Abstract
Background
Transforming growth factor-β1 (TGF-β1) is known to be a potent growth inhibitor of many cell types, including most epithelial cells. However, the mechanism of TGF-β1 action on cell growth in lymphomas and leukemia still remains to be elucidated. c-myc is a central regulator of cell proliferation and apoptosis, and telomerase is believed to play an important role in carcinogenesis. The aim of the study was to determine the effects of cell growth, c-myc gene expression and telomerase activity due to TGF-β1 and examine its mechanism of action in lymphomas and leukemia.
Methods
The cell growths of Jiyoye (Burkitt lymphoma), H9 (T cell lymphoma), and CCRF-CEM (acute lymphocytic leukemia, T cell) cell lines due to TGF-β1 were measured using the MTT assay. RT-PCR was also performed to monitor the expression of the c-myc gene in these cells with the telomerase activity measured using a TRAP assay.
Results
There was significant inhibition of cell growth in TGF-β1 (5ng/mL) treated Jiyoye cells. When treated with TGF-β1, the Jiyoye cells exhibited marked decreases in the levels of c-myc RNA and telomerase activity. However, TGF-β1 treated H9 and CCRF-CEM cells showed no cell growth inhibition or reductions in the levels of c-myc mRNA and telomerase activity. The effect of TGF-β1 on cell growth was noted to closely correlate with c-myc mRNA expression and telomerase activity.
Go to : 
REFERENCES
1). Cheifetz S, Hernandez H, Laiho M, ten Dijke P, Iwata KK, Massague J. Distinct transforming growth factor-beta (TGF-beta) receptor subsets as determinants of cellular responsiveness to three TGF-beta isoforms. J Biol Chem. 1990; 265:20533–8.

2). Blobe GC, Schiemann WP, Lodish HF. Role of transforming growth factor beta in human disease. N Engl J Med. 2000; 342:1350–8.
3). Claassen GF, Hann SR. A role for transcriptional repression of p21CIP1 by c-Myc in overcoming transforming growth factor-beta induced cell cycle arrest. Proc Natl Acad Sci USA. 2000; 97:9498–503.
4). Wolfraim LA, Fernandez TM, Mamura M, et al. Loss of Smad3 in acute T-cell lymphoblastic leukemia. N Engl J Med. 2004; 351:552–9.

5). Downing JR. TGF-beta signaling, tumor suppression, and acute lymphoblastic leukemia. N Engl J Med. 2004; 351:528–30.
6). Abba MC, Laguens RM, Dulout FN, Golijow CD. The c-myc activation in cervical carcinomas and HPV 16 infections. Mutat Res. 2004; 557:151–8.

7). Blancato J, Singh B, Liu A, Liao DJ, Dickson RB. Correlation of amplification and overexpression of the c-myc oncogene in high‐grade breast cancer: FISH, in situ hybridisation and immunohistochemical analyses. Br J Cancer. 2004; 90:1612–9.

8). Wu KJ, Grandori C, Amacker M, et al. Direct activation of TERT transcription by c-MYC. Nat Genet. 1999; 21:220–4.

9). Lundblad V. DNA ends: maintenance of chromosome termini versus repair of double strand breaks. Mutat Res. 2000; 2:227–40.

11). Autexier C, Greider CW. Telomerase and cancer: revisiting the telomere hypothesis. Trends Biochem Sci. 1996; 21:387–91.

13). Derynck R, Akhurst RJ, Balmain A. TGF-beta signaling in tumor suppression and cancer progression. Nat Genet. 2001; 2:117–29.
14). Massague J, Blain SW, Lo RS. TGF beta signaling in growth control, cancer, and heritable disorders. Cell. 2000; 103:295–309.
15). Massague J, Attisano L, Wrana JL. The TGF-beta family and its composite receptors. Trends Cell Biol. 1994; 4:172–8.
16). Amati B. Integrating Myc and TGF-beta signalling in cell cycle control. Nat Cell Biol. 2001; 3:112–3.
17). Pietenpol JA, Holt JT, Stein RW, Moses HL. Transforming growth factor beta 1 suppression of c-myc gene transcription: role in inhibition of keratinocyte proliferation. Proc Natl Acad Sci USA. 1990; 87:3758–62.

18). Djaborkhel R, Tvrdik D, Eckschlager T, Raska I, Muller J. Cyclin A down-regulation in TGFbeta1-arrested follicular lymphoma cells. Exp Cell Res. 2000; 261:250–9.
19). Inman GJ, Allday MJ. Apoptosis induced by TGF-beta 1 in Burkitt's lymphoma cells is caspase 8 dependent but is death receptor independent. J Immunol. 2000; 165:2500–10.
21). Koo SH, Kwon KC, Shin SY, et al. Genetic alterations of gastric cancer: comparative genomic hybridization and fluorescence in situ hybridization studies. Cancer Genet Cytogenet. 2000; 2:97–103.
22). Zajac-Kaye M. Myc oncogene: a key component in cell cycle regulation and its implication for lung cancer. Lung Cancer. 2001; 2:S43–6.

23). Abba MC, Laguens RM, Dulout FN, Golijow CD. The c-myc activation in cervical carcinomas and HPV 16 infections. Mutat Res. 2004; 557:151–8.

24). Augenlicht LH, Wadler S, Corner G, et al. Low level c-myc amplification in human colonic carcinoma cell lines and tumors: a frequent, p53 independent mutation associated with improved outcome in a randomized multi‐institutional trial. Cancer Res. 1997; 57:1769–75.
25). Mathew S, Lorsbach RB, Shearer P, Sandlund JT, Raimondi SC. Double minute chromosomes and c-MYC amplification in a child with secondary myelodysplastic syndrome after treatment for acute lymphoblastic leukemia. Leukemia. 2000; 14:1314–5.

26). Mulder KM, Brattain MG. Effects of growth stimulatory factors on mitogenicity and c-myc expression in poorly differentiated and well differentiated human colon carcinoma cells. Mol Endocrinol. 1989; 3:1215–22.
27). Rama S, Suresh Y, Rao AJ. TGF-beta1 induces multiple independent signals to regulate human trophoblastic differentiation: mechanistic insights. Mol Cell Endocrinol. 2003; 206:123–36.
28). Chen CR, Kang Y, Massague J. Defective repression of c-myc in breast cancer cells: a loss at the core of the transforming growth factor beta growth arrest program. Proc Natl Acad Sci USA. 2001; 98:992–9.
29). Terasawa K, Sagae S, Takeda T, Ishioka S, Kobayashi K, Kudo R. Telomerase activity in malignant ovarian tumors with deregulation of cell cycle regulatory proteins. Cancer Lett. 1999; 142:207–17.

30). Huang GT, Lee HS, Chen CH, et al. Telomerase activity and telomere length in human hepatocellular carcinoma. Eur J Cancer. 1998; 34:1946–9.

31). Kumaki F, Kawai T, Hiroi S, et al. Telomerase activity and expression of human telomerase RNA component and human telomerase reverse transcriptase in lung carcinomas. Hum Pathol. 2001; 32:188–95.

32). Kim MY, Lee JN. Pattern of telomerase activities in acute leukemias and chronic myeloid leukemia. Korean J Hematol. 2001; 36:189–96.
Go to : 
 | Fig. 1.Effect of TGF-β1 treatment on J iyoye cell growth. (A) C ontrol cell after 24 hours, (B) 24 hours after treatment of TGF-β1. (C) Control cell after 48 hours, (D) 48 hours after treatment of TGF-β1. (E) Control cell after 72 hours, (F) 72 hours after treatment of TGF-β1. |
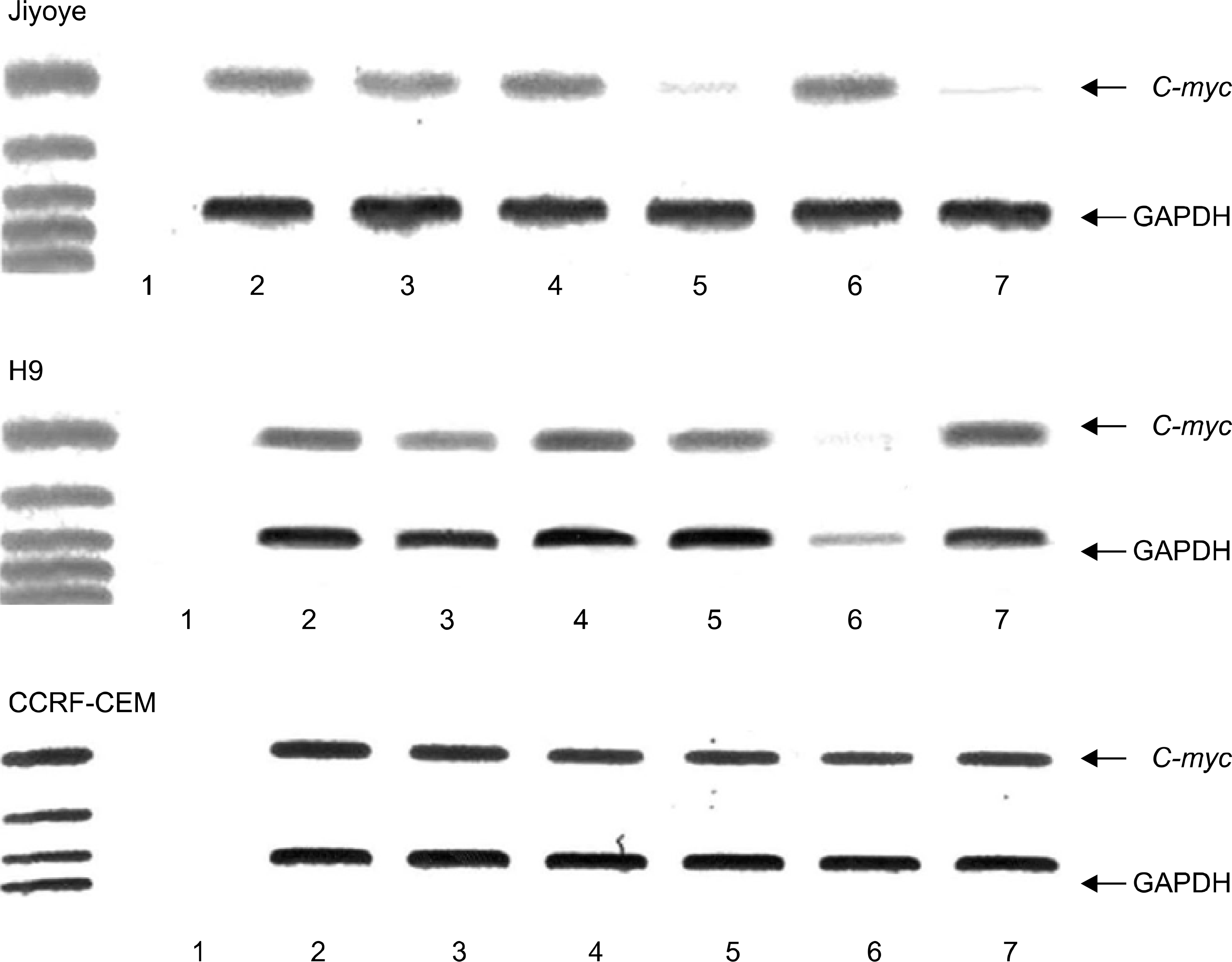 | Fig. 2.Effect of TGF-β1 treatment on c-myc RNA expression in J iyoye cell line, H9 cell line, and C C RF-C EM cell line. 1, negative control; 2, control after 24 hours; 3, 24 hours after treatment of TGF-β1; 4, control after 48 hours; 5, 48 hours after treatment of TGF-β1; 6, control after 72 hours; 7, 72 hours after treatment of TGF-β1. |
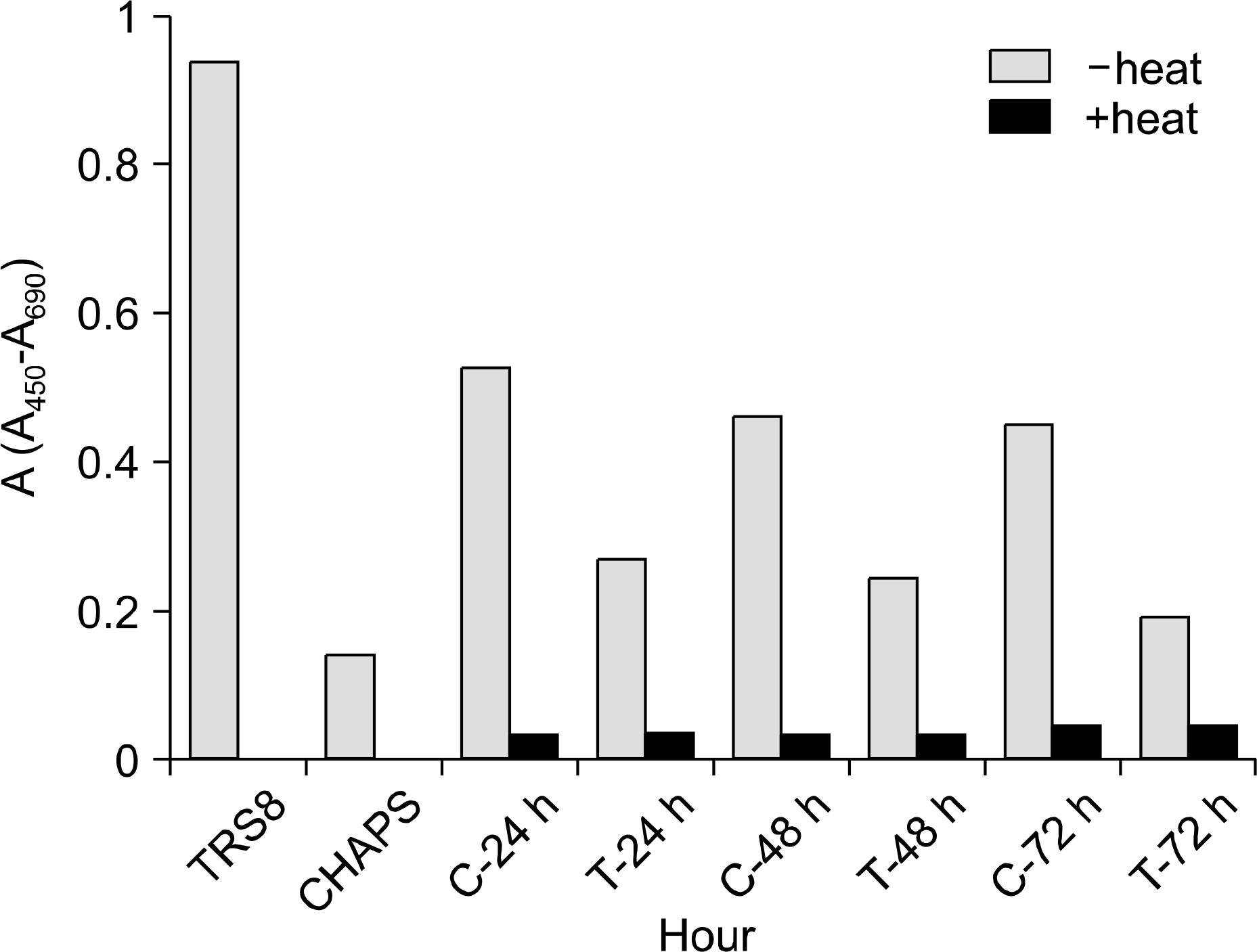 | Fig. 3.Absorbance units (A450-A690) of J iyoye cell line for telomerase activity. TRS8, positive control; CHAPS, negative control; C, control; T, TGF-β1 treated. |
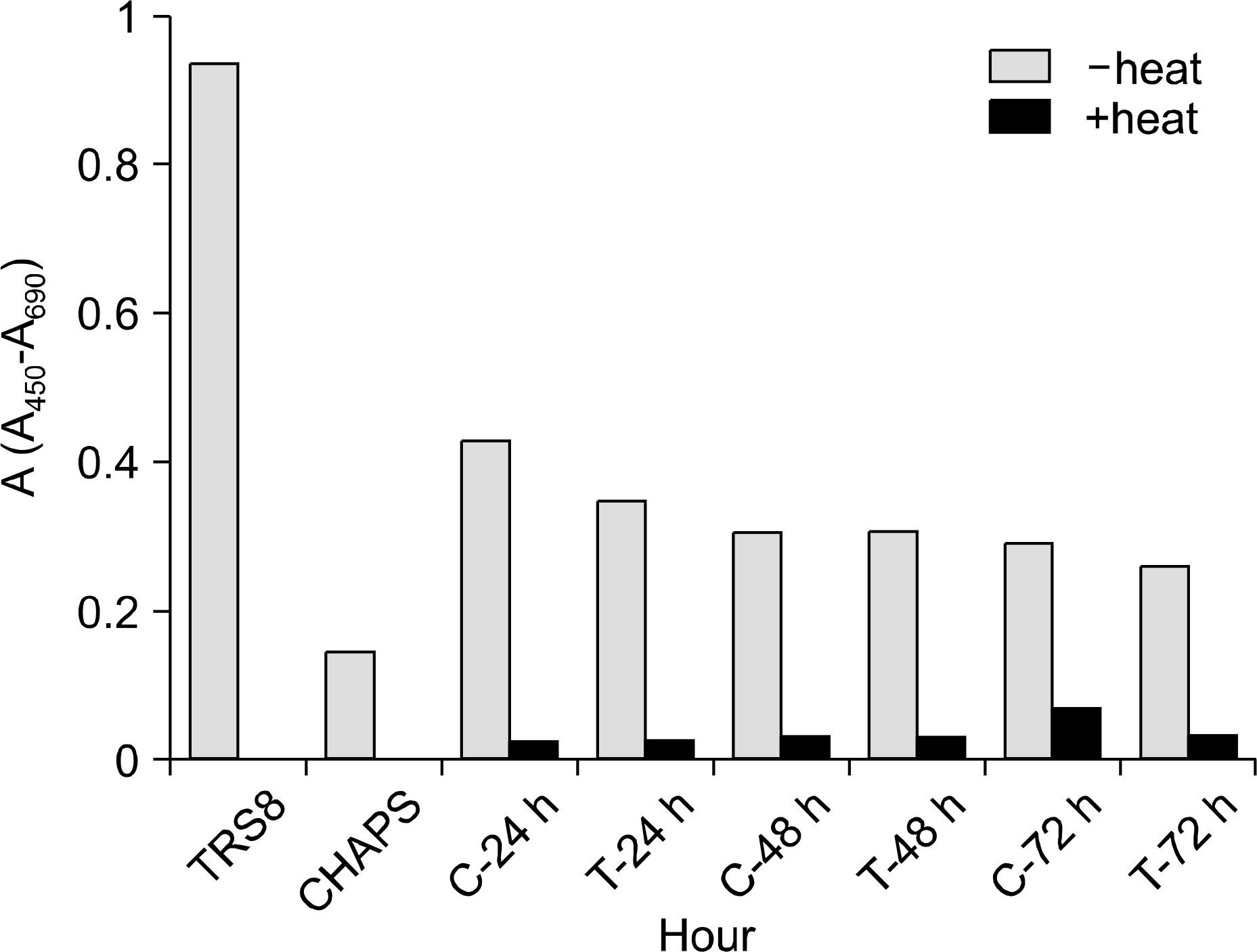 | Fig. 4.Absorbance units (A450-A690) of H9 cell line for telomerase activity. TRS8, positive control; CHAPS, negative control; C, control; T, TGF-β1 treated. |
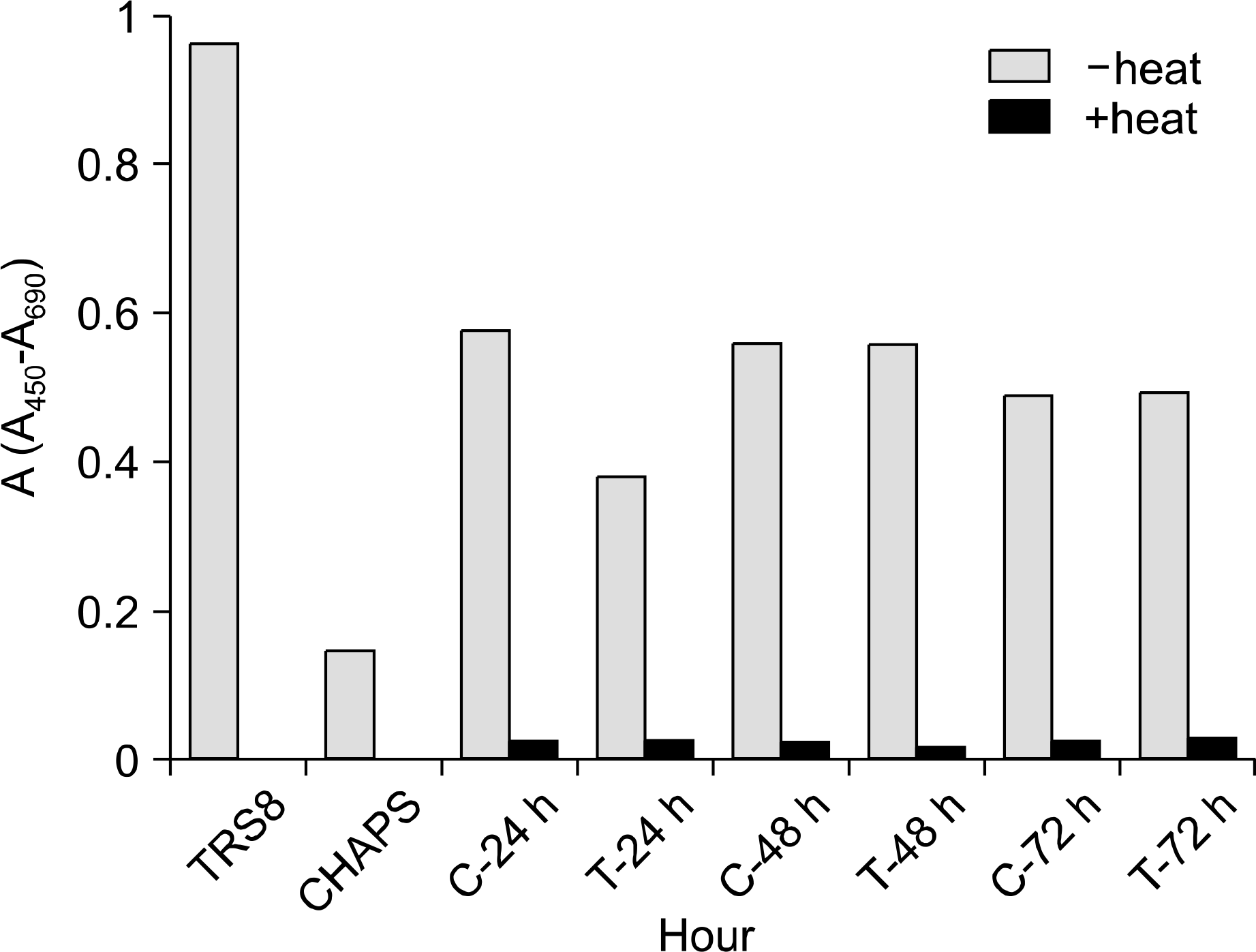 | Fig. 5.Absorbance units (A450-A690) of C C RF-C EM cell line for telomerase activity. TRS8, positive control; CHAPS, negative control; C, control; T, TGF-β1 treated. |
Table 1.
Effect on cell growth by TGF-β1 in J iyoye, H9 and C CRF-C EM cell lines
| Cell survival ratio (%) | P value | |||
|---|---|---|---|---|
| 24 hours | 48 hours 7 | 72 hours | ||
| Control | 100 | 100 | 100 | |
| J iyoye | 88.5 | 70.5 | 56.4 | <0.005 |
| H9 | 73.4 | 85.1 | 93.4 | |
| CC RF-CEM | 72.9 | 100 | 114.1 | |
Table 2.
Telomerase activity in J iyoye, H9 and CC RF-CEM cell lines
| Telomerase activity (ΔA∗) | ||||||
|---|---|---|---|---|---|---|
| 24 hours | 48 hours | 72 hours | ||||
| C ontrol | TGF-β1 | C ontrol | TGF-β1 | C ontrol | TGF-β1 | |
| J iyoye | + (0.499) | + (0.230) | + (0.428) | + (0.211) | + (0.404) | – (0.146) |
| H9 | + (0.399) | + (0.324) | + (0.296) | + (0.272) | + (0.222) | + (0.227) |
| C C RF-C EM | + (0.556) | + (0.360) | + (0.554) | + (0.541) | + (0.467) | + (0.467) |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download