Abstract
Purpose
Thyroid cancer is the most common malignancy in Korean females and can be treated with good prognosis. However, drugs to treat aggressive types of thyroid cancer such as poorly differentiated or anaplastic thyroid cancer have not yet been established. To that end, we analyzed the effects of berberine on human thyroid cancer cell lines to determine whether this compound is useful in the treatment of aggressive thyroid cancer.
Materials and Methods
The two thyroid cancer cell lines 8505C and TPC1, under adherent culture conditions, were treated with berberine and analyzed for changes in cell growth, cell cycle duration, and degree of apoptosis.
Results
Following berberine treatment, both cell lines showed a dose-dependent reduction in growth rate. 8505C cells showed significantly increased levels of apoptosis following berberine treatment, whereas TPC1 cells showed cell cycle arrest at the G0/G1 phase. Immunobloting of p-27 expression following berberine treatment showed that berberine induced a little up-regulation of p-27 in 8505c cells but relatively high up-regulation of p-27 in TPC1 cells.
Thyroid cancer is the most prevalent endocrine malignancy, and its incidence has been increasing.1,2 Well-differentiated thyroid carcinoma has a favorable prognosis with surgical and radioactive iodine therapy. Other thyroid malignancies such as poorly differentiated thyroid carcinomas and anaplastic thyroid carcinomas frequently metastasize, precluding patients from a curative resection. Therapeutic options for these patients include additional surgery for resectable lesions, external radiotherapy and chemotherapy. The result of this approach is usually disappointing and the use of novel therapeutic approaches is needed. Poorly-differentiated thyroid carcinoma and anaplastic thyroid carcinoma, which are thought to be dedifferentiated forms of pre-existing differentiated tumors, do not respond well to medical and surgical treatment. There are several therapeutic methods, including surgery, radioactive iodine therapy, and thyroid stimulating hormone suppression by thyroid hormone replacement, for treating thyroid cancer. Most thyroid cancers are well differentiated types and can be cured with ease through surgery and radioactive iodine therapy. However, poorly differentiated or anaplastic thyroid cancer shows rapid progression and is difficult to cure.
Berberine, isolated from the roots of herbal plants, is a natural compound that is non-toxic to humans.3 Berberine has been used as an agent for the treatment of inflammatory disease such as rheumatoid arthritis due to its anti-inflammatory activities.4 Berberine also can control several physical activities, including lowering-cholesterol5 and maintenance of insulin levels.6 Anti-cancer activity of berberine against a variety of cancer cell lines has been introduced. Berberine exhibits antitumor effects by inhibiting the growth,7 invasion,8 and metastatic ability9 of several cancers, including Ehrlich ascites carcinoma,10 lung cancer,11 epidermoid carcinoma,12 leukemia,13 cervical cancer14 and breast cancer.15 Though anticancer activity of berberine has been reported in many articles, its effect on thyroid cancer has not been demonstrated.
To observe the effects of berberine on thyroid cancer, we selected two cell lines, 8505C and TPC1. The 8505C cell line has been used to model anaplastic thyroid cancer, and the TPC1 cell line to model papillary thyroid cancer.16
Unless otherwise specified, all reagents were purchased from Sigma (St. Louis, MO, USA). 8505C and TPC1 cells were cultured on tissue culture dishes (Falcon; Cat no: 353003, San Jose, CA, USA) in Dulbecco's Modified Eagle's Medium (DMEM) containing 10% fetal bovine serum (FBS). For culture, 5×105 cells were cultured at 37℃ in a humidified atmosphere containing 5% CO2.
For each cell line, 5×105 cells were seeded on tissue culture dishes in DMEM containing 10% FBS. After 24 hours, the cells were washed with PBS and cultured in fresh media. Berberine (Sigma, Cat no: B3412, 95% purity, Stock solution; 100 mM in dH2O) was added at 1 µM, 10 µM, or 100 µM for 72 hours. Cells were then counted in a Neubauer chamber after incubation with 0.4% trypan blue dye.
After 10 µM berberine treatment for 72 hours, the cells were harvested, fixed in 70% ethanol for 1 hour, and washed with PBS. Cells were treated with 100 µg/mL RNase A for 1 hour at 37℃, and stained with 10 µg/mL propidium iodide (PI). Flow cytometry was performed in triplicate for each experiment using a FACS Caliber system (BD Biosciences, San Jose, CA, USA).
To analyze apoptosis, the cells treated with 10 µM berberine for 72 hours, and then both floating and adherent cells were harvested. Annexin V staining was performed according to the manufacturer's protocol (BD Pharmingen, Franklin Lakes, NJ, USA). Trypsin-EDTA was used to obtain single-cell suspensions. After centrifugation, cells were washed twice with cold PBS and resuspended in binding buffer (10 mM HEPES, pH 7.4, 150 mM NaCl, 5 mM KCl, 1 mM MgCl2, and 1.8 mM CaCl2) at a concentration of 1×106 cells/mL. An aliquot (100 µL) of the solution containing 1×105 cells was transferred to a 5-mL culture tube, and 5 µL each of Annexin V-FITC and PI were added. After vortexing, cells were incubated for 15 minutes at room temperature (25℃) in the dark and 400 µL of 1X binding buffer was added to each tube. Flow cytometry was performed using a FACS Caliber system within 1 hour.
Cells were detached using trypsin, collected by centrifugation, and cultured as described above. Cells were treated with 10 µM berberine for 72h, and total cell lysates were collected by 200 µL lysis buffer according to the manufacturer's protocol (Cell Signaling Technology, Beverly, MA, USA). To analyze expression of p27, protein concentration was measured using a Bio-Rad Protein Assay kit (Bio-Rad Laboratories, Hercules, CA, USA) according to the manufacturer's directions. Equal amounts of cell lysates were separated by 10% SDS-PAGE. The blocking of blots was processed with a 10% blocking buffer [10% nonfat dry milk in Tris-buffered saline (TBS) containing 0.1% Tween 20] for 1 h. Primary anti-p27 mouse monoclonal immunoglobulin G (IgG) antibody (1 : 1000; Cell Signaling Technology, Beverly, MA, USA) and anti-β-actin mouse monoclonal IgG antibody (1 : 1000, Santa Cruz Biotechnology, Santa Cruz, CA, USA) were incubated overnight at 4℃, and were washed three times in TBS containing 0.1% Tween 20 and secondary peroxidase-conjugated Affinipure rabbit anti-mouse IgG (1 : 2500, Jackson ImmunoResearch Laboratories, West Grove, PA, USA) or peroxidase-conjugated Affinipure mouse anti-rabbit IgG for 1 h at room temperature. Blots of p27proteins were detected with an enhanced chemiluminescence detection system (Amersham Bioscience, Piscataway, NJ, USA).
All data were compiled from a minimum of three replicate experiments. Data for statistical analysis were expressed as the mean±standard error. Comparisons of results from treated versus control cells were performed using Student's t-tests. A p-value of less than 0.05 was considered statistically significant.
We previously reported the effects of berberine in a metastatic breast cancer model.15 To observe the effects of berberine on thyroid cancer, we selected two cell lines, the anaplastic thyroid cancer cell line 8505C and the papillary thyroid cancer cell line TPC1. Both cell lines were treated with berberine at several concentrations (Fig. 1). After 72 hours of treatment, growth was inhibited in a dose-dependent manner in both cell lines (Fig. 1). These results show IC50 of berberine at 10 µM in both cell lines. Thus, 10 µM of berberine was selected for the analysis of the cell cycle and Anexin V staining in both cell lines.
Berberine is known to be an inducing drug for cell cycle arrest and apoptosis. To determine whether growth inhibition was a result of cell cycle arrest, apoptosis, or a combination of both, we analyzed the DNA content of the treated cells by propidium iodide staining and flow cytometry. Growth was inhibited in both cell lines following berberine treatment. In cell cycle analysis using propidium iodide staining, berberine induced a 2% increase in G2/M arrest at 10 µM in 8505C cells (Fig. 2A), whereas TPC1 cells showed a 6% increase in the G0/G1 phase at 10 µM of berberine (Fig. 2B). The cell lines showed different apoptotic responses to berberine treatment. In the Annexin V staining, berberine at 10 µM induced a 7% increase in Annexin V-FITC positive and PI negative 8505C cells (Fig. 3A), but no increase was detected in Annexin V-FITC positive and PI negative TPC1 cells (Fig. 3B).
We found that growth inhibition was caused by cell cycle arrest in TPC1 cells and by apoptosis in 8505c cells following berberine treatment. To confirm these results, we analyzed the expression of p-27, which is known as the molecule involved in cell cycle arrest. As shown in Fig. 4, berberine induced a little up-regulation of p-27 in 8505c cells (Fig. 4A) but relatively high up-regulation of p-27 in TPC1 cells (Fig. 4B).
Berberine is a natural compound reported to have broad anticancer effects, as well as anti-inflammatory effects. We examined the anticancer effect of berberine on the thyroid cancer cell lines 8505C and TPC1, through examination of cell cycle and apoptosis modulation. We observed that berberine treatment of these cell lines decreased growth rates in a dose-dependent manner. Berberine induced apoptosis in both cell types. However, cell cycle arrest was induced mainly in TPC1 cells.
Our results showed 6% increase of the G0/G1 cell cycle in TPC1 cells and 2% increase of the G2/M cycle in 8505C cells after berberine treatment. These results are consistent with our immunoblotting data in which berberine induced a little up-regulation of p-27 in 8505c cells but relatively high up-regulation of p-27 in TPC1 cells after berberine treatment.
Consistent with our results, it has been reported that cell growth can be inhibited by berberine through cell-cycle arrest.17 Berberine has been shown to inhibit the activity of DNA topoisomerase II alpha17 and Cdki-Cdk-cyclin,12 and to induce G0/G1 cell-cycle arrest in L1210 cells3 and G2/M-phase arrest in leukemia cells.18 Previously, we reported similar berberine-induced inhibition of growth through cell-cycle arrest and apoptosis-induction in breast cancer.15 Together with these reports, our results suggest that the effects of berberine treatment of thyroid cancer cells rely upon the induction of cell-cycle arrest in TPC1 cells. It was already demonstrated in our experiments that berberine inhibited cell growth in a dose-dependent manner in TPC1 cells and induced cell cycle arrest at the G0/G1 phase. It was confirmed by immunoblotting that berberine increased p27 in TPC1 cells. All these results suggest that berberine induced cell cycle arrest by induction of p27, which arrests cells at the G0/G1 phase of the cell cycle in TPC1 cells, and thus that berberine can be a novel candidate for an anti-cancer agent against thyroid cancer. Our results showed that berberine more significantly increased the levels of p27 and was more likely to lead to cell cycle arrest in TPC1 cells with RET/PTC1 rearrangement than in 8505C cells with BRAF V600E mutation. P27 has been shown to be targeted by the BRAF/MEK/ERK signaling pathway in distinct tumors models such as melanoma and thyroid carcinomas.19-21
In addition, previous studies have indicated that berberine could induce apoptosis in several cancers, including epidermoid carcinoma,12 human leukemia HL-60 cells,13 and Ehrlich ascites carcinoma cells.22
Here, we also observed that berberine more strongly induced apoptosis in anaplastic thyroid cancer cell line 8505C than in TPC1. In our experiment, berberine induced an 8% increase of apoptosis in Annexin V-FITC positive and PI negative 8505C cells but, 0% in Annexin V-FITC positive and PI negative TPC1 cells. Since externalization of phosphatidylserine occurs in the earlier stages of apoptosis, FITC Annexin V staining can identify apoptosis at an earlier stage than assays based on nuclear changes such as DNA fragmentation.23 Of course, we cannot confirm apoptosis using FITC Annexin V staining, and DNA Fragmentation assay should be done to clearly identify the cytotoxic or cytostatic activities of berberine. However, these results show that growth inhibition by berberine is not associated with cytotoxic effects but with cell cycle arrest through induction of p27 in TPC1 cells.
In conclusion, we have demonstrated that berberine can inhibit the growth of thyroid cancer cells as a result of apoptosis and/or cell-cycle arrest. Thus, it is possible that berberine could be a novel anticancer drug for the treatment of poorly differentiated or anaplastic thyroid cancer.
Figures and Tables
 | Fig. 1Berberine inhibited growth of 8505C and TPC1 cell lines. 8505C (A) and TPC1 (B) cells were treated with distilled water (CTL) or berberine at 1 µM, 10 µM, or 100 µM. Cells were stained with trypan blue to determine cell viability, and counted using a hematocytometer. Relative cell growth rates are shown as percent survival versus control cells after berberine treatment. The rectangles indicate IC 50 of berberine in both cell lines. The data represent the means of at least three independent experiments and the corresponding standard errors. *p-value of less than 0.05. **p-value of less than 0.01. |
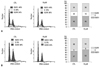 | Fig. 2Cell cycle analysis of berberine-treated 8505C and TPC1 cells. 8505C (A) and TPC1 (B) cells were treated with berberine at 10 µM doses for 72 hours. The percentage of cells at each stage of the cell cycle was analyzed by flow cytometry after DNA staining with propidium iodide. Data from a representative experiment (from a total of three) are shown. |
 | Fig. 3Berberine induced apoptosis in both 8505C cells and TPC1 cells. 8505C (A) and TPC1 (B) cells were treated with berberine at 10 µM doses for 72 hours. All cells were stained with FITC-conjugated Annexin V in a buffer containing propidium iodide and analyzed by flow cytometry. For each treatment, the percentage of viable cells is shown in the lower left quadrant, for which both Annexin V and propidium iodide levels are low. Data from a representative experiment (from a total of three) are shown. |
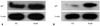 | Fig. 4Expression of p-27 after berberine treatment for 72 h. 8505C (A) and TPC1 (B) cells were treated with berberine at 10 µM doses for 72 hours. Expression of p-27 and β-actin was detected by immunoblotting. Lane 1: cells treated with distilled water (CTL); lane 2: cells treated with berberine at 10 µM doses. |
References
1. Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics, 2007. CA Cancer J Clin. 2007. 57:43–66.

2. Won YJ, Sung J, Jung KW, Kong HJ, Park S, Shin HR, et al. Nationwide cancer incidence in Korea, 2003-2005. Cancer Res Treat. 2009. 41:122–131.

3. Jantová S, Cipák L, Cernáková M, Kost'álová D. Effect of berberine on proliferation, cell cycle and apoptosis in HeLa and L1210 cells. J Pharm Pharmacol. 2003. 55:1143–1149.

4. Hu Z, Jiao Q, Ding J, Liu F, Liu R, Shan L, et al. Berberine induces dendritic cell apoptosis and has therapeutic potential for rheumatoid arthritis. Arthritis Rheum. 2011. 63:949–959.

5. Abidi P, Zhou Y, Jiang JD, Liu J. Extracellular signal-regulated kinase-dependent stabilization of hepatic low-density lipoprotein receptor mRNA by herbal medicine berberine. Arterioscler Thromb Vasc Biol. 2005. 25:2170–2176.

6. Leng SH, Lu FE, Xu LJ. Therapeutic effects of berberine in impaired glucose tolerance rats and its influence on insulin secretion. Acta Pharmacol Sin. 2004. 25:496–502.
7. Inoue K, Kulsum U, Chowdhury SA, Fujisawa S, Ishihara M, Yokoe I, et al. Tumor-specific cytotoxicity and apoptosis-inducing activity of berberines. Anticancer Res. 2005. 25:4053–4059.
8. Peng PL, Hsieh YS, Wang CJ, Hsu JL, Chou FP. Inhibitory effect of berberine on the invasion of human lung cancer cells via decreased productions of urokinase-plasminogen activator and matrix metalloproteinase-2. Toxicol Appl Pharmacol. 2006. 214:8–15.

9. Orfila L, Rodríguez M, Colman T, Hasegawa M, Merentes E, Arvelo F. Structural modification of berberine alkaloids in relation to cytotoxic activity in vitro. J Ethnopharmacol. 2000. 71:449–456.

10. Jantová S, Letasiová S, Brezová V, Cipák L, Lábaj J. Photochemical and phototoxic activity of berberine on murine fibroblast NIH-3T3 and Ehrlich ascites carcinoma cells. J Photochem Photobiol B. 2006. 85:163–176.

11. Katiyar SK, Meeran SM, Katiyar N, Akhtar S. p53 Cooperates berberine-induced growth inhibition and apoptosis of non-small cell human lung cancer cells in vitro and tumor xenograft growth in vivo. Mol Carcinog. 2009. 48:24–37.

12. Mantena SK, Sharma SD, Katiyar SK. Berberine inhibits growth, induces G1 arrest and apoptosis in human epidermoid carcinoma A431 cells by regulating Cdki-Cdk-cyclin cascade, disruption of mitochondrial membrane potential and cleavage of caspase 3 and PARP. Carcinogenesis. 2006. 27:2018–2027.

13. Lin CC, Kao ST, Chen GW, Ho HC, Chung JG. Apoptosis of human leukemia HL-60 cells and murine leukemia WEHI-3 cells induced by berberine through the activation of caspase-3. Anticancer Res. 2006. 26:227–242.
14. Mahata S, Bharti AC, Shukla S, Tyagi A, Husain SA, Das BC. Berberine modulates AP-1 activity to suppress HPV transcription and downstream signaling to induce growth arrest and apoptosis in cervical cancer cells. Mol Cancer. 2011. 10:39.

15. Kim JB, Yu JH, Ko E, Lee KW, Song AK, Park SY, et al. The alkaloid Berberine inhibits the growth of Anoikis-resistant MCF-7 and MDA-MB-231 breast cancer cell lines by inducing cell cycle arrest. Phytomedicine. 2010. 17:436–440.

16. Hoffmann S, Burchert A, Wunderlich A, Wang Y, Lingelbach S, Hofbauer LC, et al. Differential effects of cetuximab and AEE 788 on epidermal growth factor receptor (EGF-R) and vascular endothelial growth factor receptor (VEGF-R) in thyroid cancer cell lines. Endocrine. 2007. 31:105–113.

17. Kang MR, Chung IK. Down-regulation of DNA topoisomerase IIalpha in human colorectal carcinoma cells resistant to a protoberberine alkaloid, berberrubine. Mol Pharmacol. 2002. 61:879–884.

18. Lin CC, Lin SY, Chung JG, Lin JP, Chen GW, Kao ST. Down-regulation of cyclin B1 and up-regulation of Wee1 by berberine promotes entry of leukemia cells into the G2/M-phase of the cell cycle. Anticancer Res. 2006. 26:1097–1104.
19. Bhatt KV, Hu R, Spofford LS, Aplin AE. Mutant B-RAF signaling and cyclin D1 regulate Cks1/S-phase kinase-associated protein 2-mediated degradation of p27Kip1 in human melanoma cells. Oncogene. 2007. 26:1056–1066.

20. Bhatt KV, Spofford LS, Aram G, McMullen M, Pumiglia K, Aplin AE. Adhesion control of cyclin D1 and p27Kip1 levels is deregulated in melanoma cells through BRAF-MEK-ERK signaling. Oncogene. 2005. 24:3459–3471.

21. Preto A, Gonçalves J, Rebocho AP, Figueiredo J, Meireles AM, Rocha AS, et al. Proliferation and survival molecules implicated in the inhibition of BRAF pathway in thyroid cancer cells harbouring different genetic mutations. BMC Cancer. 2009. 9:387.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download