Abstract
Purpose
The aim of this study was to determine whether stratification of deceased donors by the United Network for Organ Sharing (UNOS) criteria negatively impacts graft survival.
Materials and Methods
We retrospectively reviewed deceased donor and recipient pretransplant variables of kidney transplantations that occurred between February 1995 and December 2009. We compared clinical outcomes between standard criteria donors (SCDs) and expanded criteria donors (ECDs).
Results
The deceased donors consisted of 369 patients. A total of 494 transplant recipients were enrolled in this study. Mean age was 41.7±11.4 year (range 18–69) and 273 patients (55.4%) were male. Mean duration of follow-up was 8.8±4.9 years. The recipients from ECD kidneys were 63 patients (12.8%). The overall mean cold ischemia time was 5.7±3.2 hours. Estimated glomerular filtration rate at 1, 2, and 3 years after transplantation were significantly lower in ECD transplants (1 year, 62.2±17.6 vs. 51.0±16.4, p<0.001; 2 year, 62.2±17.6 vs. 51.0±16.4, p=0.001; 3 year, 60.9±23.5 vs. 54.1±18.7, p=0.047). In multivariate analysis, donor age (≥40 years) was an independent risk factor for graft failure. In Kaplan-Meier analyses, there was no significant difference in death-censored graft survival (Log rank test, p>0.05), although patient survival was lower in ECDs than SCDs (Log rank test, p=0.011).
A rapid growing incidence in chronic kidney disease has led to an increasing number of patients awaiting kidney transplantation, despite a limited supply of organs available for donation. 123 The concept of expanded criteria donors (ECDs) for kidney transplantation was proposed as an alternative strategy to increase the donor pool. On the basis of a study by Port, et al.,4 the United Network for Organ Sharing (UNOS) introduced policy to address allocation issues and to promote use of kidney from ECDs.5 However, despite increases in available kidneys from ECDs for transplantation, organ shortages remain a major issue. Meanwhile, with increasing numbers of elderly donors with comorbidities, recent studies suggest that clinical outcomes of kidney transplantation using ECDs, compared with standard criteria donors (SCDs), have been improved.67 Accordingly, the definition of ECD needs to be re-evaluated.
The objective of this study was to evaluate whether stratifications of ECD have had a negative impact on graft survival in deceased donor kidney transplantations.
We conducted a retrospective analysis of all patients who underwent deceased donor kidney transplantation at Samsung Medical Center and Seoul National University Hospital between February 1995 and December 2009. Exclusion criteria included pediatric recipients (younger than age 18 years), multi-organ transplants, and re-transplantations.
The clinical outcomes of recipients from ECDs were compared with recipients from SCDs. We defined ECD as any donor aged ≥60 years or any donor aged 50–59 years matching two of the following three criteria: cerebrovascular accident (CVA) death, terminal creatinine >1.5 mg/dL, or history of hypertension. 4 SCD was defined as any deceased donor not meeting the above criteria. To evaluate the relationship between donor serum creatinine (sCr) levels and graft survival, we reviewed medical records for initial values at the start of management as potential organ donor in the intensive care unit, peak values, and terminal values.
Graft failure was defined as return to permanent dialysis and transplant nephrectomy after kidney transplantation. Transplant recipient death with functioning was not considered as graft failure. Delayed graft function was defined as the need for at least one hemodialysis within the first week posttransplant. Estimated glomerular filtration rate (eGFR) was calculated using the four variable modification of diet in renal disease (MDRD) equations.
Continuous variables are described as means±standard deviations and ranges. Differences between groups were compared with the t-test or chi-square test. The time to graft failure and patient death was determined using the Kaplan-Meier method, and log-rank test was used to compare groups. To identify factors associated with graft failure, we used Cox regression analysis. All p-values <0.05 were considered statistically significant.
Baseline characteristics of the deceased donors are shown in Table 1. Mean age of donors was 37.4±15.0 years. Seventy-three (19.1%) patients had a history of hypertension. The primary cause of brain death was CVA (n=170, 46.6%). The overall mean cold ischemia time (CIT) was 5.7±3.2 hours (range 1.7–18.2 hours). During donor management, acute kidney injury defined by clinical practice guidelines8 occurred in 77 patients (14.5%).
A total of 493 recipients from 365 deceased donors were categorized as SCD (n=430, 90.5%) and ECD (n=63, 11.5%). Mean duration of follow-up was 8.8±4.9 year and 53 patients (45 SCD, 10.5% and 8 ECD, 12.7%) were lost to follow-up. The characteristics of the recipients and posttransplant outcomes are outlined in Table 2. Mean CIT was similar in both groups (SCD 5.7±3.2 hours vs. ECD 5.4±3.1 hours, p>0.05). There were significant differences in recipient age (SCD 40.7±11.1 years vs. ECD 48.7±10.6 years, p<0.001) and sex distribution (male, SCD 53.5% vs. ECD 68.3%, p<0.05). The mean eGFR within 3 years posttransplant was significantly higher among recipients from SCDs, compared with recipients from ECDs (1 year, 62.2±17.6 vs. 51.0±16.4, p<0.001; 2 year, 62.3±18.2 vs. 53.9±13.5, p=0.001; 3 year, 60.9±23.5 vs. 54.1±18.7, p=0.047). However, there were no significant differences in the mean eGFR at 4 years and 5 years posttransplant. The incidences of delayed graft function and acute rejection at 1 year between recipients from SCDs and ECDs were not significantly different.
In univariate analysis, risk factors for graft failure were donor age (≥40 year), CVA, history of hypertension, and CIT (≥7.5 hours). Among these, donor age (≥40 years) was associated with significantly increased risk for graft failure in multivariate analysis (hazard ratio 2.202, p=0.009). In analyses of associations between donor sCr (initial, peak, and terminal values) with graft survival, we were unable to determine a cut-off value of negative impact for graft failure (Table 3).
The overall graft and patient survivals are shown in Figs. 1 and 2. There was no significant difference in graft survival between recipients from SCDs and ECDs (Log-rank p>0.05). However, patient survival was inferior among recipients from ECDs, compared with recipients from SCDs (Log-rank p=0.011).
To overcome critical organ shortages in kidney transplantation, it is important to expand the pool of available deceased donor. Studies that have assessed the impact of ECD kidneys on deceased donor transplantations suggest that allocation strategy using the concept of ECD is associated with an expansion in kidney transplantation.910 Despite efforts to effectively reduce discard rates of organs, many patients are still waiting for a suitable organ.11
Recently, there have been increases in ECD kidney recipients, especially from elderly or acute kidney injury accompanying with elevation of sCr. However, some authors have reported that clinical outcomes of recipients from ECD kidneys are comparable to those of recipients from SCD kidneys.1213 Therefore, we considered that the indices of ECD kidneys need to be revised according to regional differences for expansion of deceased donor pools.
In our analysis, we observed that graft survival was similar in recipients from SCDs and those from ECDs, although the recipients from ECDs showed a poor graft function within 3 years after transplantation. In addition, the incidence of DGF and acute rejection at 1 year posttransplant were not inferior in ECD recipients. These results are in agreement with those of other studies.1415
Four donor factors, including older age (≥40 years), sCr (≥1.5 mg/dL), history of hypertension, and CVA, have been shown to be associated with a significant increase risk for graft failure and identified as indices of ECDs.4 However, in our study, donor age (40 years or older) was the only independent risk factor of graft failure in Cox regression. While our study focused whether donor sCr levels have an influence on graft survival, no association was found between donor sCr levels at various time points and graft survival. Several studies have documented that the overall results of donors with high terminal sCr, especially accompanying acute kidney injury, are comparable with SCD transplants.16171819 These findings suggest that sophisticated indices to predict long-term outcomes are required to improve the stratification of ECD kidneys. It is necessary to avoid oversimplification of ECDs because of not represent the reality.20
Prolonged CITs and ischemia-reperfusion injury result in inflammation and oxidative damage on endothelial cells and tubular epithelial cells in procured kidney. These changes are potential causes of graft rejection by triggering alloimmune reactivity. 2122 Previous reports have demonstrated that increased CITs are associated with a significant risk of DGF and graft failure in deceased donor kidney transplantation.232425 However, there is a controversy as to how long CIT is associated with graft failure in kidney transplantation. According to a report by Opelz and Döhler26 increasing ischemia up to 18 hours is associated with graft survival. In a cohort study of 778 kidney transplantation, CIT of >20 hour significantly reduced 5 year graft survival. 27 In addition, analysis of 3839 recipients in a French cohort showed proportional increased risk of graft failure for each additional hour of CIT.25 In contrast, CIT was not associated with graft survival in our population. We presume that CIT (mean 5.7 hours), compared to other studies, was short because Korea is geographically small.
This study has some limitations. The small number of recipients, especially ECD transplants, could be considered insufficient to detect statistical significance for risk factors of graft failure. In addition, it was possible that posttransplant practices at each tertiary referral center differed, because of the retrospective observational nature of the study. Furthermore, our study was carried out in relatively unrepresentative settings, and we did not analyze national registry data.
In conclusion, ECD kidneys according to UNOS criteria provide unsatisfactory information on graft survival, compared with SCDs. Donors with high creatinine at various time points were not risk factor for graft failure. Modification of criteria considering regional differences for standard/expanded donor is needed to expand donor pools.
Figures and Tables
 | Fig. 1Death-censored graft survival according to donor type. ECD, expanded criteria donor; SCD, standard criteria donor. |
 | Fig. 2Patient survival according to donor type. ECD, expanded criteria donor; SCD, standard criteria donor. |
Table 1
Donor Baseline Demographics and Characteristics (n=365)

Table 2
Recipient Demographics and Outcomes (n=493)
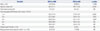
Table 3
Risk Factors for Graft Failure
References
1. Noordzij M, Kramer A, Abad Diez JM, Alonso de la Torre R, Arcos Fuster E, Bikbov BT, et al. Renal replacement therapy in Europe: a summary of the 2011 ERA-EDTA registry annual report. Clin Kidney J. 2014; 7:227–238.

2. Mathur AK, Ashby VB, Sands RL, Wolfe RA. Geographic variation in end-stage renal disease incidence and access to deceased donor kidney transplantation. Am J Transplant. 2010; 10(4 Pt 2):1069–1080.

3. Cusumano AM, Romao JE, Poblete Badal H, Elgueta Miranda S, Gomez R, Cerdas Calderon M, et al. [Latin-American dialysis and kidney transplantation registry: data on the treatment of end-stage renal disease in Latin America]. G Ital Nefrol. 2008; 25:547–553.
4. Port FK, Bragg-Gresham JL, Metzger RA, Dykstra DM, Gillespie BW, Young EW, et al. Donor characteristics associated with reduced graft survival: an approach to expanding the pool of kidney donors. Transplantation. 2002; 74:1281–1286.

5. United Network for Organ Sharing. Organ procurement and transplantation network policies. Policy 8: allocation of kidneys. 2016 March 1. Available at:http://optn.transplant.hrsa.gov/governance/policies.
6. Kim BS, Joo SH, Ahn HJ, Choi JH, Lee SH, Park HC. Outcomes of expanded-criteria deceased donor kidney transplantation in a single center. Transplant Proc. 2014; 46:1067–1070.

7. Lionaki S, Kapsia H, Makropoulos I, Metsini A, Skalioti C, Gakiopoulou H, et al. Kidney transplantation outcomes from expanded criteria donors, standard criteria donors or living donors older than 60 years. Ren Fail. 2014; 36:526–533.

8. Kidney Disease Improving Global Outcomes. KDIGO clinical practice guideline for acute kidney injury. Kidney Int. 2012; 2:Suppl 1.
9. Stratta RJ, Rohr MS, Sundberg AK, Armstrong G, Hairston G, Hartmann E, et al. Increased kidney transplantation utilizing expanded criteria deceased organ donors with results comparable to standard criteria donor transplant. Ann Surg. 2004; 239:688–695.

10. Sung RS, Guidinger MK, Lake CD, McBride MA, Greenstein SM, Delmonico FL, et al. Impact of the expanded criteria donor allocation system on the use of expanded criteria donor kidneys. Transplantation. 2005; 79:1257–1261.

11. Wynn JJ, Alexander CE. Increasing organ donation and transplantation: the U.S. experience over the past decade. Transpl Int. 2011; 24:324–332.

12. Kayler LK, Garzon P, Magliocca J, Fujita S, Kim RD, Hemming AW, et al. Outcomes and utilization of kidneys from deceased donors with acute kidney injury. Am J Transplant. 2009; 9:367–373.

13. Fraser SM, Rajasundaram R, Aldouri A, Farid S, Morris-Stiff G, Baker R, et al. Acceptable outcome after kidney transplantation using “expanded criteria donor” grafts. Transplantation. 2010; 89:88–96.

14. Serur D, Saal S, Wang J, Sullivan J, Bologa R, Hartono C, et al. Deceased-donor kidney transplantation: improvement in long-term survival. Nephrol Dial Transplant. 2011; 26:317–324.

15. La Manna G, Comai G, Cappuccilli ML, Liviano D'Arcangelo G, Fabbrizio B, Valentini C, et al. Prediction of three-year outcome of renal transplantation from optimal donors versus expanded criteria donors. Am J Nephrol. 2013; 37:158–166.

16. Ugarte R, Kraus E, Montgomery RA, Burdick JF, Ratner L, Haas M, et al. Excellent outcomes after transplantation of deceased donor kidneys with high terminal creatinine and mild pathologic lesions. Transplantation. 2005; 80:794–800.

17. Farney AC, Rogers J, Orlando G, al-Geizawi S, Buckley M, Farooq U, et al. Evolving experience using kidneys from deceased donors with terminal acute kidney injury. J Am Coll Surg. 2013; 216:645–655.

18. Jacobi J, Rebhan D, Heller K, Velden J, Hilgers KF, Wullich B, et al. Donor acute kidney injury and short-term graft outcome in renal transplantation. Clin Transplant. 2014; 28:1131–1141.

19. Heilman RL, Smith ML, Kurian SM, Huskey J, Batra RK, Chakkera HA, et al. Transplanting kidneys from deceased donors with severe acute kidney injury. Am J Transplant. 2015; 15:2143–2151.

20. Legendre C, Canaud G, Martinez F. Factors influencing long-term outcome after kidney transplantation. Transpl Int. 2014; 27:19–27.

21. Solini S, Aiello S, Cassis P, Scudeletti P, Azzollini N, Mister M, et al. Prolonged cold ischemia accelerates cellular and humoral chronic rejection in a rat model of kidney allotransplantation. Transpl Int. 2012; 25:347–356.

22. Ponticelli C. Ischaemia-reperfusion injury: a major protagonist in kidney transplantation. Nephrol Dial Transplant. 2014; 29:1134–1140.

23. van der Vliet JA, Warlé MC, Cheung CL, Teerenstra S, Hoitsma AJ. Influence of prolonged cold ischemia in renal transplantation. Clin Transplant. 2011; 25:E612–E616.

24. Pérez Valdivia MA, Gentil MA, Toro M, Cabello M, Rodríguez-Benot A, Mazuecos A, et al. Impact of cold ischemia time on initial graft function and survival rates in renal transplants from deceased donors performed in Andalusia. Transplant Proc. 2011; 43:2174–2176.

25. Debout A, Foucher Y, Trébern-Launay K, Legendre C, Kreis H, Mourad G, et al. Each additional hour of cold ischemia time significantly increases the risk of graft failure and mortality following renal transplantation. Kidney Int. 2015; 87:343–349.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download