Abstract
Purpose
House dust mites (HDMs) are an important source of indoor allergens associated with asthma, rhinitis and atopic dermatitis. Chicken immunoglobulin (Ig) Y is known to be a good alternative to mice and rabbit antibody production. In this study, we produced IgYs specific to HDMs and investigated their IgE immunoreactivities.
Materials and Methods
Total IgYs were isolated from the yolks of White Leghorn hens immunized with either Dermatophagoides pteronyssinus or D. farinae protein extract. Control antibodies were separated from the yolks of immunized hens with phosphate buffered saline. IgYs specific to HDMs were analyzed using enzyme-linked immunosorbent assay and Western blotting analysis.
Results
The concentration of egg IgY specific to D. farinae in an immunized hen increased and the highest achieved was 661.3 ug/mg (per an egg) on day 47, compared with 760 ug/mg IgY specific to D. pteronyssinus on day 16. The D. pteronyssinus or D. farinae-specific IgY was detected by binding of each mite proteins, and their immunoreactivities were elevated dependent of the specific IgY concentration.
House dust mites (HDMs) are one of the most common aeroallergens worldwide and a crucial trigger for allergic responses (asthma, rhinitis and atopic dermatitis).1,2 Exposure to HDMs in sensitized patients is associated with worse lung function, greater medication requirements, and the development of chronic asthma.3,4,5 Both D. pteronyssinus and D. farinae are two of the most predominant species, the major allergens of which are group 1 allergens: Der p 1 and Der f 1. These allergens have diverse biological functions (e.g., cysteine proteases) that contribute to allergic sensitization and share amino acid sequences with high homology.6,7,8
IgY is the principal known antibody in amphibians, reptiles, and birds.9 In hens, IgY is transported from the blood to the egg yolk, where it accumulates and provides offspring humoral immunity against avian pathogens prior to full maturation of their own immune system.10,11,12 IgY antibody concentrations range from 50 to 100 mg per egg yolk.12 Although chicken IgY shares a common ancestor with both mammalian IgG and IgE, the egg yolk antibodies neither activate the mammalian complement system, nor interact with rheumatoid factors or bacterial and human Fc receptors that could mediate an inflammatory response.11,12,13 In recent studies, chicken egg yolk has been recognized as an alternative antibody source. Immunization with egg yolk immunoglobulin (IgY) has shown diagnostic and therapeutic value against fungi, bacteria and virus causing various diseases.14,15,16,17 For example, passive immunization with egg yolk IgY through intramuscular injection, oral administration, and immersion was shown to have therapeutic value in the treatment of infectious disease evoked by rotavirus, parvovirus, E. coli, S. typhimurium, S. mutans, H. pylori, and Candida albicans.16
In this study, we aimed to produce IgYs, which are specific to D. pteronyssinus and D. farinae, from hens and to examine immunoreactivities for further diagnostic use of HDMs specific IgYs.
HDM (D. pteronyssinus or D. farinae) was pulverized in liquid nitrogen. The fatted samples were then placed in 1:1 ethyl ether/ethyl acetate and extracted with slow stirring at 4℃ for 2 days in phosphate buffered saline (PBS) (pH 7.4). The extract was then centrifuged at 9000×g for 30 minutes at 4℃, and the supernatant was dialyzed overnight and lyophilized. The extract was diluted in 0.01 M potassium phosphate buffer (pH 7.2) containing 0.1 M NaCl and mixed with Freund's complete adjuvant (FCA, Sigma, MO, USA) at a 1:1 ratio. The protein concentration of HDM was determined by Bradford protein assay and the protease activity was measured by QuantiCleave protease assay kit (Pierce, Rockford, IL, USA). The protease activity of D. pteronyssinus and D. farinae were 5 U/mL and 4.78 U/mL (per protein extract 50 ug/mL), respectively.
For the induction of HDM-specific antibodies in egg yolks, 22-week-old White Leghorns were immunized intramuscularly (specifically, in the breast tissue) with a mixture of HDM protein extract and adjuvant under conventional animal room conditions that were mite-free. Booster inoculations consisting of half the initial volume were administered to hens at week 2 and repeated in the same manner every 2 weeks (Group 2-6) (Table 1). The hens of the control group received inoculations in the same manner via a sterile saline solution (PBS). The eggs were harvested from start to finish of the experiment and stored in a refrigerator at 4℃.
Eggs were individually cracked, and the egg white was separated from the yolk. Residual egg white was carefully wiped off using a gauze sheet so as not to break the yolk. The yolk sac was ruptured with a needle and the contents were dripped into a tared beaker. After discarding the yolk sac, the weight of the separated yolk was measured. IgY from the egg yolk was purified using an EGGstract IgY purification kit (Promega, WI, USA). The method used was partly modified from that of Polson, et al.18 using polyethylene glycol and ammonium sulfate precipitation. Then, the precipitated IgYs were dissolved in PBS and stored at -20℃ until analysis.
Each HDM antigen (50 ug/mL per well) was coated previously onto an enzyme-linked immunosorbent assay (ELISA) plate for 16 hours, and diluted (1:500-1:2000) IgY samples and standards were then applied and incubated for 1 hour. Standard IgYs were produced by passing through an affinity column conjugated purified HDM. After washing 3 times, the attached IgYs were reacted with anti-IgY-alkaline phophatase conjugate for 1 hour for colorization (Promega, WI, USA). The absorbance was measured at 405 nm to determine the quantities of IgY specific to HDMs. The unit of IgY specific to HDM (ug/mg) was defined as the value of IgY specific to HDMs divided by the total protein concentration of IgY calculated in every egg, given that the amount of IgY in yolk is independent of egg weight.
Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was performed using reducing and denaturing conditions. Protein extracts from HDMs were loaded onto 12.5% gels and stained with Coomassie blue to visualize constitutive protein bands or for Western blotting analysis. The separated proteins were electro-transferred onto nitrocellulose membranes and blocked with 5% skim milk. These strips were incubated with purified IgY diluted into different ratios (v/v) and then reacted with anti-chicken IgY alkaline phosphate conjugate (Promega, WI, USA) after additional washing. Bands were made visible on the membrane using 5-bromo-4-chloro-3-indolyl-phosphate/nitro blue tetrazolium (Promega, WI, USA) color development substrates.
Extracted proteins of D. pteronyssinus and D. farinae were analyzed by SDS-PAGE. Protein bands less than 36.4 kD were strongly stained with Coomassie blue in HDMs, including Der p 1 (25 kD) and Der f 1 (36 kD), major allergens of HDMs with known proteolytic activity and maximum immunogenicity (Fig. 1).8
The HDM extracts were intramuscularly inoculated in hens 3 times every 2 weeks (Table 1), and eggs were harvested every day. IgYs were purified from egg, and IgYs specific to HDMs were detected. As shown in Fig. 2A, IgY specific to D. pteronyssinus started to increase at day 6 (89.0 ug/mg) after the first immunization at day 1 and rapidly increased from days 14 to 16 (92.8-760.2 ug/mg) when the second injection was carried out at day 14. These results suggest that the first inoculation caused a surge in specific IgYs against D. pteronyssinus. After reaching a peak at day 16 (760.2 ug/mg), however, these IgYs gradually decreased by day 29 (435.0 ug/mg), despite a third immunization at day 28. From day 30 onward, IgY specific to D. pteronyssinus was not significantly distinguished from the control. With the fourth injection, the last peak simultaneously appeared at day 42 (278.3 ug/mg) and immediately dropped toward control levels since day 43 (56.3 ug/mg), indicating the fourth injection did not affect specific IgY productivity. On the other hand, IgYs specific to D. farinae were continuously induced until day 47 (661.3 ug/mg), and an increase in these specific IgYs was detected after each immunization: at day 2 (134.9 ug/mg), 22 (425.9 ug/mg), 39 (517.1 ug/mg) and 47 (661.3 ug/mg) (Fig. 2B). These results suggest that D. farinae was more potent in producing and maintaining high amounts of specific IgY in the yolk after each injection than D. pteronyssinus.
Furthermore, we attempted to visually analyze the immunoreactivities of purified IgYs specifically against HDMs by Western blotting (Fig. 3). The immunoreactivity of HDM protein bands for specific IgYs was dependent on the dilution ratio of IgY, and IgYs from control hens did not react to any HDM protein bands. The IgYs used for Western blotting were chosen based on the highest value of IgY specific to D. pteronyssinus or D. farinae.
In this study, we produced IgY specific to HDM protein from eggs harvested after immunization, and confirmed that the purified specific IgY responded dose-dependently to D. pteronyssinus or D. farinae protein. For six weeks, including four successive sensitizations, antibody titers differed between D. pteronyssinus and D. farinae immunization groups; the anti-D. farinae group continued to produce stable results after an additional booster compared with the anti-D. pteronyssinus group (134.9-661.3 ug/mg) (Fig. 2A). Even though the productivity of anti-D. pteronyssinus group was relatively malleable, however, antibody titers remained very high in the anti-D. pteronyssinus group (40.9-760.2 ug/mg) (Fig. 2B). These findings indicate that both D. pteronyssinus and D. farinae potently induce specific IgYs, suggesting that at least a second immunization should be carried out to induce a maximal antibody level response.
In cases of D. pteronyssinus, over 30 different proteins have been found to induce IgE antibody in HDM allergic patients; 21 of these allergens demonstrated cross-reactivity with allergens from other mite species.19 Although they show immunologic cross-reactivity, D. pteronyssinus and D. farinae typically have 15-20% amino acid sequence disparity and also have unique epitopes.20 Just as anti-inflammatory medicines and immunosuppressive agents may be used to combat inflammation, allergic symptoms to HDM may be relieved by specific immunotherapies.21 Xie reported that oral administration of D. farinae extract significantly reduced goblet cell hyperplasia and eosinophilia in a D. farinae-sensitized allergic rat model.22 However, even though recent studies have emphasized the requirement for specific immunotherapy as an allergen-specific approach, they have not addressed the difficulties encountered in allergen standardization and the risk of inducing life-threatening anaphylactic side effects. Thus, efforts to develop specific antibodies against various HDM allergens absolutely requires testing proposed risk levels and assessing the corresponding airborne allergen levels, which constitute risk factors.23 Furthermore, these antibodies will also be useful in standardizing allergen extracts used for diagnosis and immunotherapy, as well as for comparing the efficacy of allergen avoidance regimes. In addition, this will make it easier to provide information on the relationship between mite allergen levels and the prevalence of allergic disease.24 For these antibodies, the use of chicken IgY instead of mammalian antibodies has numerous advantages, including increased production of antibody levels, decreased animal use, and no need for painful blood collections. The utility of IgY without a loss of specificity and sensitivity has been demonstrated in many immunological assays, and maintaining chickens was more cost-effective compared to other mammalians.12,25 Moreover, chickens from inbred strains are obtainable, thus minimizing genetic variation in antibody responses. Yolk antibodies show increased deposition in the egg during the process of transmission from the maternal line to offspring.26,27,28
In conclusion, we immunized hens with HDMs and confirmed a significant increase in the level of IgYs specific to HDMs. A number of immunoreactive proteins involving major HDM allergens of approximately 9.3 to 115 kD were also detected. Though further work is required to confirm the immunoreactivity of specific IgYs in patient sera sensitized to HDMs, our results suggest that chicken egg yolk antibodies have considerable potential as an alternative antibody source. This could be a major source of research opportunities, including diagnostic analysis and immunotherapy development in the field of allergic disease.
Figures and Tables
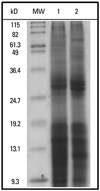 | Fig. 1SDS-PAGE of house dust mite protein extracts. MW, molecular weight marker; lane 1, D. farinae crude extract; lane 2, D. pteronyssinus extract; SDS-PAGE, sodium dodecyl sulfate polyacrylamide gel electrophoresis. |
 | Fig. 2Mean results of IgY specific to HDMs as measured by ELISA following immunization with HDMs. Plates were pre-coated with 50 ug/mL D. pteronyssinus (A) or D. farinae (B) extract. The value of IgY specific to HDMs is expressed as a dotted line and that of the control IgY as a solid line. The above arrows indicate the time of immunization with HDM. |
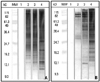 | Fig. 3Western blotting analysis of purified IgY specific against HDMs. The separated D. pteronyssinus protein bands (0.5 ug/lane) (A) and D. farinae protein bands (0.5 ug/lane) (B) were transferred to nitrocellulose membranes. MW, molecular weight marker; lane 1, control IgY diluted into 1:300; lane 2, IgY-HDM diluted into 1:500; lane 3, IgY-HDM diluted into 1:250; lane 4, IgY-HDM diluted into 1:100. |
ACKNOWLEDGEMENTS
This research was supported by Gio-Biotech Inc. and Basic Science Research Program through the National Research foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2011-0024036).
References
1. Gregory LG, Lloyd CM. Orchestrating house dust mite-associated allergy in the lung. Trends Immunol. 2011; 32:402–411.

2. Nahm DH, Kim ME. Treatment of severe atopic dermatitis with a combination of subcutaneous allergen immunotherapy and cyclosporin. Yonsei Med J. 2012; 53:158–163.

3. Trompette A, Divanovic S, Visintin A, Blanchard C, Hegde RS, Madan R, et al. Allergenicity resulting from functional mimicry of a Toll-like receptor complex protein. Nature. 2009; 457:585–588.

4. Nathan AT, Peterson EA, Chakir J, Wills-Karp M. Innate immune responses of airway epithelium to house dust mite are mediated through beta-glucan-dependent pathways. J Allergy Clin Immunol. 2009; 123:612–618.

5. Diette GB, McCormack MC, Hansel NN, Breysse PN, Matsui EC. Environmental issues in managing asthma. Respir Care. 2008; 53:602–615.
6. Yuuki T, Okumura Y, Ando T, Yamakawa H, Suko M, Haida M, et al. Cloning and expression of cDNA coding for the major house dust mite allergen Der f II in Escherichia coli. Agric Biol Chem. 1991; 55:1233–1238.

7. Chua KY, Doyle CR, Simpson RJ, Turner KJ, Stewart GA, Thomas WR. Isolation of cDNA coding for the major mite allergen Der p II by IgE plaque immunoassay. Int Arch Allergy Appl Immunol. 1990; 91:118–123.

8. Zhang J, Saint-Remy JM, Garrod DR, Robinson C. Comparative enzymology of native and recombinant house dust mite allergen Der p 1. Allergy. 2009; 64:469–477.

9. Taylor AI, Gould HJ, Sutton BJ, Calvert RA. Avian IgY binds to a monocyte receptor with IgG-like kinetics despite an IgE-like structure. J Biol Chem. 2008; 283:16384–16390.

10. Chalghoumi R, Théwis A, Portetelle D, Beckers Y. Production of hen egg yolk immunoglobulins simultaneously directed against Salmonella enteritidis and Salmonella typhimurium in the same egg yolk. Poult Sci. 2008; 87:32–40.

11. Warr GW, Magor KE, Higgins DA. IgY: clues to the origins of modern antibodies. Immunol Today. 1995; 16:392–398.

12. Dias da Silva W, Tambourgi DV. IgY: a promising antibody for use in immunodiagnostic and in immunotherapy. Vet Immunol Immunopathol. 2010; 135:173–180.

13. Hädge D, Ambrosius H. Evolution of low molecular weight immunoglobulins-IV. IgY-like immunoglobulins of birds, reptiles and amphibians, precursors of mammalian IgA. Mol Immunol. 1984; 21:699–707.

15. Casadevall A. Antibody immunity and invasive fungal infections. Infect Immun. 1995; 63:4211–4218.

16. Ibrahim el-SM, Rahman AK, Isoda R, Umeda K, Van Sa N, Kodama Y. In vitro and in vivo effectiveness of egg yolk antibody against Candida albicans (anti-CA IgY). Vaccine. 2008; 26:2073–2080.

17. Spillner E, Braren I, Greunke K, Seismann H, Blank S, du Plessis D. Avian IgY antibodies and their recombinant equivalents in research, diagnostics and therapy. Biologicals. 2012; 40:313–322.

18. Polson A, von Wechmar MB, van Regenmortel MH. Isolation of viral IgY antibodies from yolks of immunized hens. Immunol Commun. 1980; 9:475–493.

19. Griffin P, Ford AW, Alterman L, Thompson J, Parkinson C, Blainey AD, et al. Allergenic and antigenic relationship between three species of storage mite and the house dust mite, Dermatophagoides pteronyssinus. J Allergy Clin Immunol. 1989; 84:108–117.

20. Thomas WR, Heinrich TK, Smith WA, Hales BJ. Pyroglyphid house dust mite allergens. Protein Pept Lett. 2007; 14:943–953.

21. Asturias JA, Ibarrola I, Arilla MC, Vidal C, Ferrer A, Gamboa PM, et al. Engineering of major house dust mite allergens Der p 1 and Der p 2 for allergen-specific immunotherapy. Clin Exp Allergy. 2009; 39:1088–1098.

22. Xie QM, Wu X, Wu HM, Deng YM, Zhang SJ, Zhu JP, et al. Oral administration of allergen extracts from Dermatophagoides farinae desensitizes specific allergen-induced inflammation and airway hyperresponsiveness in rats. Int Immunopharmacol. 2008; 8:1639–1645.

23. Luczynska CM, Arruda LK, Platts-Mills TA, Miller JD, Lopez M, Chapman MD. A two-site monoclonal antibody ELISA for the quantification of the major Dermatophagoides spp. allergens, Der p I and Der f I. J Immunol Methods. 1989; 118:227–235.

24. Earle CD, King EM, Tsay A, Pittman K, Saric B, Vailes L, et al. High-throughput fluorescent multiplex array for indoor allergen exposure assessment. J Allergy Clin Immunol. 2007; 119:428–433.

25. Schade R, Calzado EG, Sarmiento R, Chacana PA, Porankiewicz-Asplund J, Terzolo HR. Chicken egg yolk antibodies (IgY-technology): a review of progress in production and use in research and human and veterinary medicine. Altern Lab Anim. 2005; 33:129–154.

26. Meenatchisundaram S, Parameswari G, Michael A, Ramalingam S. Neutralization of the pharmacological effects of Cobra and Krait venoms by chicken egg yolk antibodies. Toxicon. 2008; 52:221–227.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download