Abstract
Purpose
Steroids may play a role in preventing the early recurrence of atrial fibrillation (AF) after radiofrequency catheter ablation (RFCA). However, optimal doses and route of steroid delivery have not yet been determined. This study evaluated the effect of two different doses of a single bolus injection of steroids on AF recurrence after RFCA.
Materials and Methods
Of 448 consecutive AF patients who underwent RFCA, a single steroid bolus was injected into 291 patients. A low-dose steroid group (n=113) received 100 mg of hydrocortisone and a moderate-dose steroid group (n=174) received 125 mg of methylprednisolone. We used propensity-score matching to select patients as follows: control (n=95), low-dose (n=95), and moderate-dose steroid groups (n=97).
Results
Pericarditis developed in 1 (1.1%) control patient, 2 (2.1%) low-dose patients and 0 moderate-dose patients. Maximum body temperature and C-reactive protein were significantly decreased in the moderate-dose steroid group compared to the other groups (p<0.01). The number of patients of early AF recurrence (≤3 months) did not differ among three groups. Early recurrence was 24 (25%) in the control, 24 (25%) in the low-dose and 25 (26%) in the medium-dose groups (p=0.99). Compared with control group, low-dose or moderate-dose steroid treatment did not effectively decrease mid-term (3-12 months) AF recurrence [22 (23%) vs. 23 (24%) vs. 18 (19%); p=0.12].
Go to : 
The incidence of atrial fibrillation (AF) is increasing. AF is associated with a considerable number of comorbidities including a 3-fold increase in the risk of heart failure and a 5-fold increase in the risk of stroke.1,2,3 Radiofrequency catheter ablation (RFCA) is an effective treatment for drug-refractory AF with 33-86% of patients achieving favorable outcomes.4,5,6 However, studies on long-term outcomes after RFCA commonly report recurrence of AF, especially within the first few weeks after RFCA.7 For early recurrences, inflammation during RFCA is suggested to be a major precipitating factor. The role of inflammation in initiating and perpetuating AF is under investigation since inflammation is suggested to be an important cause of the high incidence of post-operative AF in cardiac surgeries.8,9 A few studies reported independent relationships between inflammatory markers and AF incidence. These studies propose that acute inflammation transiently induces electrical and structural remodeling of the left atrium.10 According to studies that investigated early AF recurrence after RFCA, inflammation might cause early recurrence, as reflected in elevated inflammatory markers and body temperatures.11,12
To prevent early AF recurrence after RFCA, corticosteroid injection is used to reduce post-procedure inflammation. Corticosteroids are potent inhibitors of the pro inflammatory cascade. Corticosteroids reduce inflammatory markers such as interleukin (IL)-6, IL-8, tumor necrosis factor α, C-reactive protein (CRP), and oxygen-free radicals after cardiac surgery.13,14,15,16 A randomized controlled trial by Koyama, et al.12 showed that a small amount of corticosteroid administered after RFCA seems to prevent immediate AF recurrence. However, the use of corticosteroids for secondary prevention of AF has not yet been confirmed and has not been thoroughly studied in a large population. In addition, an appropriate corticosteroid dosage has not been determined. Side effects of corticosteroids such as infections, hyperglycemia, gastrointestinal bleeding, adrenal suppression, hallucination, and depression have been acknowledged, and physicians realize that the benefits of steroids must outweigh their potentially dangerous side effects. Moreover, high-dose steroids are reported to cause ventricular arrhythmia and even promote AF.13 Thus, studies investigating adequate minimal dose for steroids and appropriate method of steroid administration that will prevent early AF recurrence after RFCA are warranted. This study evaluated the effect of a single bolus intravenous injection of low-dose or moderate-dose corticosteroids on preventing AF recurrence after RFCA.
Go to : 
This study enrolled 448 patients with drug-resistant AF who underwent RFCA. Patients were treated at Yonsei University Hospital (Seoul, Korea) from July 2007 through December 2012. All patients were newly enrolled in this study and none had a history of ablation. As a control group, 161 AF patients who underwent radiofrequency ablation for AF during the same period were analyzed. Patients who had previously undergone RFCA were excluded. All patients had symptomatic, paroxysmal or persistent AF. The decision to treat patients with low-dose or moderate-dose steroids was made by operating physicians. One physician (Dr. Joung) used a strategy of treatment with corticosteroids after catheter ablation and the other physicians (Dr. Lee and Dr. Pak) did not use this strategy. Patients were randomly assigned to operating physicians without this information. All operating physicians used the same ablation protocol.
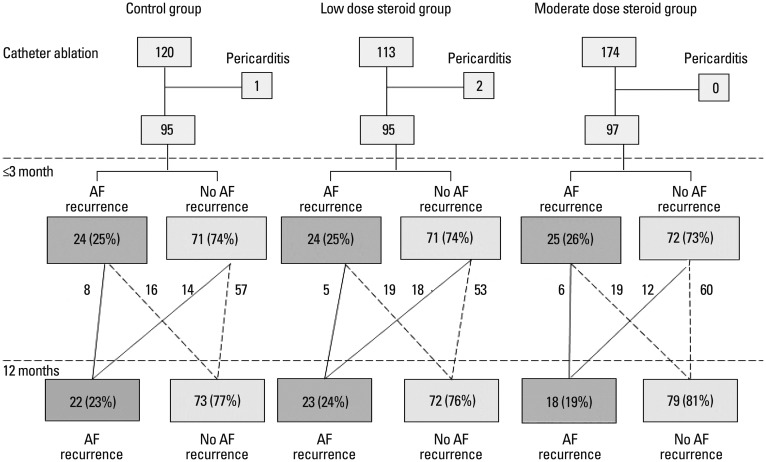
In this triple cohort study, the corticosteroid group had two subgroups based on corticosteroid potency. The low-dose steroid group (n=113) received 100 mg hydrocortisone sodium succinate (Solu-Cotef, Pfizer Manufacturing, Puurs, Belgium) in 5 mL of 0.9% sodium chloride. The moderate-dose steroid group (n=174) received a bolus injection of methyl prednisolone 125 mg (Solu-Medrol, Pfizer Manufacturing, Puurs, Belgium) in 5 mL of 0.9% sodium chloride (625 mg as in hydrocortisone equivalence). Steroid solutions were administered intravenously within 30 minutes after RFCA. Patients in the control group (n=161) were treated conservatively without steroids after ablation. From the cohort, we selected age- and gender-matched cohorts by propensity scores. The final cohorts were control (n=95, 80 men), low-dose (n=95, 80 men), and moderate-dose steroid group (n=97, 82 men) (Fig. 1).
Paroxysmal AF was defined as an AF episode that spontaneously terminated and lasted for more than 30 s, but less than 7 days. Persistent AF was defined as an AF episode that either lasted longer than 7 days or required termination by cardioversion, either with anti-arrhythmic drugs (AADs) or by direct current cardioversion. All AADs were discontinued at 5 half-lives before the ablation procedure. Other drugs were administered throughout the study period. The study protocol was approved by the Institutional Review Board of Severance Cardiovascular Hospital, Seoul, Korea and complied with the Declaration of Helsinki.
Three-dimensional geometry of the left atrial (LA) was reconstructed with a CARTO (Biosense Webster, Inc., Diamond Bar, CA, USA) or NavX™ (St. Jude Medical, Inc., St. Paul, MN, USA) electroanatomic mapping system. Before ablation, triggers were evaluated after direct current cardioversion of AF. Circumferential pulmonary vein isolation (PVI) was performed on all patients using a 3.5-mm irrigated-tip catheter (ThermoCool, Biosense Webster, Diamond Bar, CA, USA) with the maximum temperature set at 50℃ and power output 25-35 W. We used a circular mapping catheter (Lasso, Biosense Webster) to confirm PVI. The endpoint of the PVI was creation of a bidirectional conduction block between the atrium and the pulmonary veins (PVs). A cavo-tricuspid isthmus block line was created with confirmation of the bidirectional block. After PVI, if AF was sustained, additional ablation, consisting of linear ablation, complex fractionated atrial electrogram (CFAE) ablation and/or superior vena cava (SVC) isolation, was performed.17 If AF did not terminate, sinus rhythm was restored by transthoracic cardioversion.17
Patients remained hospitalized under continuous rhythm monitoring (IntelliVue Telemetry System, Philips Healthcare, Amsterdam, the Netherlands) for at least 48 hours after RFCA with subsequent follow-up. Body temperature was measured on the morning of the ablation and every 8 hours for 72 hours after ablation using an eardrum thermometer (Infrared thermometer IRT4020, Braun GmbH, Kronberg, Germany) in the ear canal. CRP, C3, C4 levels and complete blood cell counts were measured 1 day and 1 week after catheter ablation.
After discharge, all patients underwent follow-up at 1 week and 1 month post-procedure and every three months thereafter. At each hospital visit, patients underwent 12-lead electrocardiography and intensive questioning regarding arrhythmia-related symptoms (palpitation, chest discomfort and dizziness) since the previous follow-up visit. Holter monitoring for 24 hour was performed at 1 and 3, 6, 12, 18, and 24 months after RFCA. Portable event electrocardiographic monitoring (EV-101, Parama-Tech, Fukuoka, Japan) was performed to evaluate arrhythmic assessment in patients with intermittent symptoms.
We defined early recurrence as any episode of AF or atrial tachycardia (AT) of at least 30 seconds that occurred within 3 months after AF ablation. Mid-term recurrence was defined as any AF or AT of at least 30 seconds that occurred between 3 and 12 months.18
Three patients: 1 (1.1%) from the control group, and 2 (2.1%) from the low-dose group developed pericarditis after RFCA; these patients were excluded from the analysis of recurrence because they were treated with higher doses of corticosteroids for pericarditis
If electrocardiograms (ECGs) showed any AF episodes during the follow-up, patients received a diagnosis of clinical recurrence of AF, irrespective of the presence of symptoms.19 Patients with AF recurrence were treated temporarily with Class I, II, III, and/or IV AADs, and AADs were discontinued if stable sinus rhythm was maintained. Discontinuation of AADs was attempted in patients whose recurrent AF disappeared after temporary treatment; if sinus rhythm was maintained, the drugs were stopped permanently.
SPSS version 19.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. Continuous variables were expressed as mean±standard deviation, and categorical variables were described using numbers or percentages. Comparisons of differences between two groups were made by chi-square analysis and unpaired Student's t-test. Time to atrial fibrillation recurrence 31 days after catheter ablation was estimated by the Kaplan-Meier method, with comparisons made using the log-rank test. A p value <0.05 was considered statistically significant.
Go to : 
Baseline clinical characteristics are shown in Table 1. The groups contained 95, 95, and 97 patients, respectively. Baseline characteristics of mean age, gender predominance, co morbidities, and echocardiographic parameters were not different among the 3 groups. Although the proportion of patients taking class III AADs was different among the 3 groups [39 (41%) vs. 22 (23%) vs. 30 (31%), p=0.04], other medications were not significantly different among the groups.
Catheter ablation parameters and recurrence rates of AF are presented in Table 2. Circumferential PVI was successfully performed in all patients, and bidirectional conduction block was created at all 4 PVs. A cavo-tricuspid isthmus block line with confirmation of bidirectional block was successfully created in 81 (90%) in the control group, 88 (98%) in the low-dose group, and 84 (91%) in the moderate-dose group. No differences were observed in linear ablation, CFAE or superior vena cava (SVC) isolation, except posterior box and lateral box linear ablations. Total procedure duration time, total fluoroscopy time and duration of RFCA were not statistically different among the three groups.
We measured white blood cell (WBC) counts, CRP, and body temperature after RFCA (Table 3). Patients in the moderate-dose steroid group had the lowest maximal body temperature (37.1±0.5℃ control vs. 37.0±0.5℃ low-dose vs. 36.8±0.2℃ moderate-dose; p<0.01) and the lowest CRP (23.7±13.4 mg/L control vs. 20.3±12.8 mg/L low-dose vs. 5.2±8.4 mg/L moderate-dose; p<0.01). Although not statistically significant, the control group had the highest mean WBC count compared to low- and moderate-dose steroid group (9279±2668/µL control vs. 8880±2332/µL low-dose vs. 9122±2559/µL moderate-dose; p=0.61). Post-RFCA complications, specifically development of pericarditis, were 1 (1.1%) in control, 2 (2.1%) in low-dose and 0 in moderate-dose.
No major complications were documented in any group during the follow-up period, which had a median of 26±12 months. Rates of infection and hematoma were not different among the 3 groups. One year after RFCA, 16 of 24 (67%) early AF recurrence patients in control group maintained sinus rhythm (Fig. 1). For low-dose and moderate-dose steroid group, 19 of 24 (79%) and 19 of 25 (76%) early AF recurrence maintained to be sinus rhythm. We compared recurrence rates among the 3 groups. The number of patients of early AF recurrence (≤3 months) did not differ among the 3 groups. Early recurrence was 24 (25%) in the control, 24 (25%) in the low-dose and 25 (26%) in the medium-dose groups (p=0.99). Low-dose and moderate-dose steroids did not decrease mid-term AF recurrence (3-12 months) [22 (23%) vs. 23 (24%) vs. 18 (19%); p=0.12] after ablation, compared with controls (Table 3).
Fig. 2 shows Kaplan-Meier survival curves for AF recurrence in each group, with no significant differences in the rates of patients with no AF recurrence (p=0.11 by log-rank test). The first three months was presumed to be a blanking period, because AF recurrences early after RFCA are considered to be transient.17 By multivariate Cox-regression analysis, persistent AF (HR 1.67, 95% CI 1.18-2.37; p=0.04) was the only independent predictor of late recurrence after adjusting for steroid use, age >75, diabetes mellitus, LA size and ablation time.
Go to : 
The main finding of this study was that a single bolus injection of low-dose or moderate-dose corticosteroids did not prevent AF recurrence after RFCA. Moderate doses of corticosteroids resulted in decreased inflammation after RFCA, which was reflected in significantly lower WBC counts, CRP, and body temperature, compared to the control group. However, the decrease in inflammatory markers was not accompanied by a decreased incidence in recurrence. Our data suggested that a single bolus injection of low-dose or moderate-dose corticosteroids might not be sufficient to prevent post-ablation AF recurrence.
The use of corticosteroids to prevent AF has mainly been evaluated in cardiothoracic surgery.20,21 Meta-analyses revealed that corticosteroid therapy is associated with a decrease in more than 50% postoperative AF episodes,20 although heterogeneity in variable dosages and steroid dosing intervals must be considered. AF after cardiac and non cardiac surgery had been known to be usually prevented with steroid, however, it must be noted that the surgery is different from catheter ablation, causing more severe systemic inflammation and infection.
Dernellis and Panaretou22 demonstrated that 16 mg methyl prednisolone for 4 weeks, tapering to 4 mg for 4 months, significantly reduced AF recurrence (50% vs. 2.9%, p<0.001). Koyama, et al.12 reported that intravenous hydrocortisone (2 mg/kg) immediately after ablation and oral prednisolone (0.5 mg/kg/day) for 3 days after the procedure resulted in a marked decrease in immediate AF recurrence. Moreover, AF-free states were maintained without AADs in a corticosteroid-treated group (85% vs. 71%, p<0.05). The efficacy of steroids and proper doses and dosing intervals to prevent AF recurrence after RFCA are still debatable.
In this study, single doses of steroids did not prevent AF recurrence after RFCA. The total dosage of steroid was not different between our study and the study by Koyama, et al.12 For example, a total dosage of steroid given to a 60 kg body weight patient was 625 mg as in hydrocortisone equivalence for moderate-dose group of our study, while 480 mg as in hydrocortisone equivalence was injected in the study by Koyama, et al.12 Therefore, the negative result of this study is more likely caused by administration method. Recently, Nascimento, et al.23 reported that a single prophylactic corticosteroid before AF ablation did not prevent systemic inflammation or alter the healing of the ablation lesion in pigs. This result is consistent with our study.
Effective doses or dosing intervals to decrease AF recurrence are not fully established, even in cardiac surgery studies. An intermediate-dose group (50-210 mg dexamethasone, 1333-5600 mg as in hydrocortisone equivalence) significantly reduced incidences of AF after cardiothoracic surgery, compared with placebo.21,24 Both low-dose (<200 mg/d) and high-dose steroids (>1000 mg/d) were ineffective compared to moderate doses (200-1000 mg/d) of corticosteroid for preventing AF after cardiac surgery.20 However, high-dose steroids do not prevent postoperative AF.21,25,26 Moreover, high-dose steroids induced AF by modulating potassium channels. Our previous study showed that a bolus injection of low-dose steroids was not effective in preventing AF recurrence.27
Whether a single bolus injection of steroids is effective for prevent inflammation is unknown. A previous study that investigated steroid doses to affect survival in acute respiratory distress syndrome suggested that more than a single bolus injection of steroids was effective at reducing systemic inflammatory responses.28 In this study, however, a single bolus injection of moderate-dose steroids significantly lowered inflammation after RFCA without noticeable side effects of corticosteroid.
This study had several limitations. First, data were reviewed retrospectively and low-dose or moderate-doses of steroids were not randomly allocated. However, using propensity score matching, no significant difference was seen in patient baseline characteristics, ablation protocol, or procedural time among the 3 groups. Second, since we used conventional electrocardiogram and ambulatory Holter monitoring, asymptomatic AF recurrence might not be detected. A continuous recording for a longer period would provide more insight into the efficacy of steroids.
A single injection of moderate-dose steroid decreased inflammation. However, single bolus injections of low-dose or moderate-dose steroids were not effective in preventing immediate, early or midterm AF recurrence after RFCA. Our study suggested that a single dose of corticosteroids might not be effective to prevent AF after RFCA.
Go to : 
ACKNOWLEDGEMENTS
This study was supported in part by the Basic Science Research Program through the National Research Foundation of Korea, funded by the Ministry of Education, Science and Technology (NRF-2010-0021993, NRF-2012R1A2A2A 02045367), and the Korean Healthcare Technology R&D Project, Ministry of Health & Welfare (HI12C1552).
Go to : 
References
1. Wilke T, Groth A, Mueller S, Pfannkuche M, Verheyen F, Linder R, et al. Incidence and prevalence of atrial fibrillation: an analysis based on 8.3 million patients. Europace. 2013; 15:486–493. PMID: 23220354.

2. Wang TJ, Larson MG, Levy D, Vasan RS, Leip EP, Wolf PA, et al. Temporal relations of atrial fibrillation and congestive heart failure and their joint influence on mortality: the Framingham Heart Study. Circulation. 2003; 107:2920–2925. PMID: 12771006.
3. Chin JY, Youn HJ. The effect of ablation for paroxysmal atrial fibrillation on left atrial volume and function: a one-year follow-up study. Yonsei Med J. 2014; 55:895–903. PMID: 24954316.

4. Gerstenfeld EP, Callans DJ, Dixit S, Zado E, Marchlinski FE. Incidence and location of focal atrial fibrillation triggers in patients undergoing repeat pulmonary vein isolation: implications for ablation strategies. J Cardiovasc Electrophysiol. 2003; 14:685–690. PMID: 12930245.
5. Shah DC, Haïssaguerre M, Jaïs P, Hocini M, Yamane T, Deisenhofer I, et al. Electrophysiologically guided ablation of the pulmonary veins for the curative treatment of atrial fibrillation. Ann Med. 2000; 32:408–416. PMID: 11028689.

6. Gerstenfeld EP, Guerra P, Sparks PB, Hattori K, Lesh MD. Clinical outcome after radiofrequency catheter ablation of focal atrial fibrillation triggers. J Cardiovasc Electrophysiol. 2001; 12:900–908. PMID: 11513440.

7. Oral H, Knight BP, Ozaydin M, Tada H, Chugh A, Hassan S, et al. Clinical significance of early recurrences of atrial fibrillation after pulmonary vein isolation. J Am Coll Cardiol. 2002; 40:100–104. PMID: 12103262.

8. Issac TT, Dokainish H, Lakkis NM. Role of inflammation in initiation and perpetuation of atrial fibrillation: a systematic review of the published data. J Am Coll Cardiol. 2007; 50:2021–2028. PMID: 18021867.
9. Bruins P, te Velthuis H, Yazdanbakhsh AP, Jansen PG, van Hardevelt FW, de Beaumont EM, et al. Activation of the complement system during and after cardiopulmonary bypass surgery: postsurgery activation involves C-reactive protein and is associated with postoperative arrhythmia. Circulation. 1997; 96:3542–3548. PMID: 9396453.
10. Aviles RJ, Martin DO, Apperson-Hansen C, Houghtaling PL, Rautaharju P, Kronmal RA, et al. Inflammation as a risk factor for atrial fibrillation. Circulation. 2003; 108:3006–3010. PMID: 14623805.
11. McCabe JM, Smith LM, Tseng ZH, Badhwar N, Lee BK, Lee RJ, et al. Protracted CRP elevation after atrial fibrillation ablation. Pacing Clin Electrophysiol. 2008; 31:1146–1151. PMID: 18834466.

12. Koyama T, Tada H, Sekiguchi Y, Arimoto T, Yamasaki H, Kuroki K, et al. Prevention of atrial fibrillation recurrence with corticosteroids after radiofrequency catheter ablation: a randomized controlled trial. J Am Coll Cardiol. 2010; 56:1463–1472. PMID: 20951321.
13. Chiappini B, El Khoury G. Risk of atrial fibrillation with high-dose corticosteroids. Expert Opin Drug Saf. 2006; 5:811–814. PMID: 17044807.

14. Jahangiri M, Camm AJ. Do corticosteroids prevent atrial fibrillation after cardiac surgery? Nat Clin Pract Cardiovasc Med. 2007; 4:592–593. PMID: 17724480.

15. Liakopoulos OJ, Schmitto JD, Kazmaier S, Bräuer A, Quintel M, Schoendube FA, et al. Cardiopulmonary and systemic effects of methylprednisolone in patients undergoing cardiac surgery. Ann Thorac Surg. 2007; 84:110–118. PMID: 17588396.

16. Bourbon A, Vionnet M, Leprince P, Vaissier E, Copeland J, McDonagh P, et al. The effect of methylprednisolone treatment on the cardiopulmonary bypass-induced systemic inflammatory response. Eur J Cardiothorac Surg. 2004; 26:932–938. PMID: 15519185.

17. Oral H, Chugh A, Lemola K, Cheung P, Hall B, Good E, et al. Noninducibility of atrial fibrillation as an end point of left atrial circumferential ablation for paroxysmal atrial fibrillation: a randomized study. Circulation. 2004; 110:2797–2801. PMID: 15505091.

18. Calkins H, Kuck KH, Cappato R, Brugada J, Camm AJ, Chen SA, et al. 2012 HRS/EHRA/ECAS Expert Consensus Statement on Catheter and Surgical Ablation of Atrial Fibrillation: recommendations for patient selection, procedural techniques, patient management and follow-up, definitions, endpoints, and research trial design. Europace. 2012; 14:528–606. PMID: 22389422.

19. Koyama T, Sekiguchi Y, Tada H, Arimoto T, Yamasaki H, Kuroki K, et al. Comparison of characteristics and significance of immediate versus early versus no recurrence of atrial fibrillation after catheter ablation. Am J Cardiol. 2009; 103:1249–1254. PMID: 19406267.

20. Marik PE, Fromm R. The efficacy and dosage effect of corticosteroids for the prevention of atrial fibrillation after cardiac surgery: a systematic review. J Crit Care. 2009; 24:458–463. PMID: 19327322.

21. Ho KM, Tan JA. Benefits and risks of corticosteroid prophylaxis in adult cardiac surgery: a dose-response meta-analysis. Circulation. 2009; 119:1853–1866. PMID: 19332460.
22. Dernellis J, Panaretou M. Relationship between C-reactive protein concentrations during glucocorticoid therapy and recurrent atrial fibrillation. Eur Heart J. 2004; 25:1100–1107. PMID: 15231367.

23. Nascimento T, Mota F, dos Santos LF, de Araujo S, Okada M, Franco M, et al. Impact of prophylactic corticosteroids on systemic inflammation after extensive atrial ablation in pigs. Europace. 2012; 14:138–145. PMID: 21846643.

24. Baker WL, White CM, Kluger J, Denowitz A, Konecny CP, Coleman CI. Effect of perioperative corticosteroid use on the incidence of postcardiothoracic surgery atrial fibrillation and length of stay. Heart Rhythm. 2007; 4:461–468. PMID: 17399635.

25. Morariu AM, Loef BG, Aarts LP, Rietman GW, Rakhorst G, van Oeveren W, et al. Dexamethasone: benefit and prejudice for patients undergoing on-pump coronary artery bypass grafting: a study on myocardial, pulmonary, renal, intestinal, and hepatic injury. Chest. 2005; 128:2677–2687. PMID: 16236942.
26. Chaney MA, Nikolov MP, Blakeman BP, Bakhos M, Slogoff S. Hemodynamic effects of methylprednisolone in patients undergoing cardiac operation and early extubation. Ann Thorac Surg. 1999; 67:1006–1011. PMID: 10320243.

27. Won H, Kim JY, Shim J, Uhm JS, Pak HN, Lee MH, et al. Effect of a single bolus injection of low-dose hydrocortisone for prevention of atrial fibrillation recurrence after radiofrequency catheter ablation. Circ J. 2013; 77:53–59. PMID: 23006726.

28. Minneci PC, Deans KJ, Banks SM, Eichacker PQ, Natanson C. Meta-analysis: the effect of steroids on survival and shock during sepsis depends on the dose. Ann Intern Med. 2004; 141:47–56. PMID: 15238370.

Go to : 




 PDF
PDF ePub
ePub Citation
Citation Print
Print






 XML Download
XML Download