Abstract
Purpose
Cutaneous lymphocyte-associated antigen (CLA)-expressing CD8+T cells have been known to play an important role in the pathogenesis of atopic dermatitis (AD). However, the mechanisms underlying the loss of self-tolerance remain unclear. Regulatory T cells (Tregs) play a key role in the development of homeostasis in the immune system. We, therefore, hypothesized that a reduced ability of Tregs to inhibit autologous CD8+CLA+T cells might be underlying mechanism in AD.
Materials and Methods
CD8+CLA+T cells and Tregs were obtained from the peripheral blood of AD patients and control volunteers. The frequencies of CD8+CLA+T cells were evaluated. The proliferative responses of CD8+CLA+T cells were assessed by flow cytometry, and the levels of transforming growth factor-β1 (TGF-β1) and interleukin-10 (IL-10) in culture supernatants were detected by enzyme-linked immunosorbent assay.
Results
Our results revealed higher frequency and increased expression of perforin and granzyme-B in peripheral CD8+CLA+T cells in AD, and lower inhibitory ability of Tregs on proliferation of CD8+CLA+T cells in AD. Meanwhile, the levels of TGF-β1 produced by Tregs were significantly lower in AD, and anti-TGF-β1 abolished such suppression.
Atopic dermatitis (AD) is a T-cell-mediated highly pruritic chronic inflammatory skin disease.1 Recent findings have suggested that CD8+T cells are required for the development of AD lesion.2,3 Cutaneous lymphocyte-associated antigen (CLA) is an important skin-homing receptor displayed by memory/effector T cells which recognize skin-related allergens.4 In peripheral blood, CD8+subsets of CLA+CD45RO+T cells in AD are in an activated state, and the CD8+CLA+ memory/effector T cells are capable of responding to superantigenic stimulation.5 Furthermore, increased frequencies of CLA-positive CD8+T cells producing type 2 cytokines have been detected in the blood of patients with AD.6 However, the mechanisms involved in the induction and activation of CD8+CLA+T cells in AD are not clear.
Regulatory T cells (Tregs) play a crucial role in the maintenance of immunologic-self-tolerance via dominant control of self-reactive T cells.7 It has been suggested that Tregs are able to suppress the proliferation of effector T cells in vitro and to inhibit the development and progression of systemic T cell-mediated autoimmune disorders in vivo; Foxp3-infected T cells can be successfully used to treat autoimmune and inflammatory disorders in mice.8,9,10 In AD, a dysregulation of disease-causing effector T cells is observed in skin biopsies, in association with an impaired Tregs infiltration.11 In contrary, however, the presence of Tregs in infiammatory lesions in AD has also been observed,12,13,14 suggesting the role of Tregs in the AD pathogenesis. Therefore, we hypothesized that an imbalance between activated CD8+CLA+T cells and Tregs exists in peripheral blood of patients with AD. However, the basic mechanisms of suppression mediated by Tregs in AD have not been reported. The purpose of this study is to analyze the inhibition ability of peripheral Tregs on autologous CD8+CLA+T cells in AD, and investigate the mechanisms involved.
In this study, thirty patients (median age 25.6 years; 16 female, 14 male) with history of AD of Yidu Central Hospital of Weifang and 25 healthy controls, age- and gender-matched, were enrolled in the study. AD was diagnosed according to the criteria proposed by Hanifin and Rajka.15 All volunteers had no other autoimmune-associated disorders and no medications had been used for at least 4 weeks prior to the study. All participants signed informed consent and the study was approved by the Ethics Committee.
The following monoclonal antibodies were used for flow cytometry experiments: anti-CD3-APC, anti-CD8-PerCP, anti-CLA-FITC, anti-CD4-FITC, anti-CD25-PECY7, FOXP3-APC, purified anti-CD3 NA/LE and anti-CD28 NA/LE (anti-CD3/CD28), and FACSCalibur Flow Cytometer, all of which were obtained from BD Biosciences (Franklin Lakes, NJ, USA). CD8+T Cell Isolation Kit, anti-CLA MicroBead Kit, CD4+CD25+Regulatory T Cell Isolation Kit, 5-(and 6)-carboxyfluorescein diacetate succinimidyl ester (CFSE), MS/LD Columns, and MiniMACS separator were purchased from Miltenyi Biotec (Cologne, Germany). Biotin-conjugated anti-transforming growth factor-β1 (anti-TGF-β1) or biotin-conjugated chicken IgG for a negative control were from Calbiochem (Darmstadt, Germany). RPMI 1640 medium, 100 U/mL penicillin G and 100 µg/mL streptomycin were purchased from Invitrogen (Carlsbad, CA, USA).
Peripheral blood samples were drawn in ethylene diamine tetraacetic acid (EDTA)-treated tubes, and the samples were processed within 24 h after collection for flow cytometry. Briefly, for the surface staining, appropriate blood was incubated for 15 min in the dark with the following antibodies: anti-CD3, anti-CD8, and anti-CLA. A lysin solution was then added to the tubes. After 10-min incubation, the cells were centrifuged, and the supernatants were discarded. The remainder was then washed in phosphate buffered saline (PBS), the cells were centrifuged again, and the supernatant was discarded. Next, the cells were re-suspended in PBS and were analysed immediately. For intracellular labeling of perforin or granzyme-B, appropriate blood was incubated for 15 min in the dark with the following antibodies: anti-CD3, anti-CD8, and anti-CLA. Then, Reagent A was added to the tubes and incubated for 5 min according to the manufacturer's protocol of IntraSure Kit. Next, lysing solution was added and incubated for 10 min. The mixture was centrifuged and supernatants were then discarded. Reagent B and anti-perforin or anti-granzyme-B were added to the tubes and incubated for 15 min. PBS was added to wash the cells, the cells were centrifuged again and the supernatant discarded. Lastly, the cells were re-suspended in PBS and were analysed immediately. Isotype-matched controls were used for non-specific staining and analysis gates. Data were collected and analyzed.
Peripheral blood mononuclear cells (PBMCs) were isolated on Ficoll-Hypaque gradient. CD8+CLA+T cells were isolated by a two-step procedure; CD8+T cells were purified by negtive selection (using CD8+T Cell Isolation Kit), and then CLA+T cells were isolated by positive selection (using anti-CLA MicroBead Kit) on the MidiMACS magnetic separation system, according to the manufacturer's instructions, with final purities >93%. Tregs were isolated by a two-step procedure using CD4+CD25+ Regulatory T Cell Isolation Kit, according to the manufacturer's instructions, and the purities were confirmed >95% (Fig. 1).
For the proliferation assay, freshly isolated CD8+CLA+T cells or Tregs were labeled with CFSE immediately. Briefly, cells were resuspended in pre-warmed (37℃) PBS/0.1% albumin from bovine serum (BSA) at a final concentration of 1×106 cells/mL, and CFSE solution was added for a final working concentration of 2.5 µM. Cells were incubated at 37℃ for 10 min. Then, staining was quenched by adding 5 volumes of ice-cold culture media (RPMI 1640 supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin G and 100 µg/mL streptomycin) and the cells were incubated on ice for 5 min. Cells were then washed 3 times and resuspended in the culture media.
CFSE labeled cells were plated in 96-well U-bottom plates, precoated overnight with 5 µg/mL anti-CD3 NA/LE and 5 µg/mL anti-CD28 NA/LE (anti-CD3/CD28), at 4℃. CFSE labeled CD8+CLA+T cells were divided into two groups; namely, cells cultured in the absence (5×104 cells/well) and presence (5×104 cells/well) of unlabeled autologous Tregs. After incubation for 4 days, the cells were harvested and washed in PBS containing 0.5 mM EDTA. CFSE labeled cells were resuspended and analyzed on the Flow Cytometer. According to the fluorescence intensity of CFSE, the proliferating (CFSEDIM) and non-proliferating (CFSEBRIGHT) populations of T cells were gated.
PBMCs obtained from 30 AD patients and 25 normal controls were analyzed for the frequencies of CD8+CLA+T cells and the expression of perforin and granzyme-B by flow cytometry. There was significantly higher number of peripheral CD8+CLA+T cells in the AD group than the control group (p=0.000) (Fig. 2). Furthermore, the expression levels of perforin on CD8+CLA+T cells were significantly higher in the AD group than the control group (p=0.003). Similarly, the expression levels of granzyme-B on CD8+CLA+T cells were also significantly higher in the AD group than the control group (p=0.001) (Fig. 3).
CD8+CLA+T cells from either AD patients (n=8) or healthy volunteers (n=8) were labelled with CFSE and stimulated with anti-CD3/CD28 in the absence of Tregs for 4 days. Flow cytometric analysis of CFSE intensity revealed no significant difference in the percentages of proliferating CD8+CLA+T cells between the two groups (Fig. 4).
Our preliminary analysis showed no significant difference in the frequencies of Tregs between the two groups (data not shown): Tregs were confirmed not to proliferate after stimulation with anti-CD3/CD28. Therefore, CFSE labeled CD8+CLA+T cells from either 8 AD patients or 8 normal control subjects were stimulated as described above and co-cultured with autologous Tregs. Four days later, the percentages of proliferating CD8+CLA+T cells were analyzed and found to be significantly higher in the AD group than the control group (Fig. 5). Furthermore, Tregs showed an attenuated suppressive function on the proliferation of CD8+CLA+T cells in the AD group.
We determined the ability of CD8+CLA+T cells and Tregs to produce cytokines under the above established condition 4 days later. Results showed that CD8+CLA+T cells stimulated with anti-CD3/CD28 alone secreted undetectable amounts of TGF-β1 and only minimal amounts of IL-10. However, in the cocultures of Tregs and CD8+CLA+T cells, Tregs could produce high levels of TGF-β1 and IL-10 in both groups. Nevertheless, the levels of TGF-β1 were significantly lower in the AD group than the control group (p=0.025), whereas the levels of IL-10 were not significantly different between the two groups (Fig. 6), demonstrating that the levels of TGF-β1 produced by Tregs in the cocultures were decreased in AD.
We further investigated whether such production mediates immunosuppression: whether Tregs mediate immunosuppression in the presence or absence of anti-TGF-β1. Fig. 7 shows that, in cultures containing 50 µg/mL of anti-TGF-β1, the Tregs-mediated suppression decreased and the CD8+CLA+T cells showed increased proliferation. More importantly, in the cultures containing 100 µg/mL of anti-TGF-β1, Tregs-mediated suppression was greatly weakened, while the percentages of proliferating CD8+CLA+T cells were further increased, with no significant difference between the two groups. These results suggest that the suppression of CD8+CLA+T cells proliferation by Tregs is mediated, at least in part, by TGF-β1 in AD.
It has been proven that CD8+CLA+T cells play an important role in immune-related diseases, such as psoriasis and alopecia areata.16,17 Recently, Hennino, et al.2,3 showed that CD8+T cells are recruited early to allergen exposure sites in atopy patch test reactions in human atopic dermatitis and allergen-primed CD8+T cells initiate atopic dermatitis lesions. Furthermore, CLA+T cells isolated from peripheral blood of atopic individuals were shown to contain and spontaneously release cytokines IL-4 and IL-13,18,19 and staphylococcal enterotoxin B treatment of atopic PBMC in vitro resulted in up-regulation of CLA expression on T cells, thereby facilitating the homing of T cells in the AD skin.20 Akdis, et al.5 demonstrated that CLA+CD8+T cells isolated from the skin or peripheral blood of AD patients responded to superantigenic stimulation to the same extent as CD4+T cells: they spontaneously proliferated ex vivo, secreting high levels of IL-5 and IL-13, and therefore, were capable of preventing spontaneous eosinophil apoptosis and enhancing IgE. In the present study, we showed that patients with AD have a higher frequency of CD8+CLA+T cells and increased expression of perforin and granzyme-B. Similarly, Yawalkar, et al.21 found that perforin and granzyme-B are cytotoxic molecules produced by activated cytotoxic CD8+T cells, which are strongly expressed in AD lesional skin. Moreover, CD8+granzyme-B+T cells infiltrating the epidermis are associated with the development of positive atopy patch test.3 These results together with the present study are consistent with the hypothesis that these hyper-activated cytotoxic T cells are responsible for the apoptosis of keratinocytes and for the epidermal spongiosis, which are both pathological hallmarks of AD.22,23
The functional hallmark of Tregs is their remarkable capacity to suppress T effector/memory (Teff/mem) cell activation including T-cell proliferation.24 Recent reviews have updated the idea that Tregs can inhibit activated T cells to maintain peripheral tolerance.25,26 The suppressive capacity of Tregs is diminished in patients with autoimmune diseases such as psoriasis27 and bullous pemphigoid,28 and Tregs showed a dysfunction to CD4+CD25-T cells27 or CD8+T cells in theses diseases.29 Considering the fact that CD8+CLA+T cells are hyper-activated in AD, to assay the suppressive function of Tregs on CD8+CLA+T cells appears to be highly significant. Our study showed that proliferative responses of peripheral CD8+CLA+T cells in the absence of autologous Tregs stimulated with anti-CD3/CD28 had no significant difference between AD and control groups. However, after co-cultured with autologous Tregs, proliferative responses of CD8+CLA+T cells were significantly higher in the AD group, demonstrating that the suppressive effect of Tregs on CD8+CLA+T cells was decreased in AD patients. On the other hand, however, there is a possibility that the CD8+CLA+T cells are resistant to the suppression in AD patients, and crisscross experiments are required to rule it out (e.g., to assay the proliferation of CD8+CLA+T cells of a patient with AD in the presence of Tregs of healthy donor). However, it is difficult to perform such experiments and beyond the scope of the present study.
In many model systems, the cell contact-dependent immunosuppression by Tregs is mediated by inhibitory cytokines including TGF-β and IL-10.30 Two major modes of action exist for these cytokines to promote Tregs suppressive functions. First, these cytokines can directly inhibit activation and/or survival of Teff cells themselves, thereby dampening autoreactive Teff cell activation in the setting of autoimmunity. Secondly, these cytokines can also generate peripherally derived Tregs and contribute to the peripheral homeostasis and survival of these cells.31 Nakamura, et al.32 found that Tregs exert immunosuppression by cell-cell interaction and via presentation of TGF-β to TGF-βR on target cells. Similarly, we found that the TGF-β1 levels in culture supernatants in the presence of anti-CD3/CD28 were decreased in AD, suggesting that TGF-β1 may play a role in the pathogenesis of AD. Furthermore, our present data showed that Tregs-mediated suppression was weakened in the presence of anti-TGF-β1 dose-dependently, and the difference in the proliferation of CD8+CLA+T cells co-cultured with Tregs between the two groups was abolished in the presence of 100 µg/mL of anti-TGF-β1. These results suggest that the suppressive function of Tregs in AD is mediated, at least in part, by TGF-β1.
In summary, our results reveal hyper-activated CD8+CLA+T cells and lower inhibitory ability of Tregs in AD, which are mediated by TGF-β1, and support the hypothesis that attenuated suppressive function of Tregs on autologous CD8+CLA+T cells plays an important role in the pathogenesis of AD.
Figures and Tables
Fig. 1
The purities of isolated Tregs (CD4+CD25+T cells). PBMCs were isolated on Ficoll-Hypaque gradient. According to the manufacturer's instructions of CD4+CD25+ Regulatory T Cell Isolation Kit, CD4+T cells were purified by negtive selection, and then CD25+T cells were isolated by positive selection. The purities were confirmed >95%. A representative profile of purified Tregs was showed. PBMC, peripheral blood mononuclear cell; FITC, fluorescein isothiocyanate; PE, phycoerythrin.

Fig. 2
Frequencies of peripheral CD8+CLA+T cells. The frequencies of CD8+CLA+T cells in peripheral blood were measured by flow cytometry, and the representative results (A) from AD patients and healthy volunteers were shown by dot plot graphs. (B) The data showed the frequencies of CD8+CLA+T cells were significantly higher in the AD group (n=30) compared to the control group (n=25, *p<0.05). CLA, cutaneous lymphocyte-associated antigen; AD, atopic dermatitis; PE, phycoerythrin.

Fig. 3
Expression levels of cytotoxic molecules (perforin and granzyme-B) on peripheral CD8+CLA+T cells. The percentages of cytotoxic molecules (perforin and granzyme-B) on CD8+CLA+T cells in peripheral blood were measured by flow cytometry, and the representative results (A and C) from AD patients and healthy volunteers were shown by dot plot graphs. (B and D) The data showed the expression levels of both perforin and granzyme-B on CD8+CLA+T cells were significantly higher in the AD group (n=30) compared to the control group (n=25, *p<0.05). CLA, cutaneous lymphocyte-associated antigen; AD, atopic dermatitis; PE, phycoerythrin; FITC, fluorescein isothiocyanate.

Fig. 4
Proliferation of peripheral CD8+CLA+T cells. CFSE-labeled peripheral CD8+CLA+T cells stimulated with anti-CD3/CD28 in the absence of Tregs for 4 days. According to the CFSE intensity by flow cytometry, the percentages of proliferating CD8+CLA+T cells were detected. (A) Representative CFSE profiles from an AD patient and a healthy volunteer were shown. (B) The data showed the percentages of proliferating CD8+CLA+T cells were of no significant difference between the AD group (n=8) and the control group (n=8). CFSE, carboxyfluorescein diacetate succinimidyl ester; CLA, cutaneous lymphocyte-associated antigen; AD, atopic dermatitis.

Fig. 5
Tregs suppress proliferation of autologous CD8+CLA+T cells. CFSE-labeled peripheral CD8+CLA+T cells stimulated with anti-CD3/CD28 in the presence of autologous Tregs for 4 days. According to the CFSE intensity by flow cytometry, the percentages of proliferating CD8+CLA+T cells were detected. (A) Representative CFSE profiles from an AD patient and a healthy volunteer were shown. (B) The data showed the percentages of proliferating CD8+CLA+T cells were significantly higher in the AD group (n=8) compared to the control group (n=8, *p<0.05). (C) The suppressive function of Tregs on the proliferation of autologous CD8+CLA+T cells was significantly lower in the AD group (n=8) compared to the control group (n=8, *p<0.05). CFSE, carboxyfluorescein diacetate succinimidyl ester; Tregs, regulatory T cells; CLA, cutaneous lymphocyte-associated antigen; AD, atopic dermatitis.
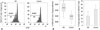
Fig. 6
Levels of TGF-β1 and IL-10 in culture supernatants of Tregs and/or CD8+CLA+T cells. 5×104 Tregs co-cultured with 5×104 autologous CD8+CLA+T cells stimulated with anti-CD3/CD28 in 96-well plates. Four days later, the amount of TGF-β1 (A) and IL-10 (B) in culture supernatant was measured by ELISA. The levels of TGF-β1 produced by Tregs were significantly lower in the AD group (n=8) compared to the control group (n=8, *p<0.05). However, the levels of IL-10 produced by Tregs showed no difference between the two groups. And 5×104 CD8+CLA+T cells stimulated with anti-CD3/CD28 in 96-well plates for 4 days, secreted undetectable amounts of TGF-β1 and minimal amounts of IL-10 in the two groups. CLA, cutaneous lymphocyte-associated antigen; AD, atopic dermatitis; Tregs, regulatory T cells; ELISA, enzyme-linked immunosorbent assay; TGF-β1, transforming growth factor-β1; IL-10, interleukin-10.

Fig. 7
Suppression of CD8+CLA+T cells proliferation by Tregs is mediated by TGF-β1. 5×104 Tregs co-cultured with 5×104 autologous CD8+CLA+T cells from AD patients (n=8, gray bars) or healthy volunteers (n=8, white bars) stimulated with anti-CD3/CD28 in 96-well plates in the presence of anti-TGF-β1 or control IgG. Four days later, the percentages of proliferating CD8+CLA+T cells were detected. The CD8+CLA+T cells exhibited increased proliferation in the presence of 50 µg/mL of anti-TGF-β1, and their proliferation were further increased and of no significant difference between the two groups in the presence of 100 µg/mL of anti-TGF-β1. Tregs, regulatory T cells; CLA, cutaneous lymphocyte-associated antigen; AD, atopic dermatitis; TGF-β1, transforming growth factor-β1.

References
2. Hennino A, Jean-Decoster C, Giordano-Labadie F, Debeer S, Vanbervliet B, Rozières A, et al. CD8+ T cells are recruited early to allergen exposure sites in atopy patch test reactions in human atopic dermatitis. J Allergy Clin Immunol. 2011; 127:1064–1067.

3. Hennino A, Vocanson M, Toussaint Y, Rodet K, Benetière J, Schmitt AM, et al. Skin-infiltrating CD8+ T cells initiate atopic dermatitis lesions. J Immunol. 2007; 178:5571–5577.

4. Sallusto F, Lenig D, Förster R, Lipp M, Lanzavecchia A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature. 1999; 401:708–712.

5. Akdis M, Simon HU, Weigl L, Kreyden O, Blaser K, Akdis CA. Skin homing (cutaneous lymphocyte-associated antigen-positive) CD8+ T cells respond to superantigen and contribute to eosinophilia and IgE production in atopic dermatitis. J Immunol. 1999; 163:466–475.
6. Teraki Y, Hotta T, Shiohara T. Increased circulating skin-homing cutaneous lymphocyte-associated antigen (CLA)+ type 2 cytokine-producing cells, and decreased CLA+ type 1 cytokine-producing cells in atopic dermatitis. Br J Dermatol. 2000; 143:373–378.

7. Sakaguchi S. Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu Rev Immunol. 2004; 22:531–562.
8. Loser K, Hansen W, Apelt J, Balkow S, Buer J, Beissert S. In vitro-generated regulatory T cells induced by Foxp3-retrovirus infection control murine contact allergy and systemic autoimmunity. Gene Ther. 2005; 12:1294–1304.

9. Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol. 2003; 4:330–336.

10. Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell development by the transcription factor Foxp3. Science. 2003; 299:1057–1061.

11. Verhagen J, Akdis M, Traidl-Hoffmann C, Schmid-Grendelmeier P, Hijnen D, Knol EF, et al. Absence of T-regulatory cell expression and function in atopic dermatitis skin. J Allergy Clin Immunol. 2006; 117:176–183.

12. Caproni M, Antiga E, Torchia D, Volpi W, Barletta E, Gitti G, et al. FoxP3-expressing T regulatory cells in atopic dermatitis lesions. Allergy Asthma Proc. 2007; 28:525–528.

13. Schnopp C, Rad R, Weidinger A, Weidinger S, Ring J, Eberlein B, et al. Fox-P3-positive regulatory T cells are present in the skin of generalized atopic eczema patients and are not particularly affected by medium-dose UVA1 therapy. Photodermatol Photoimmunol Photomed. 2007; 23:81–85.

14. Szegedi A, Baráth S, Nagy G, Szodoray P, Gál M, Sipka S, et al. Regulatory T cells in atopic dermatitis: epidermal dendritic cell clusters may contribute to their local expansion. Br J Dermatol. 2009; 160:984–993.

15. Hanifin JM, Rajka G. Diagnostic features of atopic dermatitis. Acta Derm Venereol (Stockh 92). 1980; Suppl 92. 44–47.
16. Sigmundsdóttir H, Gudjónsson JE, Jónsdóttir I, Lúdvíksson BR, Valdimarsson H. The frequency of CLA+ CD8+ T cells in the blood of psoriasis patients correlates closely with the severity of their disease. Clin Exp Immunol. 2001; 126:365–369.

17. Yano S, Nakamura K, Okochi H, Tamaki K. Analysis of the expression of cutaneous lymphocyte-associated antigen on the peripheral blood and cutaneous lymphocytes of alopecia areata patients. Acta Derm Venereol. 2002; 82:82–85.

18. Santamaria Babi LF, Picker LJ, Perez Soler MT, Drzimalla K, Flohr P, Blaser K, et al. Circulating allergen-reactive T cells from patients with atopic dermatitis and allergic contact dermatitis express the skin-selective homing receptor, the cutaneous lymphocyte-associated antigen. J Exp Med. 1995; 181:1935–1940.

19. Akdis M, Akdis CA, Weigl L, Disch R, Blaser K. Skin-homing, CLA+ memory T cells are activated in atopic dermatitis and regulate IgE by an IL-13-dominated cytokine pattern: IgG4 counter-regulation by CLA- memory T cells. J Immunol. 1997; 159:4611–4619.
20. Leung DY, Gately M, Trumble A, Ferguson-Darnell B, Schlievert PM, Picker LJ. Bacterial superantigens induce T cell expression of the skin-selective homing receptor, the cutaneous lymphocyte-associated antigen, via stimulation of interleukin 12 production. J Exp Med. 1995; 181:747–753.

21. Yawalkar N, Schmid S, Braathen LR, Pichler WJ. Perforin and granzyme B may contribute to skin inflammation in atopic dermatitis and psoriasis. Br J Dermatol. 2001; 144:1133–1139.

22. Pastore S, Mascia F, Girolomoni G. The contribution of keratinocytes to the pathogenesis of atopic dermatitis. Eur J Dermatol. 2006; 16:125–131.
23. Akdis M, Trautmann A, Klunker S, Daigle I, Kucuksezer UC, Deglmann W, et al. T helper (Th) 2 predominance in atopic diseases is due to preferential apoptosis of circulating memory/effector Th1 cells. FASEB J. 2003; 17:1026–1035.

24. Hilchey SP, Bernstein SH. Use of CFSE to monitor ex vivo regulatory T-cell suppression of CD4+ and CD8+ T-cell proliferation within unseparated mononuclear cells from malignant and non-malignant human lymph node biopsies. Immunol Invest. 2007; 36:629–648.

25. Toubi E. The role of CD4+CD25+ T regulatory cells in autoimmune diseases. Clin Rev Allergy Immunol. 2008; 34:338–344.

26. Costantino CM, Baecher-Allan CM, Hafler DA. Human regulatory T cells and autoimmunity. Eur J Immunol. 2008; 38:921–924.

27. Sugiyama H, Gyulai R, Toichi E, Garaczi E, Shimada S, Stevens SR, et al. Dysfunctional blood and target tissue CD4+CD25high regulatory T cells in psoriasis: mechanism underlying unrestrained pathogenic effector T cell proliferation. J Immunol. 2005; 174:164–173.

28. Antiga E, Quaglino P, Volpi W, Pierini I, Del Bianco E, Bianchi B, et al. Regulatory T cells in skin lesions and blood of patients with bullous pemphigoid. J Eur Acad Dermatol Venereol. 2013; [Epub ahead of print].

29. Lili Y, Yi W, Ji Y, Yue S, Weimin S, Ming L. Global activation of CD8+ cytotoxic T lymphocytes correlates with an impairment in regulatory T cells in patients with generalized vitiligo. PLoS One. 2012; 7:e37513.

30. Umetsu DT, Akbari O, Dekruyff RH. Regulatory T cells control the development of allergic disease and asthma. J Allergy Clin Immunol. 2003; 112:480–487.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download