Abstract
Background:
The autosomal dominant giant platelet syndromes (GPS), characterized by triads of giant platelets, thrombocytopenia, and Döhle-like leukocyte inclusions are caused by MYH9 mutation, a gene encoding the nonmuscle myosin heavy chain-IIA. This study was aimed to identify the Korean GPS patients and to define clinical findings and molecular characteristics on them.
Methods:
After taking a family history, platelets were counted using hematologic autoanalyzer and peripheral blood smear (PBS) was examined for platelet size and number, and the presence of leukocyte inclusions. Mutation of MYH9 was studied from mononuclear cells from PB by direct sequencing of previously known 8 exons after PCR amplification of genomic DNA.
Results:
Twenty patients from 5 unrelated families were diagnosed as GPS. Giant platelets, greater than red cells on PBS, were found to be 3.1% of platelet (range, 1~11%). The median platelet count was 61,000/μL. Inclusion bodies were found in 3 families. Two families had previously reported mutations. Family I had Arg1944Ter in exon 40, located in the tail portion of myosin, while Family IV had Lys373Asn in exon 10, located in the proximal portion of myosin head. The mutations were found only in affected patients, but not in normal siblings or unrelated families.
Conclusion:
In this study, we identified several families with autosomal dominant GPS. Two families had known MYH9 mutations, Arg1944Ter and Lys373Asn. The search for unknown mutations in the remaining families as well as study of protein structural and functional alteration seems to be necessary for further delineation of these rare genetic disorders.
Go to : 
REFERENCES
1). Balduini CL., Iolascon A., Savoia A. Inherited throm-bocytopenias: from genes to therapy. Haematologica. 2002. 87:860–80.
2). Mhawech P., Saleem A. Inherited giant platelet disorders. Classification and literature review. Am J Clin Pathol. 2000. 113:176–90.
3). Kunishima S., Matsushita T., Kojima T, et al. Immunofluorescence analysis of neutrophil nonmuscle myosin heavy chain-A in MYH9 disorders: association of subcellular localization with MYH9 mutations. Lab Invest. 2003. 83:115–22.

4). Seri M., Cusano R., Gangarossa S, et al. Mutations in MYH9 result in the May-Heggllin anomaly, and Fechtner and Sebastian syndromes. The May-Hegglin/Fechtner Syndrome Consortium. Nat Genet. 2000. 26:103–5.
5). Peterson LC., Rao KV., Crosson JT., White JG. Fechtner syndrome: a variant of Alport's syndrome with leukocyte inclusions and macrothrombocytopenia. Blood. 1985. 65:397–406.
6). Epstein CJ., Sahud MA., Piel CF, et al. Hereditary macrothrombocytopathia, nephritis and deafness. Am J Med. 1972. 52:299–310.

7). Greinacher A., Nieuwenhuis HK., White JG. Sebastian platelet syndrome: a new variant of hereditary mac-rothrombocytopenia with leukocyte inclusions. Blut. 1990. 61:282–8.

8). Kelley MJ., Jawien W., Ortel TL., Korczak JF. Mutation of MYH9, encoding nonmuscle myosin heavy chain A, in May-Hegglin anomaly. Nat Genet. 2000. 26:106–8.

9). Kunishima S., Kojima T., Matsushita T, et al. Mutations in the NMMHC-A gene cause autosomal dominant macrothrombocytopenia with leukocyte inclusions (May-Hegglin anomaly/Sebastian syndrome). Blood. 2001. 97:1147–9.

10). Seri M., Savino M., Bordo D, et al. Epstein syndrome: another renal disorder with mutations in the no-nmuscle myosin heavy chain 9 gene. Hum Genet. 2002. 110:182–6.
11). Lalwani AK., Goldstein JA., Kelley MJ., Luxford W., Castelein CM., Mhatre AN. Human nonsyndromic hereditary deafness DFNA17 is due to a mutation in nonmuscle myosin MYH9. Am J Hum Genet. 2000. 67:1121–8.

12). Seri M., Pecci A., Di Bari F, et al. MYH9-related disease: May-Hegglin anomaly, Sebastian syndrome, Fechtner syndrome, and Epstein syndrome are not distinct entities but represent a variable expression of a single illness. Medicine (Baltimore). 2003. 82:203–15.
13). Lee AN., Nam CH., Kim BS., Song KS. A family case of May-Hegglin anomaly. Korean J Hematol. 1992. 27:195–9.
14). Lee HY., Seo JJ. Ultrastructural and functional study on the platelet of May-Hegglin anomaly. Korean J Pediatr Hematol-Oncol. 1995. 2:62–73.
15). Song KS., Choi JR., Song JW., Ha SK. A nonsense C5797T (R1933X) mutation of MYH9 gene in a family with May-Hegglin anomaly. Korean J Hematol. 2001. 36:253–6.
16). Baek HJ., Kook H., Byun HS, et al. MYH9-related disorder in a family (Autosomal dominant Epstein giant platelet syndrome). Korean J Pediatr Hematol-Oncol. 2003. 10:99–104.
17). Park JA., Kim EK., Park ES, et al. A case of Fechtner syndrome. The 54th annual fall meeting of Korean Pediatric Society. 2004. 258.
18). Gershoni-Baruch R., Baruch Y., Viener A., Lichtig C. Fechtner syndrome: clinical and genetic aspects. Am J Med Genet. 1988. 31:357–67.

19). Kunishima S., Kojima T., Tanaka T, et al. Mapping of a gene for May-Hegglin anomaly to chromosome 22q. Hum Genet. 1999. 105:379–83.

20). Kelley MJ., Jawien W., Lin A, et al. Autosomal dominant macrothrombocytopenia with leukocyte inclusions (May-Hegglin anomaly) is linked to chromosome 22q12-13. Hum Genet. 2000. 106:557–64.

21). Toren A., Rozenfeld-Granot G., Rocca B, et al. Autosomal-dominant giant platelet syndromes: a hint of the same genetic defect as in Fechtner syndrome owing to a similar genetic linkage to chromosome 22q11-13. Blood. 2000. 96:3447–51.

22). Marigo V., Nigro A., Pecci A, et al. Correlation between the clinical phenotype of MYH9-related disease and tissue distribution of class II nonmuscle myosin heavy chains. Genomics. 2004. 83:1125–33.
23). Simons M., Wang M., McBride OW, et al. Human nonmuscle myosin heavy chains are encoded by two genes located on different chromosomes. Circ Res. 1991. 69:530–9.

25). Strehler EE., Strehler-Page MA., Perriard JC., Peria-samy M., Nadal-Ginard B. Complete nucleotide and encoded amino acid sequence of a mammalian myosin heavy chain gene. Evidence against intron-dependent evolution of the rod. J Mol Biol. 1986. 190:291–317.
26). Kunishima S., Matsushita T., Kojima T, et al. Identification of six novel MYH9 mutations and genotype-phenotype relationships in autosomal dominant macrothrombocytopenia with leukocyte inclusions. J Hum Genet. 2001. 46:722–9.

27). Arrondel C., Vodovar N., Knebelmann B, et al. Expression of the nonmuscle myosin heavy chain IIA in the human kidney and screening for MYH9 mutations in Epstein and Fechtner syndromes. J Am Soc Nephrol. 2002. 13:65–74.

28). Heath KE., Campos-Barros A., Toren A, et al. Non-muscle myosin heavy chain IIA mutations define a spectrum of autosomal dominant macrothrombocyt-openias: May-Hegglin anomaly and Fechtner, Sebastian, Epstein, and Alport-like syndromes. Am J Hum Genet. 2001. 69:1033–45.

29). Ghiggeri GM., Caridi G., Magrini U, et al. Genetics, clinical and pathological features of glomerulonephritis associated with mutations of nonmuscle myosin IIA (Fechtner syndrome). Am J Kidney Dis. 2003. 41:95–104.
30). Martignetti JA., Heath KE., Harris J, et al. The gene for May-Hegglin anomaly localizes to a <1-Mb region on chromosome 22q12.3-13.1. Am J Hum Genet. 2000. 66:1449–54.

31). Hu A., Wang F., Sellers JR. Mutations in human non-muscle myosin IIA found in patients with May-Hegglin anomaly and Fechtner syndrome result in impaired enzymatic function. J Biol Chem. 2002. 277:46512–7.

32). Lodish H., Berk A., Matsudaira P, et al. Chap. 9: Molecular genetic techniques and genomics. In: Molecular cell biology. New York: W.H. Freeman and Company. 2004. 351–403.
33). Pecci A., Noris P., Invernizzi R, et al. Immunocytochemistry for the heavy chain of the non-muscle myosin IIA as a diagnostic tool for MYH9-related disorders. Br J Haematol. 2002. 117:164–7.
Go to : 
 | Fig. 1(A) Blood smear shows thrombocytopenia with giant platelets. (B) Döhle-like inclusion body in leukocyte in patient 3C (arrow) (Wright's Giemsa×1,000). |
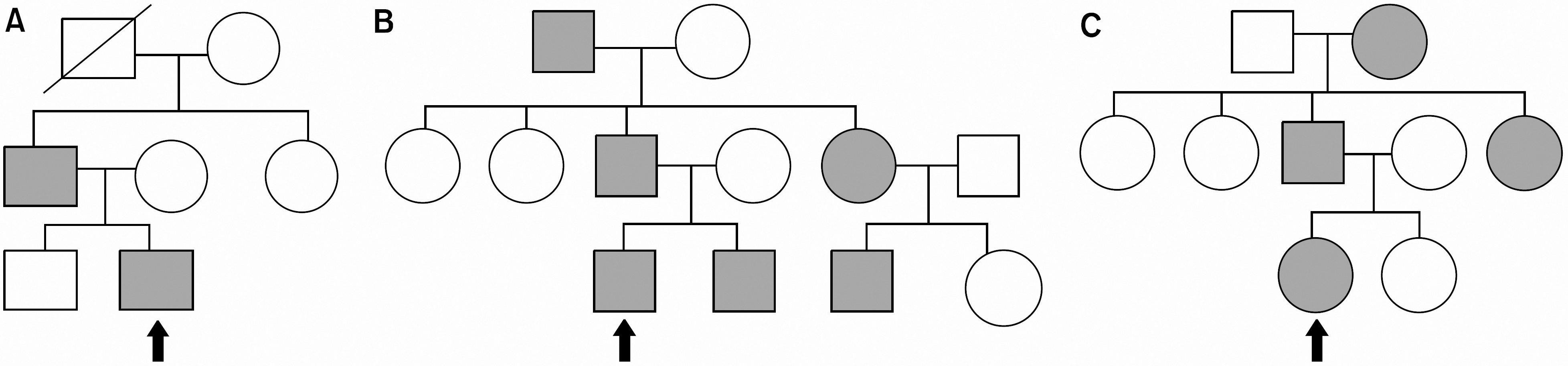 | Fig. 2Pedigrees of the Family II (A), Family III (B) and Family V (C). All the families show the autosomal dominant inheritance spanning 3 generations. |
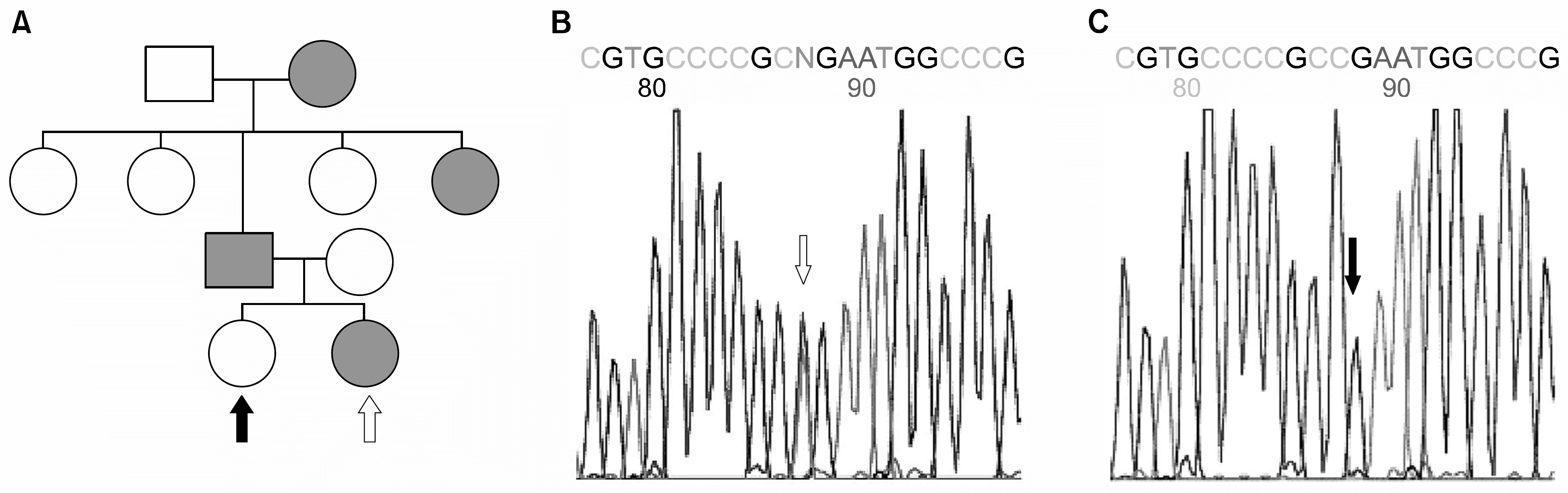 | Fig. 3Pedigree and electropherograms showing genetic alterations of MYH9 gene in the Family I. The candidate exons of MYH9 in the affected patients and in their normal siblings were subjected to genotyping after PCR amplification. (A) Pedgree shows the typical autosomal dominant trait of penetration. (B, C) Electropherogram showing the alteration of single nucleotide in the 40th exon in the proband white arrow in A & B), comparing with the normal sibling (black arrow in A & C). Note that equal amounts of thymidine (T) and cytidine (C) were reported as N, suggesting two different alleles are present. No other mutations were found in the patient. And this mutation was present only in this family. |
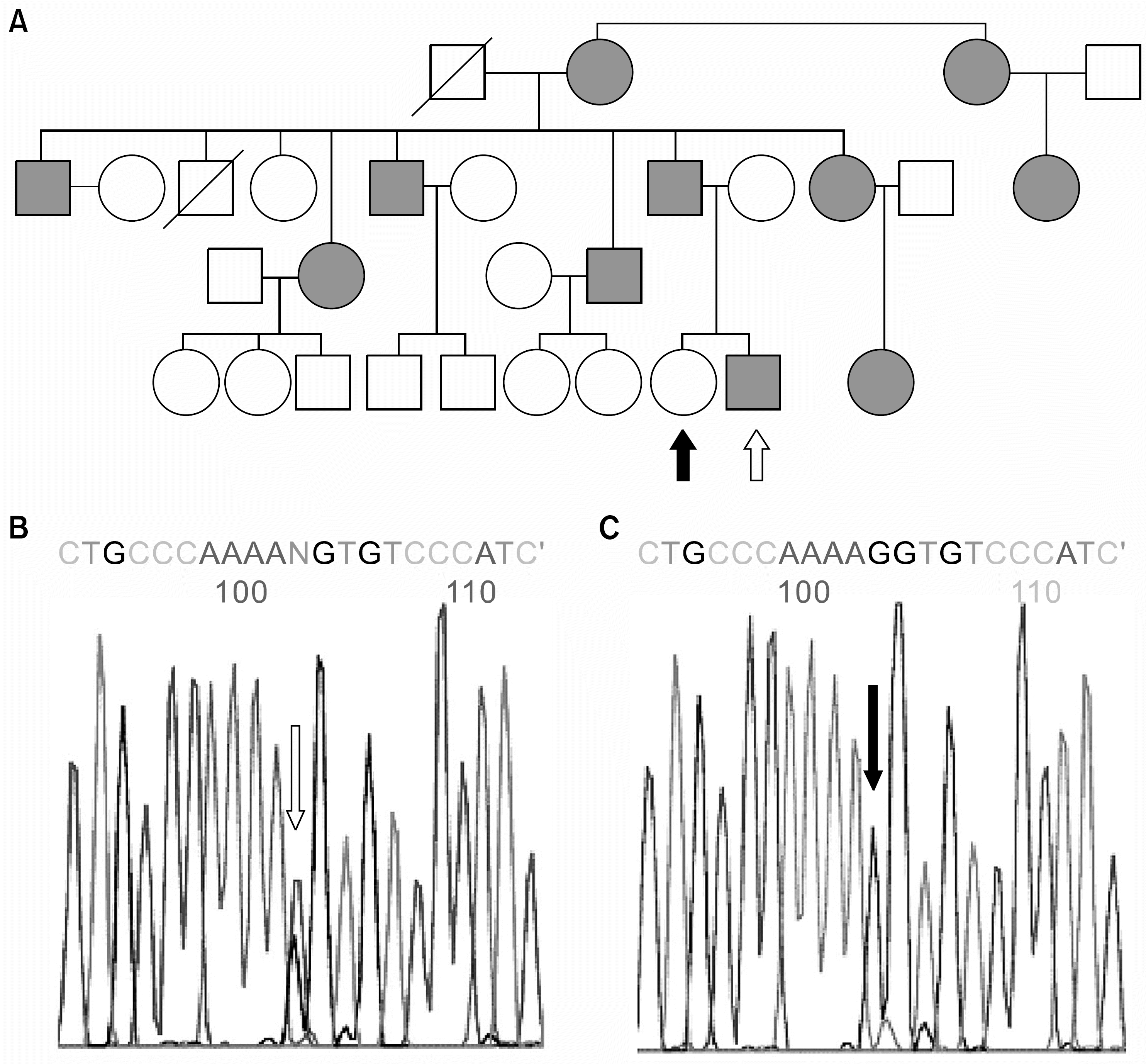 | Fig. 4Pedigree and electropherograms showing genetic alterations of MYH9 gene in the Family IV. The candidate exons of MYH9 in the affected patients and in their normal siblings were subjected to genotyping after PCR amplification. (A) Pedgree shows the typical autosomal dominant trait of penetration. (B, C) Electropherogram showing the alteration of single nucleotide in the 10th exon in the proband (white arrow in A & B), comparing with the normal sibling (black arrow in A & C). Genotyping of 10th exon shows the patient has two different alleles of cytidine (C) and guanosine (G). |
 | Fig. 5The linear protein structure was obtained from pfam (www.sanger.ac.uk) and the relative location of the mutations was pointed out. One mutation from the Family IV was located in the head portion of the myosin, while that of the Family I was at the tail portion of the protein. |
Table 1.
Discriminating features of MYH9-related disorders (Modified from ref. 1)
∗Ultrastructure of leukocyte inclusion: type 1, clusters of ribosomes aligned along parallel filaments; type 2, dispersed filaments and randomly distributed ribosomes. Abbreviations: MHA, May-Hegglin anomaly; SBS, Sebastian syndrome; FTNS, Fechtner syndrome; EPS, Epstein syndrome; DFNA17, nonsyndromic autosomal domonant form of deafness.
Table 2.
Primer set for exon amplifications
Table 3.
Clinical and hematologic findings of patients with Giant Platelet syndromes




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download