Abstract
In this paper, we report on a 5-year-old girl who developed a renal stone while following the ketogenic diet to treat refractory seizure disorder. Three months after initiating the ketogenic diet, she developed severe abdominal pain and vomiting. The spot urine calcium-to-creatinine (Ca/Cr) ratio and 24-hour urine evaluation showed hypercalciuria. Computed tomography (CT) imaging revealed a stone in the right ureteropelvic junction, resulting in hydronephrosis of the right kidney. The renal stone disappeared 5 days after conservative treatment; the patien's microscopic hematuria resolved concurrently. In light of this case report, we recommend regularly monitoring the urine Ca/Cr ratio with ultrasonography for further development of renal stones in patients following the ketogenic diet. If these patients exhibit evidence of symptomatic hypercalciuria or cyristalluria, liberalization of fluid restriction and urine alkalization using oral potassium citrate should be considered.
The ketogenic diet was introduced by Wilder in 1921 to treat medically intractable childhood epilepsy.1 Recently published studies have demonstrated the diet's antiepileptic effects.2-6 Several hypotheses have been put forward to explain the mechanism behind its efficacy.7,8 Kalapos8 proposed that ketone bodies might result in reduced neuronal excitability, resulting in a direct anticonvulsant effect. Janigro7 suggested that the diet's efficacy may be related to increased availability of beta-hydroxybutyrate, a ketone body readily transported through the blood-brain barrier. However, the mechanisms underlying the protective effect of these compounds are not completely understood.
There have been several reports about renal calculi developing in children on the ketogenic diet since the first report more than 30 years ago.9-11 The prevalence of renal caculi in people on the ketogenic diet is 3-10%, compared with 1 in several thousand in the general population.12 Chronic acidosis, dehydration, low urine pH, and fat malabsorption all contribute to the formation of uric acid and calcium oxalate stones.9,10 In this paper, we describe the case of a 5-year-old girl who developed a renal stone after beginning a ketogenic diet to control refractory complex partial seizures.
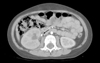
A 5-year-old girl presented with severe abdominal pain, nausea, and vomiting for one day. Her past medical history was significant for eyelid fluttering with or without facial paresis, at least 50-100 times per day. Complex partial seizures were diagnosed at 4-years-old; therapy included 2 months of antiepileptic drugs such as zonisamide, valproate, vigabatrin, and clonazepam at an outside hospital. Despite this treatment, her seizures did not improve, so she was subsequently referred to our hospital's Epilepsy Center. A subtotal right frontal lobectomy preserving the motor cortex was performed 4 months after localizing the epileptogenic focus using a brain magnetic resonance image, single photon emission computed tomography, and positron emission tomography studies. The initial electroencephalogram pattern was focal slowings and a frequent sharp wave discharged from the right frontal area and focal slowings on the centro-temporal areas. Its pattern changed to a rhythmic sharp wave discharged from the right temporal areas after a right subtotal frontal lobectomy. Despite the epileptic surgery, seizure frequency remained at 50-100 times per day as well as similar seizure patterns and intensity. Ten days after surgery, she was started on a 1,300 kcal ketogenic diet with a non-lipid: lipid ratio of 4 : 1. Although seizure frequency decreased 5 times per day, she developed severe abdominal pain, nausea, and vomiting after 3 months. On a physical examination, she had direct tenderness without rebound tenderness in the right lower abdominal quadrant. Her blood pressure was 107/72 mmHg; chest and abdomen X-rays were unremarkable. Laboratory findings showed a white blood cell count of 12,420/µL, hemoglobin 13.8 g/dL and platelets 395×103/µL. Serum total protein was 7.0 g/dL, albumin 4.6 g/dL, blood urea nitrogen 7.7 mg/dL, creatinine 0.3 mg/dL, calcium 9.3 mg/dL, uric acid 3.9 mg/dL, sodium 142 mmol/L, potassium 3.9 mmol/L, chloride 102 mmol/L, and total CO2 12 mmol/L. Urinalysis showed a specific gravity of 1.030, pH 5.0, hematuria (3-5/high power field), and 3-plus ketones. The spot urine calciumto-creatinine ratio was 1.0 mg/mg (reference level: < 0.2) and 24-hour urine calcium excretion was 5.9 mg/kg (reference level: < 4 mg/kg), suggesting hypercalciuria. Twenty-four hour uric acid excretion was within normal limits. In order to evaluate acute appendicitis, abdominal CT was initially performed. It revealed a stone in the right ureteropelvic junction, resulting in hydronephrosis (Fig. 1).
The patient was treated with aggressive hydration and she spontaneously passed the stone, so we could not check the calculus composition. An intravenous pyelogram demonstrated no definite intrinsic abnormalities in the kidneys, Ureters, or bladder; renal ultrasonography indicated the complete resolution of previously noted right side abnormalities.
After 1 month, a follow-up ultrasonography showed no abnormal findings. Because seizure frequency increased by over 40 times per day, she received a right frontal lobectomy and cortisectomy of the periinsular and superior temporal gyri, finally attaining seizure-free status.
The ketogenic diet has been increasingly used by neurologists over the past 20 years for children with medically intractable epilepsy. Recent studies have reported that 23% to 44% of patients show a reduction in seizure frequency of > 50%, including 7% to 22% who become seizure-free after 12 months on the classic ketogenic diet.3,4,13,14 However, side effects have previously been reported in patients following a ketogenic diet.9,10 Renal stones are a significant complication of the ketogenic diet. Calculus composition has been reported as uric acid, calcium oxalate, or a mixture of calcium oxalate and calcium phosphate/uric acid.10,15 There are several reasons underlying the elevated risk of developing renal calculi in patients on the ketogenic diet. First, hypercalciuria can develop due to chronic metabolic acidosis. This metabolic acidosis not only decreases calcium reabsorption in the renal tubules, thus increasing urinary calcium excretion,10 but also increases bone demineralization because bone phosphate acts as an acid buffer.16 Second, children on a ketogenic diet show hypocitraturia. Citrate normally binds urine calcium, lowering its concentration, acting as an inhibitor of calcium crystallization.16 Acidosis induces proximal tubules to both increase citrate absorption and decreases its excretion. As a result, acidosis not only reduces urinary citrate excretion but also increases urinary calcium excretion, aggravating renal stone formation. Third, chronic acidosis persistently causes low urinary pH, which facilitates uric acid crystal formation due to lowered uric acid solubility. These crystals can act as a nidus for calcium stone formation.16 Lastly, dehydration may be the most significant factor in calculus formation in children on the ketogenic diet,15 primarily because ketosis has been shown to interfere with the normal thirst mechanism.10
In light of these complications, fluid liberalization and urine alkalization using oral potassium citrate should be considered as prophylaxis to prevent renal stone formation in children beginning this diet.10,15,17 Potassium citrate increases urine pH and solublizes calcium, thereby decreasing the concentration of free calcium available to crystallize. One of our authors has suggested that initial fasting and fluid restriction are not essential to the ketogenic diet.18 Because an increase in fluid intake does not diminish the efficacy of the ketogenic diet in controlling seizures and blood ketone levels,11 these preventive measures may be useful in reducing the formation of renal stones.
In conclusion, the ketogenic diet is a risk factor for kidney stones, and hypercalciuria was more common in those with kidney stones. We recommend maximizing fluid intake and alkalinizing the urine to prevent the development of renal stones. Regular urinary studies including calcium-to-creatinine ratio and ultrasonography are highly recommended to detect this possible complication in children receiving the ketogenic diet.9,10 Any evidence of hematuria, dysuria, or crystalluria should be evaluated with both a renal ultrasonography and a nephrology referral.
Figures and Tables
References
1. Wilder RM. The effects of ketonemia on the course of epilepsy. Mayo Clin Bull. 1921. 2:307–308.
2. Nordli DR Jr, De Vivo DC. The ketogenic diet revisited: back to the future. Epilepsia. 1997. 38:743–749.

3. Vining EP, Freeman JM, Ballaban-Gil K, Camfield CS, Camfield PR, Holmes GL, et al. A multicenter study of the efficacy of the ketogenic diet. Arch Neurol. 1998. 55:1433–1437.
4. Freeman JM, Vining EP, Pillas DJ, Pyzik PL, Casey JC, Kelly LM. The efficacy of the ketogenic diet-1998: a prospective evaluation of intervention in 150 children. Pediatrics. 1998. 102:1358–1363.
5. Kang HC, Kim YJ, Kim DW, Kim HD. Efficacy and safety of the ketogenic diet for intractable childhood epilepsy: Korean multicentric experience. Epilepsia. 2005. 46:272–279.
7. Janigro D. Blood-brain barrier, ion homeostatis and epilepsy: possible implications towards the understanding of ketogenic diet mechanisms. Epilepsy Res. 1999. 37:223–232.

8. Kalapos MP. Possible mechanism for the effect of ketogenic diet in cases of uncontrolled seizures. The reconsideration of acetone theory. Med Hypotheses. 2007. 68:1382–1388.
9. Kang HC, Chung DE, Kim DW, Kim HD. Early- and late-onset complications of the ketogenic diet for intractable epilepsy. Epilepsia. 2004. 45:1116–1123.
10. Furth SL, Casey JC, Pyzik PL, Neu AM, Docimo SG, Vining EP, et al. Risk factors for urolithiasis in children on the ketogenic diet. Pediatr Nephrol. 2000. 15:125–128.

11. Livingsone S. Comprehensive Management of epilepsy in infancy, childhood, and adolescence. 1972. Springfield, Ill: Thomas.
12. Dodson WE, Prensky AL, DeVivo DC, Goldring S, Dodge PR. Management of seizure disorders: selected aspects. Part II. J Pediatr. 1976. 89:695–703.

13. Swink TD, Vining EP, Freeman JM. The ketogenic diet: 1997. Adv Pediatr. 1997. 44:297–329.
14. DiMario FJ Jr, Holland J. The ketogenic diet: a review of the experience at Connecticut Children's Medical Center. Pediatr Neurol. 2002. 26:288–292.

15. Kielb S, Koo HP, Bloom DA, Faerber GJ. Nephrolithiasis associated with the ketogenic diet. J Urol. 2000. 164:464–466.

17. Sampath A, Kossoff EH, Furth SL, Pyzik PL, Vining EP. Kidney stones and the ketogenic diet: risk factors and prevention. J Child Neurol. 2007. 22:375–378.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download