Abstract
Purpose
Rapid growth of prescription drug expenditures is a problem in South Korea. The objective of this study was to assess the contributions of four variables (therapeutic choice, drug-mix, original use, and price changes) to increases in drug expenditures paid by the National Health Insurance (NHI) in Korea.
Materials and Methods
A retrospective cohort study was conducted between January 1, 2008 and June 30, 2012 utilizing data from the NHI Claims Database of the Health Insurance Review and Assessment Service. The number of target drug types for final analysis was 13959. To analyze the growth rates of drug expenditures, this study used Fisher ideal index and the Laspeyres and Paasche indexes.
In Korea, 97% of the population is registered for National Health Insurance (NHI), social insurance provided by the government through a single plan. Among total medical costs in 2010, 74.5% were at least partially covered by health insurance benefits, and 62.7% were fully covered.1 Of medical costs covered by health insurance, prescription drugs accounted for 26.1% of costs in 2013, slightly decreasing after reform of the Korea Pharmaceutical Pricing Regulations in 2012.2 General drug expenditures were 21.1% of total health expenditures per capita, which is greater than the Organization for Economic Cooperation and Development (OECD) average of 16.4%. However, per-capita expenditures are lower than other countries because of the low average medical costs in Korea. Although healthcare expenditures per capita in France are almost twice those in Korea, drug expenditures per capita in France are lower than those in Korea. Moreover, drug expenditures in Korea are growing, increasing at an average of 12.4% from 2004 to 2010, which is very high, relative to other developed countries. The average annual growth rate of drug expenditure per capita for people aged 10 and up from 2002 to 2009 was 8.1%, which is double the average rate of OECD countries and three to four times greater than that of Denmark and Norway (where drug expenditures per capita are low).3
Although the economic incentives for medical institutions to prescribe drugs were eliminated after the separation of prescription and dispensary practices in Korea in 2000,4 drug expenditures have continuously increased in accordance with an ageing population, increased prevalence of chronic diseases, increased preferences of doctors and patients for expensive drugs, and changing prescription patterns.56 Although the government has implemented various policies to rationalize drug expenditure and made many attempts to reduce drug prices and promote the proper use of drugs,7 it has been met with little success. In order to effectively control increases in drug expenditures, the causes there of need to be examined. The degree to which increases in drug expenditures can be attributed to the prices of expensive prescription drugs or to a pattern of drug use tied to therapeutic methods or disease prevalence must be compared in order to establish effective policy measures.689
In order to describe expenditure trends, Steve Morgan classified factors affecting drug expenditures as a volume effect, a price effect, and a therapeutic choice.10 Volume effect is divided into volume of prescriptions and prescription size. Price effect is divided into the use of generics and price changes. Therapeutic choice is grouped into therapeutic mix and drug mix.
These factors explain trends as a mutually exclusive final factor, which is derived from and complexly affected by changing market conditions. These include changes arising from an ageing population and disease patterns (e.g., a rise in chronic diseases), changes in therapeutic patterns because of new drugs, and increased demand because of successful marketing.
This research used Morgan's model to identify the contribution rates of factors affecting drug expenditure in Korea. We examined the extent to which the following four variables contributed to the increase in drug expenditures paid for by health insurance in Korea: therapeutic choice, drug-mix, original use, and price changes. In other words, a deterministic model designed to describe the rate of increase in drug expenditure per capita as the product of each factor's growth rate was developed to examine the respective contribution rate of each factor.
We referenced two databases from January 2008 to October 2012: detailed statements on treatments covered by health insurance and the NHI Claims Database of the Health Insurance Review & Assessment Service (HIRA), which includes medical claims filed by medical institutions through the NHI and that are compiled and referenced through the Electronic Data Interchange. These data include information on all prescription drugs. Of these, medical treatment statements from January 2008 to June 2012 were analyzed.
Morgan's model used Fisher ideal indexes to identify the contribution rate of four factors that affect drug expenditure: the price effect, drug-mix, original use for the drugs, and therapeutic choice.10 The Fisher ideal approach is the full-attribution of the expenditure approach and the geometric mean of the Laspeyres and Paasche indexes, which show growth rates compared with the previous period. The product of the Fisher indexes of the four factors affecting drug expenditure is titled the Fisher ideal formula, implying the level of change in total drug expenditure per exposed population in the applicable period.
The Laspeyres index shows the growth rate of drug expenditure using a change in one of the factors and assuming that the other factors remain constant. Thus, the Laspeyres index presents a "forward-looking approach," because it shows the future rate of change over the base period. On the other hand, the Paasche index shows the growth rate of expenses using a change in one variable when the other factors are assumed to remain constant in the current period. Thus, it presents a "backward- looking approach" (Appendix 1).
Using exposure to drug treatment (exposure) as an example, the Laspeyres index shows the effect of an increase or reduction of one unit of expenditure in the current period when a patient uses a drug in accordance with the rate of the base period (Appendix 1).
The Laspeyres price index mathematically equals the price index of all drug purchases. The original index shows the effect of switching between brands of drugs and generic use with the same ingredients. This index presents the change in market share of drug brands as a weighted average of price. The exposure to drug treatment (category) index shows the effect of the change in the composition of the population exposed to the drug treatment. The drug-mix index indicates the effect of changes among prescription drugs within the same therapeutic scope.
The Anatomical Therapeutic Chemical Classification System (ATC) codes used in this research are established items developed by the HIRA as therapeutic classifications used for evaluation according to target group classification. The therapeutic subgroup at the second level of classification is set as a standard.
The target drugs used in this analysis comprised all 16528 drugs used from January 2008 to June 2012. Of these, 15728 drugs provided and used new product codes. The number of target drug types used for final analysis was 13959, excluding drugs that did not have ATC codes, were not included in the original classification defined by the HIRA, or were not in use.
The Laspeyres and Paasche indexes were used to adjust and identify the rate of change in drug expenditure according to original use, drug-mix, and therapeutic choice (Table 1). According to the Laspeyres indexes, which show the level of change in price under the assumption that other factors remain constant from the base year, the price dropped by nearly 10% in 2012, whereas it decreased by 1–2% in other years. The significant price drop in 2012 was a result of overall drug price reductions implemented in April 2012.
The rate of change in drug expenditure by therapeutic choice was explored using both the Laspeyres and Paasche indexes. The Laspeyres index, which fixes other factors in the base year and shows the effect of the change in composition of people exposed to drug treatment by therapeutic scope (category) on drug expenditure, increased by almost 6% in 2009, compared to the previous year. Furthermore, it increased by 2.5% in 2010, 3.8% in 2011, and 1.3% in 2012. This implies that therapeutic choice had a significant effect on annual drug expenditure.
The Laspeyres and Paasche indexes were also used to explore the contribution rate of a change in drug-mix on drug expenditure. Controlling for other factors, the annual growth rate of drug expenditure by drug-mix with general ingredients within the therapeutic scope increased by 7.6% in 2009, followed by a continuous increase of 1–3% every year.
The Laspeyres and Paasche indexes were also used to explore the effect of original use on the rate of change in drug expenditure, compared with the previous year; however, original use had almost no effect on the rate of change in drug expenditure.
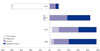
Table 2 presents the results of our analysis using the Fisher ideal indexes that explored the contribution rate of price changes, original use, drug-mix, and therapeutic choice to the rate of change in drug expenditure. Original use had almost no effect on drug expenditure, whereas drug-mix and therapeutic choice were factors continuously affected drug expenditure. Of these, therapeutic choice had a greater effect on drug expenditure. Although overall drug expenditure in 2011–2012 fell because of artificial price adjustments, the contribution rate of drug-mix continuously increased (Fig. 1).
The contribution rate of each factor to the rate of change in drug expenditure was calculated. Because the product of the Fisher drug-mix index is the rate of change in drug expenditure, the Fisher ideal index of the rate of change for all drug expenditure was logged. Accordingly, the contribution rate was calculated by summing the logs of each factor's Fisher ideal index and calculating the absolute value for each factor. The contribution rate was thus calculated as a distribution ratio of each factor with a sum of 100.
With regard to the effect by year, the price dropped significantly in 2012 because of the government's overall price reduction policy, accounting for 80% of the total change in drug prices in the period studied and leading to an overall reduction in price. With the exception of 2012, therapeutic choice and drugmix contributed to about 40–60% and 30–40%, respectively, of the annual increase in drug expenditure. Original use typically had a contribution rate of less than 10% (Table 3, Fig. 2).
Although increases in drug expenditures resulting from an ageing population and an increased prevalence of chronic diseases are a global trend, the difference is striking in Korea, compared to other OECD countries. The 9.8% average annual growth rate over the past 5 years is serious to the extent that it can threaten the financial sustainability of the NHI.2611 In order to control the increase in drug expenditures, the overall prices of 6506 drugs, which accounted for 47.1% of drugs reimbursed in April 2012, were reduced.2
Nevertheless, the increase in drug expenditure was caused not only by price but also by various factors, such as volume of drug use and drug-mix.10121314151617 Thus, it is questionable whether this increase can be controlled using only overall drug price reductions. This study explored mechanisms that led to increasing drug expenditure over the past 4 years and estimated the contribution rate of each factor to this increase.
The model used in this study assessed four factors affecting drug expenditure, based on a study by Morgan.10 These factors include price changes, original use, drug-mix, and therapeutic choice. Of these, the increase in drug expenditure in Korea was mainly attributed to therapeutic choice and drug-mix. Therapeutic choice contributed to over 50% of the increase in drug expenditure, mostly due to changes in the rate and pattern of exposure to drug treatments.13 In Korea, the number of drugs per prescription was 4.16, which is very high, compared to other OECD countries.18 Thus, more therapeutic mixes were prescribed. If there is a pattern of prescribing expensive drugs that have recently been developed, the contribution rate of therapeutic choice will increase. Even the effect of original use is excluded, the annual growth rate of drug expenditure because of drug-mix was 20–30%, which has remained consistent. This reveals a tendency to prefer expensive drugs under the assumption that an increase in the volume of drug use per patient is limited.
The result of this study is similar to that by Morgan,13 which targeted a population of elderly people in Canada and showed that contribution rates of drug and therapeutic choices for drug treatments over 5 years from 1985 to 1989 were 40% and 38%, respectively. Although the contribution rate of drug price was approximately 22% over 5 years in Canada, it was less than 5% in Korea (and sometimes negative), with the exception of 2012. This implies that the government typically controlled drug prices. Furthermore, because of an overall drug price reduction in 2012, holding other factors constant, drug prices per capita were reduced by 9%. This contributed to 80% of the overall change in drug prices and played a significant role in reducing drug prices by 7–8%.
Drug expenditures paid for by the NHI, accounted for 26.1% of the total health insurance expenditure in 2013, which is high.19 An inability to control of steep increases in drug expenditure can threaten the financial sustainability of the NHI. Although the government has focused on prices, it has failed to control increases in drug expenditure. This is because the root of the increase in drug expenditures in Korea resides in therapeutic choice and drug-mix rather than in price. Thus, in order to reduce drug expenditures in Korea, various prescription drugs for patients should be controlled, and policies to prevent patterns of prescribing expensive drugs that have not been proven effective and incentives for cost-effective drugs within the therapeutic scope are required. Despite limitations in the data on claims made to Korea's NHI, this study is significant in that it examined the causes of the increased in drug expenditures using mass data at the national level for all Korean people.
Figures and Tables
Table 1
Laspeyres and Paasche Indexes for Factors Affecting Drug Cost by Year
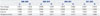
Table 2
Fisher Ideal Indexes for Factors Affecting Drug Expenditure by Year

Appendix
APPENDIX 1
1. Laspeyres indexes
Exposure: patterns according to therapeutic choice
Drug: drug-mix
Original: original use for the drugs
Prices: price changes
2. Paasche indexes
Exposure: patterns according to therapeutic choice
Drug: drug-mix
Original: original use for the drugs
Prices: price changes
References
1. Park M, Chol Y, Tae Y, Chol J, Paek S, Lee H. 2010 Survey on National Health Insurance out-of-pocket expenditure. Seoul: National Health Insurance Service Health Insurance Police Research Institute;2011.
2. Ministry of Health and Welfare. Evaluation of price cuts for drugs whose patents are expired after one year. Ministry of Health and Welfare;2013. accessed on 2013 July 24. http://www.129.go.kr/news/news02_view.jsp?n=7293.
3. OECD. Health at a glance 2011: OECD indicators. Paris: OECD Publishing;2013.
4. Lee YK, Chang S, Sin JK, Park JY. Separation of prescription and dispensing: changes in pharmaceutical costs and related policy issues. Seoul: Korea Institute for Health and Social Affairs;2001.
5. Ess SM, Schneeweiss S, Szucs TD. European healthcare policies for controlling drug expenditure. Pharmacoeconomics. 2003; 21:89–103.

6. Lee IH, Park S, Lee EK. Generic utilization in the Korean National Health Insurance Market; cost, volume and influencing factors. Yakhak Hoeji. 2014; 58:99–106.
7. Shin Y, Whang D, Kang K, Bae EY, Lee S, Lee C. The National Health Insurance and its policy tasks. Seoul: Korea Institute for Health and Social Affairs;2010.
8. Bae EY. Study on the drug expenditure trend in Korea. Korean J Health Econ Policy. 2007; 13:39–54.
9. Choi YJ, Shalowitz JI. Major growths of Korean pharmaceutical expenditure. J Korea Soc Health Inform Stat. 2011; 36:15–24.
10. Morgan S. Drug expenditure trends in the Canadian provinces: magnitude and causes from 1998 to 2004. Healthc Policy. 2005; 1:85–99.

11. Cheong I, Lee S. A study on an incentive system for reducing pharmaceutical expenditure in Korea. Health Soc Welf Rev. 2009; 29:243–267.

12. Chernew ME, Smith DG, Kirking DM, Fendrick AM. Decomposing pharmaceutical cost growth in different types of health plans. Am J Manag Care. 2001; 7:667–673.
13. Morgan SG. Quantifying components of drug expenditure inflation: the British Columbia seniors' drug benefit plan. Health Serv Res. 2002; 37:1243–1266.

14. Prieto L, Flöter S. The new EMCDDA standard table on public expenditure: a step forward in quantifying drug-related costs in Europe. Suchttherapie. 2009; 10:S211.

15. Jang S, Park CM, Bae G, Lee HJ, Kim HS. Determinants of publicly funded drug expenditure in South Korea. Seoul: Health Insurance Review & Assessment Service;2011.
16. Park SE, Lim SH, Choi HW, Lee SM, Kim DW, Yim EY, et al. Evaluation on the first 2 years of the positive list system in South Korea. Health Policy. 2012; 104:32–39.

17. Bae G, Park C, Lee H, Han E, Kim DS, Jang S. Effective policy initiatives to constrain lipid-lowering drug expenditure growth in South Korea. BMC Health Serv Res. 2014; 14:100.

18. IMS Health intelligence applied, HIRA. Benchmarking prescribing behaviour for 207 disease conditions in Korea against those in specific reference markets, and to explore the drivers of any disparities in prescribing behaviour. Seoul: IMS Health intelligence applied, Health Insurence Review & Assecement;2006.
19. National Health Insurance Service, Health Insurance Review & Assessment Service. 2012 National Health Insurance statistical yearbook. Seoul: National Health Insurance Service, Health Insurance Review & Assessment Service;2013.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




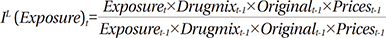
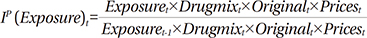
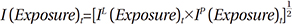
 XML Download
XML Download