Abstract
Purpose
To determine whether levels of nerve growth factor (NGF) and heparin-binding epidermal growth factor-like growth factor (HB-EGF) can be used to objectively assess overactive bladder syndrome (OAB) treatment outcome and to evaluate the effects of fixed-dose fesoterodine on OAB symptoms.
Materials and Methods
This study included 124 participants (62 patients with OAB and 62 controls) in Severance Hospital between 2010 and 2012. In patients with OAB, 4 mg fesoterodine was administered once daily. Repeated evaluations of putative biomarker levels, urine creatinine (Cr) levels, and questionnaire responses, including the Overactive Bladder Symptom Score (OABSS) and the Overactive Bladder Questionnaire (OAB q), were performed from baseline to 16 weeks.
Results
Urinary levels of NGF/Cr (OAB: 1.13±0.9 pg/mg; control: 0.5±0.29 pg/mg) and HB-EGF/Cr (OAB: 8.73±6.55 pg/mg; control: 4.45±2.93 pg/mg) were significantly higher in subjects with OAB than in controls (p<0.001). After 16 weeks of fixed-dose fesoterodine treatment, urinary NGF/Cr levels (baseline: 1.13±0.08 pg/mg; 16 weeks: 0.60±0.4 pg/mg; p=0.02) and HB-EGF/Cr levels significantly decreased (baseline: 8.73±6.55 pg/mg; 16 weeks: 4.72±2.69 pg/mg; p=0.03, respectively). Both the OABSS and OAB q scores improved (p<0.001). However, there were no a statistically significant correlations between these urinary markers and symptomatic scores.
Overactive bladder syndrome (OAB) is highly prevalent and exhibits lower urinary tract symptoms (LUTS) of frequency, nocturia, and urgency, with or without urgency urinary incontinence; urgency is the primary symptom defining OAB.1,2 Painful bladder syndrome/interstitial cystitis (PBS/IC) is another prevalent urologic disease characterized by urinary frequency, urgency, and chronic bladder pain.3 Although many studies have investigated OAB and PBS/IC, its causes and mechanisms remain poorly understood. A high percentage of symptom overlap with PBS/IC has been noted for OAB, which implies a possible common pathophysiology, despite these being different diseases.4 Although diagnoses are made based on the evaluation of a patient's symptoms, the role of urinary biomarkers in assessing either OAB or PBS/IC has garnered greater interest. Measurement of biomarkers in urine have been shown to be simple and easy to look at potential pathophysiologies.5,6,7,8,9 In fact, previous studies have identified nerve growth factor (NGF) as a potential diagnostic biomarker for OAB and as a prognostic marker for measuring treatment response in patients with PBS/IC.5,6,7
Three biomarkers have been identified in urine from PBS/IC patients: epidermal growth factor (EGF), heparin-binding epidermal growth factor-like factor (HB-EGF), and antiproliferative factor (APF).8 Among these, HB-EGF is functionally antagonized by other factors via a mitogen-APF complex, and its level significantly changes between PBS/IC patients with and without ulcers.9 Urinary urgency and frequency are related to a defective urothelium in patients with PBS/IC, and HB-EGF is involved in this pathway.10 In OAB patients, urgency and frequency are also core symptoms.1,2 To our knowledge, no studies have investigated the diagnostic role of urinary HB-EGF levels with OAB and the effect of OAB treatment on HB-EGF levels.
Therefore, this study was designed to examine whether urinary NGF and HB-EGF levels can be used as objective tools for assessing the therapeutic outcomes of antimuscarinic OAB treatment. We also evaluated the effects of a fesoterodine fixed-dosing regimen on OAB symptoms in OAB subjects using standardized questionnaires.
Between December 2010 and June 2012, 124 subjects were recruited from Yonsei University Severance Hospital in Seoul, Korea. The study was approved by the hospital's Institutional Review Board, and all patients provided informed consent to participate in the study. OAB was diagnosed based on the clinical symptoms of urgency or urgency incontinence, and further confirmed using a 3-day voiding diary. The inclusion criterion involved the patient reporting at least three episodes of urgency/urgency urinary incontinence within a 3-day period without previous history of diagnosis or treatment for OAB. OAB patients were treated with fixed-dose fesoterodine (4 mg, daily) for 4-6 months. No one stopped or increased the quantity of medication. OAB patients were also classified as OAB wet (n=39) or OAB dry (n=23) based on their voiding record of having urgency urinary incontinence or not, respectively. Control subjects were recruited from among gynecologic patients who were scheduled to undergo hysterectomy or myomectomy and who did not have urinary tract disease or LUTS. The exclusion criteria were the presence of asthma, chronic obstructive pulmonary disease, allergic diseases, a history of psychiatric disorders, urinary tract disease (infection, stone, cystitis, urothelial cell cancer, etc.), neurological diseases, gynecologic cancers, or gynecologic inflammatory diseases (pelvic inflammatory disease, endometriosis, etc.).
All enrolled patients underwent urogynecologic assessments by the same doctor, according to the algorithm from the Fourth International Consultation on Incontinence.11 OAB patients also underwent cystoscopy and urodynamic measurements to evaluate uroflowmetry, detrusor overactivity (DO), postvoid residual urine, and total bladder capacity. Urine samples were collected from the control patients preoperatively and 1 month postoperatively. In OAB patients, the urine samples were collected before treatment and at 1 and 4 months after treatment. Urinalysis was performed at the time of urine collection to confirm the absence of pyuria or detectable bacteria. Subjective urinary, coital, and bowel symptoms were assessed in OAB patients through an interview utilizing two questionnaires, the Overactive Bladder Symptom Score (OABSS) and Overactive Bladder Questionnaire (OAB q).
To assess symptomatic changes, OAB subjects completed standardized OAB-specific questionnaires including the OABSS12 and the OAB q13 at baseline and 4 months after starting fesoterodine treatment. The OABSS evaluated four symptoms: daytime frequency, nighttime frequency, urgency, and urgency urinary incontinence. The overall score was the simple sum of the four symptom scores. The OAB q comprises an eight-item Symptom Bother scale and a 25-item health-related quality of life (HRQL) scale with four domains (concern, coping, sleep, and social interaction).
The primary endpoints were changes in urinary levels of NGF and HB-EGF in OAB patients from baseline to 4 months after fesoterodine therapy. The secondary endpoints included changes in OABSS and OAB q scores at the same time-points. The questionnaire was self-administered; beforehand, the interviewer thoroughly explained each questionnaire and the questions. Only questionnaires that contained responses to all items were evaluated (i.e., incomplete questionnaires were excluded).
Clean-catch urine specimens were obtained from both OAB and control patients. Aliquots (20-30 mL) of the samples were immediately put on ice and centrifuged at 3000 rpm for 10 min at 4℃. The supernatants were carefully collected and frozen (-80℃) until enzyme-linked immunosorbent assays (ELISAs) were performed. At the same time, 3-mL urine aliquots were used to measure urinary creatinine (Cr) levels.
Urinary NGF levels were measured using the Emax® Immuno Assay System (Promega, Madison, WI, USA) with an ELISA kit (USCN Life Science, Wuhan, Hubei, China). Assays were performed according to the manufacturer's instructions, as previously reported.5 For the HB-EGF ELISA, each well of a 96-well Immulon II plate (Dynatech Laboratories, Chantilly, VA, USA) was coated with 200 µL of urine at 4℃, overnight, as previously reported.14 The plates were blocked with 5% fetal bovine serum, and anti-HB-EGF antibody (1 µg/mL) (R&D Systems, Minneapolis, MN, USA) was added in addition to biotinylated anti-goat IgG/avidin D-horseradish peroxidase. The total urinary NGF and HB-EGF levels were further normalized to the concentration of urinary creatinine (NGF/Cr and HB-EGF/Cr levels).
Urinary NGF/Cr and HB-EGF/Cr levels were compared between controls and patients with OAB at different times following treatment. The differences in urinary NGF/Cr and HB-EGF/Cr levels were also compared between OAB-dry and OAB-wet groups. A repeated measures analysis was performed using a linear mixed model with post hoc analysis of OAB patients who underwent repeated measurements of NGF/Cr and HB-EGF/Cr levels at baseline and 1 and 4 months. p-values were adjusted using the Bonferroni correction. Changes in UDS parameters were evaluated by a McNemar's test and paired t-test. Correlation analysis with Pearson's correlation coefficient was used to assess associations between urinary NGF/Cr and HB-EGF/Cr. OABSS and OAB q scores in OAB patients were compared between those at baseline and those at 4 months after treatment. A paired t-test was used for intra-group statistical analysis. Differences in urinary markers and symptomatic scores for intragroup were also assessed by correlation analysis. Data were analyzed using Statistical Analysis System software (version 9.2, SAS, Cary, NC, USA); all p-values <0.05 were considered statistically significant.
Mean ages were not statistically different between the OAB-wet (60.4±11.2 years) and OAB-dry (56.4±12.3 years) groups; the controls were younger (52.9±8.8 years), however. Table 1 shows the initial urinary levels of NGF/Cr and HB-EGF/Cr in the OAB and control subjects.
In patients with OAB, we checked the urinary levels of NGF, HB-EGF, NGF/Cr, and HB-EGF/Cr at baseline and at 1 and 4 months after beginning fesoterodine therapy. The control patients underwent these measurements only twice since equivalent values were observed at 4 months. Urinary levels of NGF/Cr and HB-EGF/Cr were significantly higher in OAB patients than in controls, at baseline [1.13±0.90 (OAB) vs. 0.50±0.29 (Control) pg/mg; 8.73±6.53 (OAB) vs. 4.45±2.93 (Control) pg/mg; p<0.001 for both comparisons]. After continuing fesoterodine treatment for 4 months, urinary levels of NGF/Cr and HB-EGF/Cr significantly decreased in patients with OAB [0.70±0.57 (1 month), 0.60±0.40 (4 months), p=0.02; 5.76±4.24 (1 months), 4.72±2.69 (4 months) pg/mg, respectively, p=0.03] (Fig. 1). In addition, higher mean urinary levels of NGF/Cr and HB-EGF/Cr were observed in OAB wet patients than in OAB dry patients at baseline [1.26±1.07 (OAB wet), 0.91±0.44 (OAB dry); 9.4±7.73 (OAB wet), 7.59±3.68 (OAB dry) pg/mg, p<0.001]. Mean bladder capacity also significantly increased (p=0.037) in OAB patients after 4 months of fesoterodine treatment; DO decreased by half (Table 2). In addition, Table 3 showed a strong correlation between urinary NGF/Cr and HB-EGF/Cr levels from baseline to 4 months after treatment.
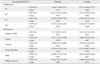
The secondary outcomes (i.e., OABSS and OAB q scores) also changed from baseline to 4 months after treatment in the OAB group. The mean OABSS and OAB q scores changed significantly (p<0.001); more specifically, the post-treatment scores for the questions, "How many times do you typically urinate after waking in the morning until you go to sleep?", "How many times do you typically wake up to urinate from the time you go to sleep at night until you awaken in the morning?", "How often do you have a sudden desire to urinate, which is difficult to defer?", and "How often do you leak urine because you cannot defer your sudden urge to urinate?" were lower than their corresponding pretreatment scores (p<0.001, for all comparisons) (Fig. 2A). Thus, patients reported less urinary frequency and nocturia (p<0.001) after fesoterodine therapy. Urgency and urgency incontinence, as measured by OABSS, also decreased after anticholinergic treatment (p<0.001, for both comparisons). In addition, OAB q scores, including Symptom Bother and the four HRQL subscales, improved after anticholinergic treatment (p<0.001) (Fig. 2B). Mean OABSS and OAB q scores in patients significantly improved, whereas correlation analyses between urinary markers and symptomatic scores in patients were not statistically meaningful; in detail, NGF/Cr level exhibited a weak correlation with the symptom bother domain included in the OAB q scale at baseline (r=0.355, p=0.006). HB-EGF/Cr level was also related to Q2, "How many times do you typically wake up to urinate from the time you fall asleep at night until you awaken in the morning?" in the OABSS questionnaire (r=-0.261, p=0.046). However, the correlation analysis between these urinary makers and clinical symptomatic scales was not statistically significant at 16 weeks (Table 4 and 5).
This study identified specific uses for previously reported OAB and PBS/IC urinary biomarkers, NGF and HB-EGF, in patients with OAB. Urinary levels of NGF have proven to be a useful marker in patients with OAB; however, urinary HB-EGF levels in OAB patients undergoing fixed-dose fesoterodine treatment have not been reported.5,6,7 We simultaneously evaluated the urinary levels of both HB-EGF and NGF to verify their effectiveness as potential markers of urinary HB-EGF. We found that urinary levels of both NGF/Cr and HB-EGF/Cr were higher in OAB patients than in controls. After 4 months of anticholinergic therapy, both NGF/Cr and HB-EGF/Cr levels decreased to normal. A previous study reported that urinary NGF levels commonly increased in OAB and PBS/IC patients, whereas HB-EGF levels were decreased in patients with PBS/IC.15,16 Overall, our findings demonstrated that urinary levels of NGF/Cr and HB-EGF/Cr may be useful biomarkers for differentiating between OAB and PBS/IC.
HB-EGF, as an autocrine regulator of the growth of human bladder epithelial cells, has been proven to antagonize APF via activation of the mitogen-activated protein kinase pathway.9 A defective glycosaminoglycan layer and a permeable urothelium allow urinary potassium to depolarize nerves in the urothelium, detrusor, and perivesical area, leading to urinary urgency and frequency.10 We consider that this mechanism might also be related to OAB symptoms and have a keen interest in studying urinary HB-EGF levels in OAB patients because of its potential use as a biomarker; hence, this study evaluated HB-EGF levels adjusted to the Cr level. This Cr correction for urinary levels of biomarkers is routinely used in urine proteomics and has been considered valid for studies involving measurement of urinary proteins that are believed to be passively released into urine from the urothelium during the storage phase of micturition.17,18 In this study, we discerned that elevation of urinary HB-EGF against increased urinary NGF was a defense mechanism in bladder epithelium of patients with OAB. The protective urothelial barrier was disrupted with broken defense mechanism in PBS/IC, while the restoration pathway worked normally in OAB.
A previous study by Liu and Kuo,5 reported that urinary levels of NGF/Cr were higher in OAB wet patients than in OAB dry patients. We also found higher mean urinary levels of NGF/Cr and HB-EGF/Cr in OAB wet patients than in OAB dry patients. A possible reason for these differences of both urinary markers between OAB dry and OAB wet is strongly associated with urgency symptoms and that higher levels of NGF/Cr and HB-EGF/Cr might impact the occurrence of DO. In a previous study, a higher incidence of DO was shown to be related to urgency incontinence in OAB wet but not OAB dry patients.5 We suggest that elevated NGF concurrently stimulates HB-EGF in relation to the protective mechanism of irritation. These observations indicate that both NGF and HB-EGF are potential biomarkers that reflect the effects of fesoterodine in patients with OAB.
Our secondary outcomes were assessed to evaluate improvements in LUTS in OAB patients. The results presented here showed that 4-mg fesoterodine treatment significantly improves OABSS and OAB q scores, as well as the OAB subjects' assessment of their bladder-related problems, symptoms bother, and their overall HRQL. However, the relevance between urinary markers and OAB symptoms was not significantly different from our expectations.
This is the first report to describe the effects of fixed-dose fesoterodine on LUTS and both urinary biomarkers in patients with OAB. The obvious advantage of the use of noninvasive urinary biomarkers is tempered by the increased variability that arises due to intra- and inter-individual changes in urine volume, pH, and other urine constituents.19,20 Urinary creatinine concentrations are influenced by age, gender, race/ethnicity, diet, and the specific time of urine collection. We controlled for those factors and the effects of different therapeutic regimens by adjusting for Cr concentrations and using only a fixed-dosing regimen. The fesoterodine dose was not increased in any of the subjects in this study, as the outcome improvements associated with escalated doses are controversial.21,22 Our study also employed specific standardized questionnaires to assess OAB symptoms in a well-defined group of women.
The limitation of this study is that patients in the OAB population were older than the controls. Second, we cannot suggest standardize measuring methods and cut-off values of our urinary markers. It is not yet established standards of measuring methods and cut-off value of various urinary markers in OAB patients in spite of previous studies.5,6,23 Our study did not verify sensitivity and specificity, because it compared levels of these markers between controls and OAB patients only. A future study with more broad-spectrum patients and more standard criteria for these urinary markers would strengthen the results.
In conclusion, urinary levels of NGF and HB-EGF may be potential biomarkers for evaluating OAB therapeutic results. Statistically significant improvements in OAB symptoms were seen in OAB patients after fixed-dose fesoterodine treatment. Although this study was limited in that we failed to detect a meaningful relationship between urinary markers and symptomatic scores, it provides useful conclusions and suggestions for future study.
Figures and Tables
 | Fig. 1Changes in the urinary levels of nerve growth factor/creatinine (NGF/Cr) and heparin-binding epidermal growth factor-like growth factor/creatinine (HB-EGF/Cr) in patients with overactive bladder (OAB) syndrome at baseline and 1 and 4 months after treatment and in the control patients at baseline and 1 month after surgery (mean±SD). A post hoc analysis was used to test statistical significance (A), (B) urinary NGF/Cr and HB-EGF/Cr levels in OAB patients. Group 1: OAB patients, Group 2: controls. *p<0.05. |
 | Fig. 2Symptomatic changes in patients with overactive bladder (OAB) syndrome are shown at baseline and after 4 months of fesoterodine treatment. (A) Mean Overactive Bladder symptomatic score (OABSS) with standard deviation (SD) for the four questions. (B) Mean Overactive Bladder Questionnaire (OAB q) scores with SD for total health-related quality of life (HRQL), the four HRQL domains, and symptom bother. A positive score change in total HRQL and its domains indicates improvement; a negative score change in symptom bother indicates improvement (*p<0.001 vs. baseline). |
Table 1
Putative Biomarker Levels in Patients with Overactive Bladder Syndrome and Controls at Baseline (Mean±SD)

| Control (n=62) | OAB (n=62) | p value | |
|---|---|---|---|
| Age (yr) | 52.85±8.78 | 58.89±11.72 | 0.002* |
| Urinary NGF/Cr (pg/mg) | 0.50±0.29 | 1.13±0.9 | <0.001* |
| Urinary HB-EGF/Cr (pg/mg) | 4.45±2.93 | 8.73±6.55 | <0.001* |
Table 2
Changes in Parameters during a Urodynamic Study of Patients with Overactive Bladder Syndrome (n=62)

Table 3
Significant Associations between Urinary NGF/Cr and HB-EGF/Cr in OAB Patients

| Urinary NGF/Cr and HB-EGF/Cr | r† | 95% CI | p value |
|---|---|---|---|
| Baseline | 0.848 | 0.798, 0.925 | <0.001* |
| 1 month after treatment | 0.951 | 0.875, 0.983 | <0.001* |
| 4 months after treatment | 0.949 | 0.608, 0.903 | 0.004* |
Table 4
The Correlation of Both Urinary Markers with Urinary Symptoms and Quality of Life Via Overactive Bladder Symptom Score (OABSS) and Overactive Bladder Questionnaire (OAB q) Scores in Patients with Overactive Bladder Syndrome after Antimuscarinic Treatment: NGF/Cr Level with OABSS and OAB q Scores
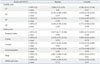
Table 5
The Correlation of Both Urinary Markers with Urinary Symptoms and Quality of Life Via Overactive Bladder Symptom Score (OABSS) and Overactive Bladder Questionnaire (OAB q) Scores in Patients with Overactive Bladder Syndrome after Antimuscarinic Treatment: HB-EGF/Cr Level with OABSS and OAB q Scores
ACKNOWLEDGEMENTS
This study was supported by a faculty research grant of Yonsei University College of Medicine in 2010 (6-2010-0115).
References
1. Irwin DE, Milsom I, Hunskaar S, Reilly K, Kopp Z, Herschorn S, et al. Population-based survey of urinary incontinence, overactive bladder, and other lower urinary tract symptoms in five countries: results of the EPIC study. Eur Urol. 2006; 50:1306–1314.

2. Abrams P, Kelleher CJ, Kerr LA, Rogers RG. Overactive bladder significantly affects quality of life. Am J Manag Care. 2000; 6:11 Suppl. S580–S590.
3. Hanno P, Dmochowski R. Status of international consensus on interstitial cystitis/bladder pain syndrome/painful bladder syndrome: 2008 snapshot. Neurourol Urodyn. 2009; 28:274–286.

4. Rodríguez MA, Afari N, Buchwald DS. National Institute of Diabetes and Digestive and Kidney Diseases Working Group on Urological Chronic Pelvic Pain. Evidence for overlap between urological and nonurological unexplained clinical conditions. J Urol. 2009; 182:2123–2131.

5. Liu HT, Kuo HC. Urinary nerve growth factor level could be a potential biomarker for diagnosis of overactive bladder. J Urol. 2008; 179:2270–2274.

6. Liu HT, Kuo HC. Urinary nerve growth factor levels are increased in patients with bladder outlet obstruction with overactive bladder symptoms and reduced after successful medical treatment. Urology. 2008; 72:104–108.

7. Steers WD, Kolbeck S, Creedon D, Tuttle JB. Nerve growth factor in the urinary bladder of the adult regulates neuronal form and function. J Clin Invest. 1991; 88:1709–1715.

8. Keay SK, Zhang CO, Shoenfelt J, Erickson DR, Whitmore K, Warren JW, et al. Sensitivity and specificity of antiproliferative factor, heparin-binding epidermal growth factor-like growth factor, and epidermal growth factor as urine markers for interstitial cystitis. Urology. 2001; 57:6 Suppl 1. 9–14.

9. Kim J, Keay SK, Freeman MR. Heparin-binding epidermal growth factor-like growth factor functionally antagonizes interstitial cystitis antiproliferative factor via mitogen-activated protein kinase pathway activation. BJU Int. 2009; 103:541–546.

10. Parsons CL, Greenberger M, Gabal L, Bidair M, Barme G. The role of urinary potassium in the pathogenesis and diagnosis of interstitial cystitis. J Urol. 1998; 159:1862–1866.

11. Abrams P, Cardozo L, Khoury S, Wein A. Incontinence. 4th International Consultation on Incontinence. Paris: Health Publication Ltd.;2009.
12. Homma Y, Yoshida M, Seki N, Yokoyama O, Kakizaki H, Gotoh M, et al. Symptom assessment tool for overactive bladder syndrome--overactive bladder symptom score. Urology. 2006; 68:318–323.

13. Coyne K, Revicki D, Hunt T, Corey R, Stewart W, Bentkover J, et al. Psychometric validation of an overactive bladder symptom and health-related quality of life questionnaire: the OAB-q. Qual Life Res. 2002; 11:563–574.
14. Keay S, Zhang CO, Kagen DI, Hise MK, Jacobs SC, Hebel JR, et al. Concentrations of specific epithelial growth factors in the urine of interstitial cystitis patients and controls. J Urol. 1997; 158:1983–1988.

15. Jacobs BL, Smaldone MC, Tyagi V, Philips BJ, Jackman SV, Leng WW, et al. Increased nerve growth factor in neurogenic overactive bladder and interstitial cystitis patients. Can J Urol. 2010; 17:4989–4994.
16. Erickson DR, Tomaszewski JE, Kunselman AR, Stetter CM, Peters KM, Rovner ES, et al. Urine markers do not predict biopsy findings or presence of bladder ulcers in interstitial cystitis/painful bladder syndrome. J Urol. 2008; 179:1850–1856.

17. Goto T, Morioka J, Inamura H, Yano M, Kodaira K, Igarashi Y, et al. Urinary eosinophil-derived neurotoxin concentrations in patients with atopic dermatitis: a useful clinical marker for disease activity. Allergol Int. 2007; 56:433–438.

18. Zimmerli LU, Schiffer E, Zürbig P, Good DM, Kellmann M, Mouls L, et al. Urinary proteomic biomarkers in coronary artery disease. Mol Cell Proteomics. 2008; 7:290–298.

19. Good DM, Thongboonkerd V, Novak J, Bascands JL, Schanstra JP, Coon JJ, et al. Body fluid proteomics for biomarker discovery: lessons from the past hold the key to success in the future. J Proteome Res. 2007; 6:4549–4555.

20. Candela JV, Park E, Gerspach JM, Davidoff R, Stout L, Levy SM, et al. Evaluation of urinary IL-1alpha and IL-1beta in gravid females and patients with bacterial cystitis and microscopic hematuria. Urol Res. 1998; 26:175–180.

21. Khullar V, Rovner ES, Dmochowski R, Nitti V, Wang J, Guan Z. Fesoterodine dose response in subjects with overactive bladder syndrome. Urology. 2008; 71:839–843.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download