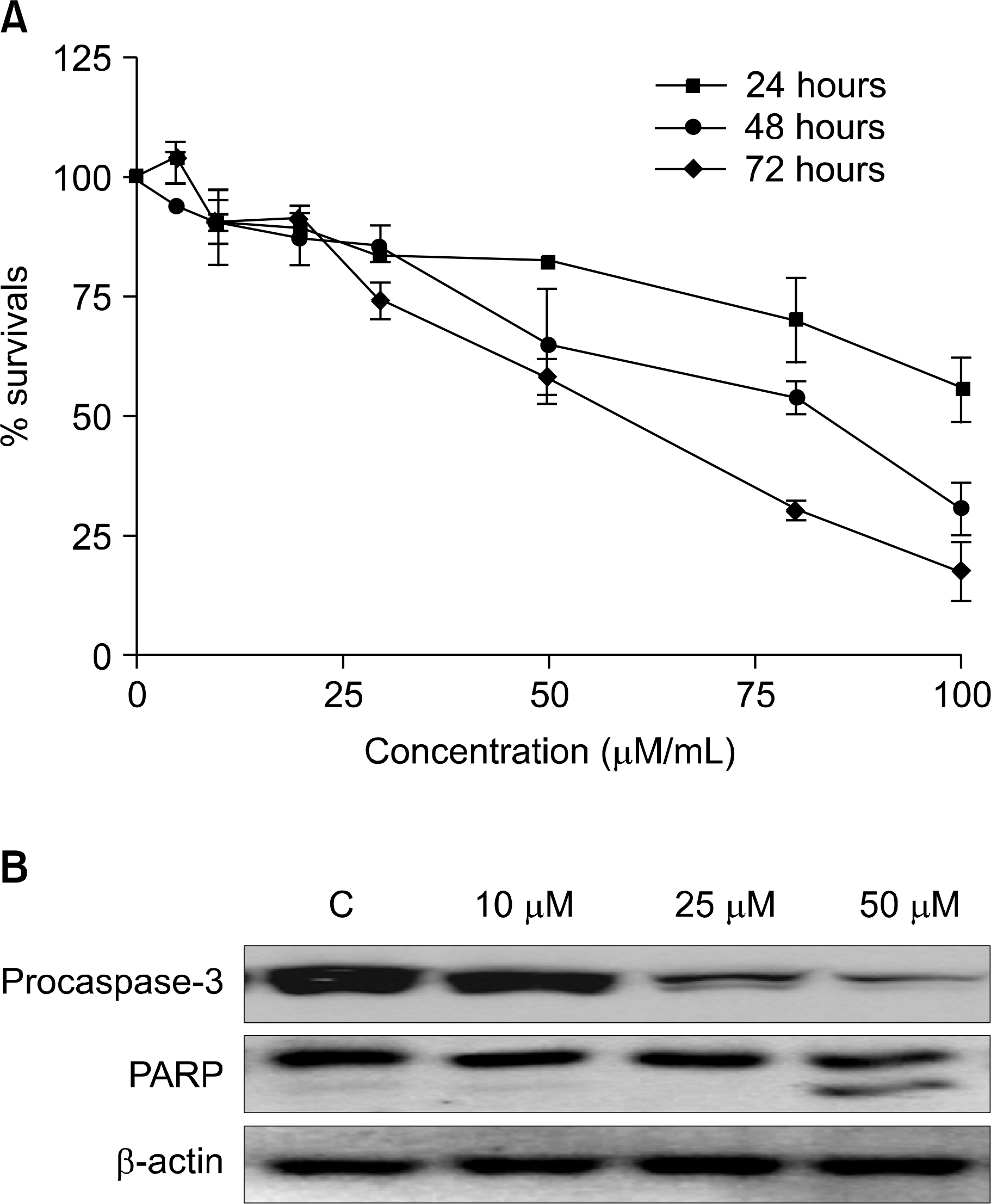
Fig. 2
Sub-G1 was induced by curcumin treatment via inactivation of cyclin D1 in U266 cells. Sub-G1 phase increased after treating U266 cells with curcumin 25μM for 24 hrs (A). U266 cell treated with indicated concentration of curcumin for 24 hrs. Curcumin treatment decreased expression of cyclinD1 and CDK4. Its treatment increased expression of p21 (B).
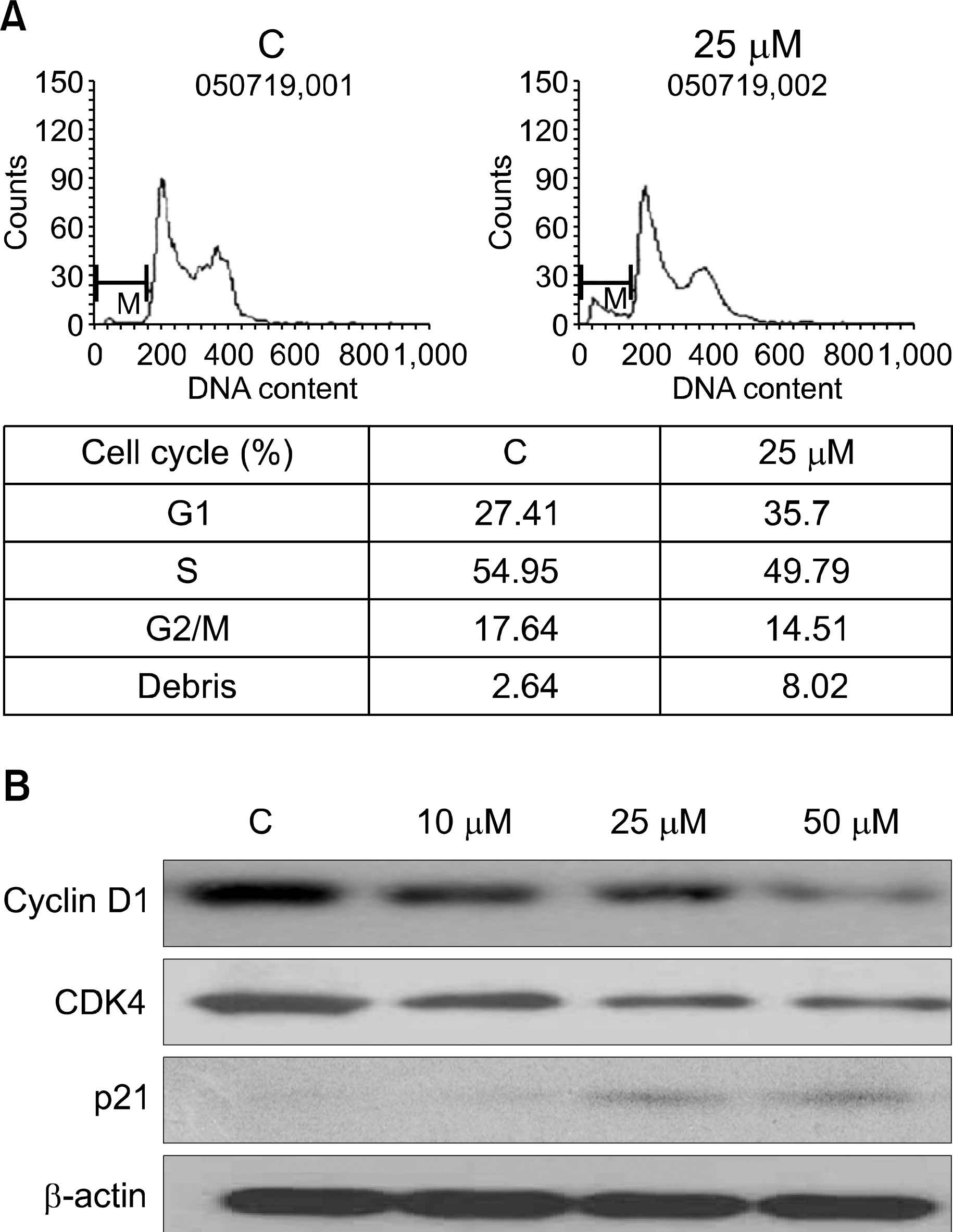
Fig. 3
Effect of curcumin including various signal transduction inhibitors on IL-6 mediated cell signal in U266 cells. U266 cells were incubated with serum free culture media (RPMI-1640) for 24 hours. U266 cell pretreated with various signal transduction inhibitors including curcumin for 1hr and then stimulated with 5ng human recombinant IL-6 for 15 min. 30μg of whole-cell extracts were analyzed by Western blot for phosphorylated STAT1, STAT3, total STAT1, STAT3, phosphorylated Erk, and total Erk (A). Cyclin D1, c-myc and β-actin (B). Lane: 1. control; 2. 5ng/mL IL-6; 3. 25μM curcumin, 5ng/mL IL-6; 4. 20μM LY294002, 5ng/mL IL-6; 5. 10 μM PD98059, 5ng/mL IL-6; 6. 20μM SP600125, 5ng/mL IL-6; 7. 20μM AG490, 5ng/mL IL-6; 8. 10μM SB203580, 5ng/mL IL-6; 9. 2μM 6-amino-4-quinazoline, 5ng/mL IL-6.
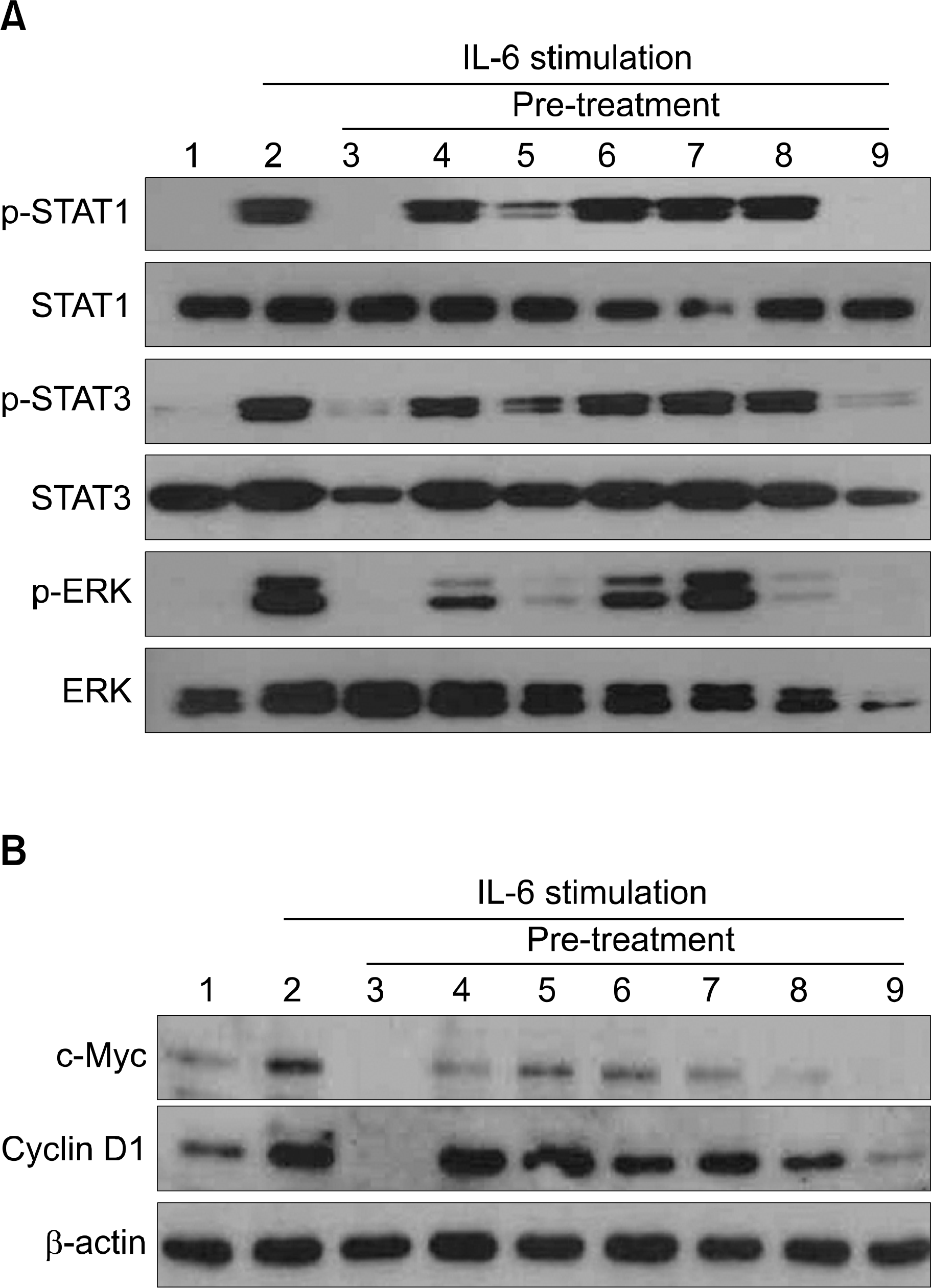
Fig. 4
Secretion of IL-6 and sIL-6R were inhibited by curcumin treatment. U266 cell treated with indicated concentration of curcumin for 24 hrs after culture in serum free media for 24 hrs. The secretion of IL-6 (A) and sIL-6R (B) was decreased at treated with curcumin.
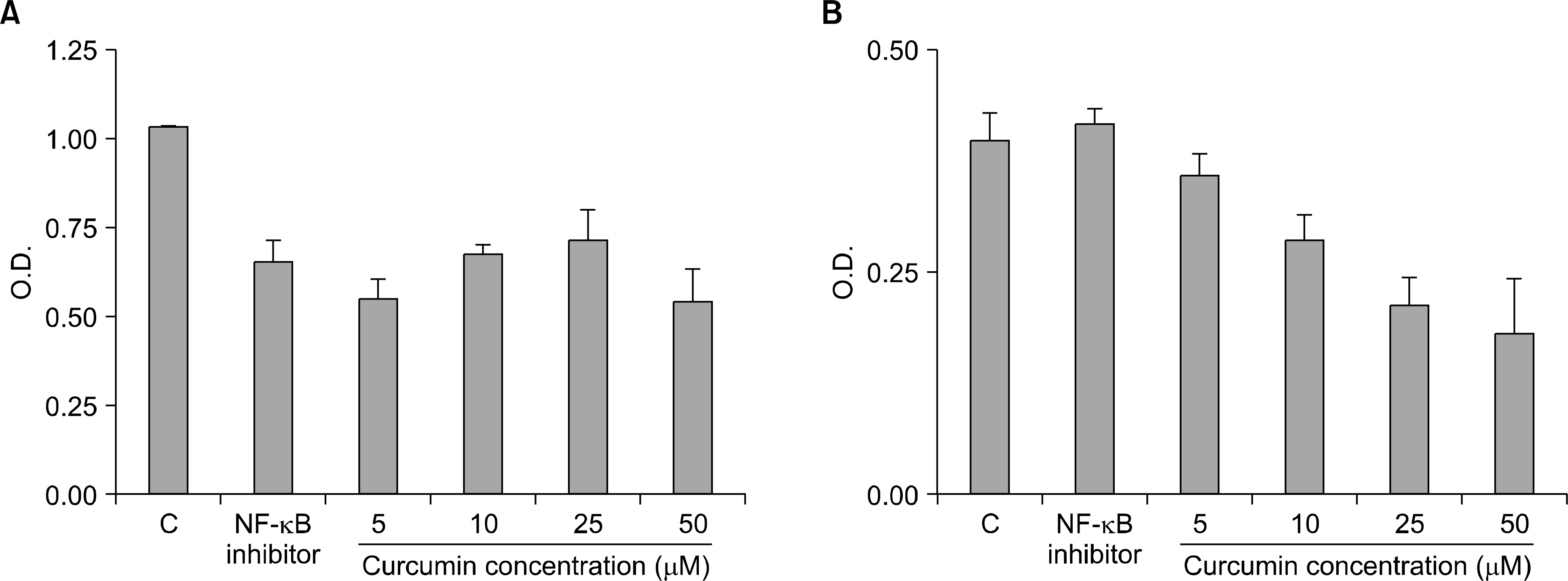
Fig. 5
Effect of curcumin in U266 animal model using NOD/SCID mouse. After U266 (5×105) cells were given via NOD/SCID mouse lateral tail vein, 25mg/kg was injected via intraperitoneum. 6 weeks later, development of bone lesions was examined using whole body micro CT imaging of NOD/SCID mouse. (A) Control, (B) Curcumin-injected SCID mouse.
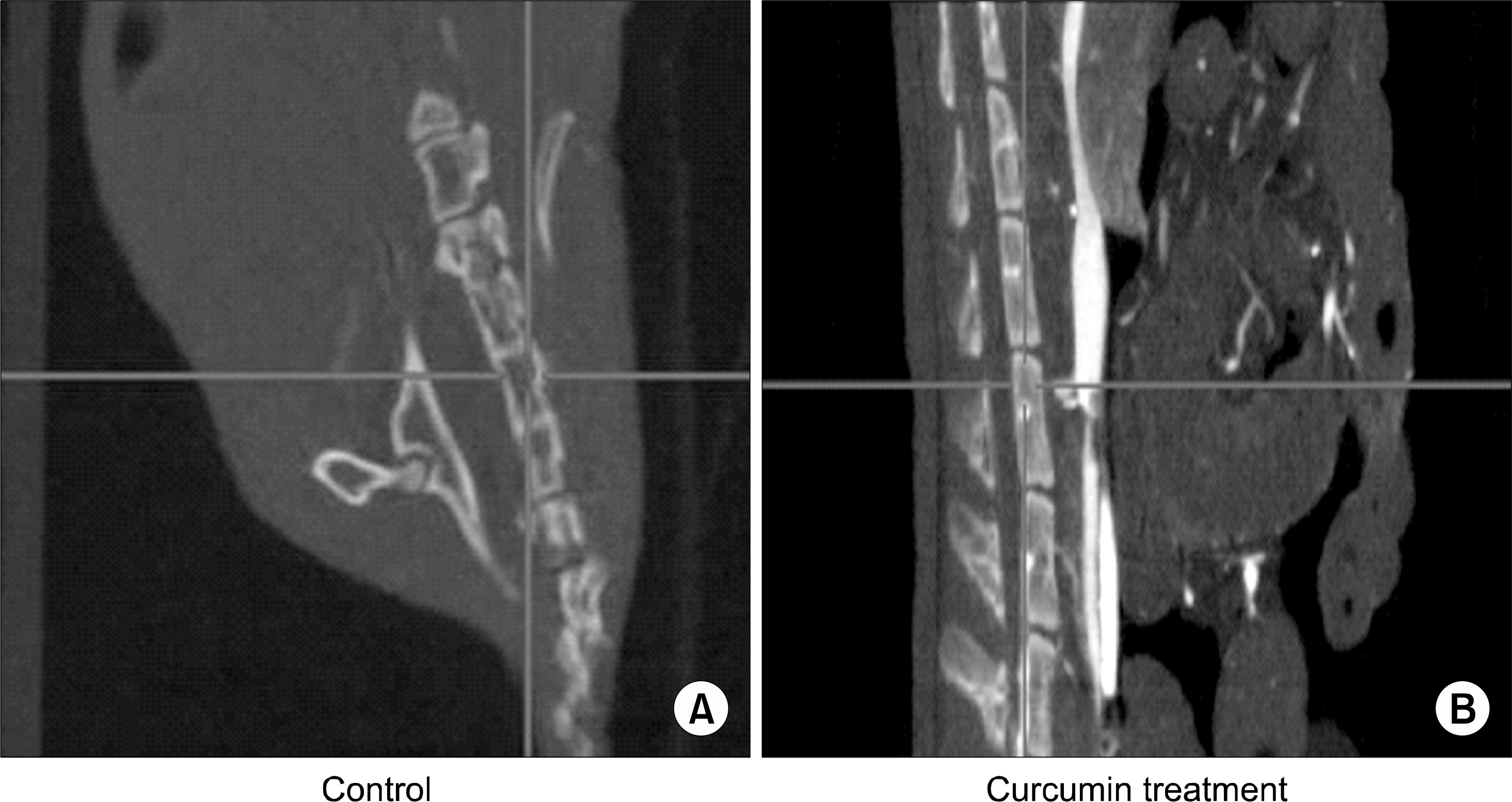
Fig. 6
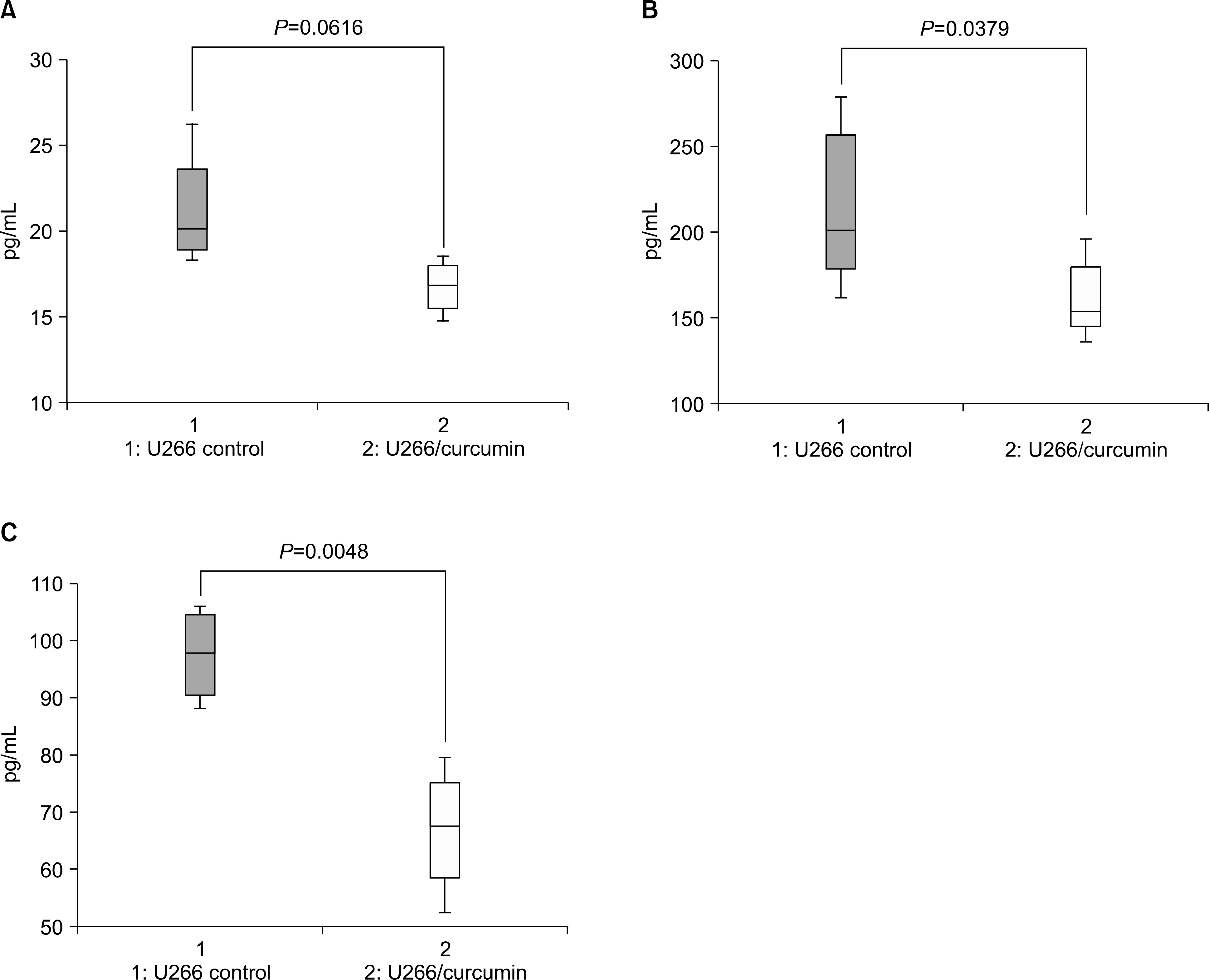
Secretion of IL-6 and sIL-6R were inhibited by curcumin treatment. After administration of 25mg/kg curcumin to U266 animal model for 6 weeks, IL-6, sIL-6R and IL-8 expression levels were examined using ELISA assay. The expression of IL-6 (A), sIL-6R (B) and IL-8 (C) in serum of administration of curcumin group was decreased with statically significance.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download