Abstract
Purpose
Enhancer of zeste homolog 2 (EZH2), a kind of transcriptional repressor, is reportedly over-expressed in metastatic prostate cancer. In this study, we analyzed EZH2 mRNA in circulating tumor cells (CTCs) in peripheral blood as a biomarker in patients with metastatic prostate cancer.
Patients and Methods
Ber-EP4 coated immunomagnetic beads were used to harvest CTCs, and mRNA was isolated by oligo-dT conjugated immunomagnetic beads. Reverse transcriptase-polymerase chain reaction for EZH2 mRNA was performed and the expression density was measured. The sensitivity of this test for detection of EZH2 mRNA was determined by serial dilutions of a human prostate cancer cell line. Blood samples were collected from 20 patients each with metastatic or localized prostate cancer and 10 healthy volunteers.
Results
Sensitivity experiments showed that the test was highly sensitive as it could detect 10 tumor cells per 5 mL. EZH2 mRNA expression was obtained from peripheral blood samples of patients and control subjects. EZH2 mRNA expression density in the metastatic prostate cancer group was significantly higher than in the control (p = 0.023) and localized prostate cancer groups (p = 0.019). There was no difference between the control and localized prostate cancer groups (p > 0.05).
Prostate cancer is the most commonly diagnosed male malignancy and the second leading cause of cancer death in men in the Western world. In Korea, prostate cancer is the most rapidly increasing male malignancy by incidence. Although the introduction of serum prostate specific antigen (PSA) measurement has allowed early detection of the disease, clinicians are unable to determine the potential aggressiveness of individual tumors with certainty, resulting in the over-or under-treatment of many patients.
The reverse transcriptase-polymerase chain reaction (RT-PCR) technique has been used to search for cells expressing PSA or prostate-specific membrane antigen in extraprostatic sites, including the peripheral blood, lymph nodes, and bone marrow, with various ranges of positivity.1-10 These approaches have only limited value, however, because dissemination of cells is an early event in prostate cancer that may or may not lead to the development of metastases.
Enhancer of zeste homolog 2 (EZH2) is a known repressor of gene transcription. Varambally et al. first suggested that dysregulated expression of EZH2 might be involved in the progression of prostate cancer,11 and Bryant et al. demonstrated that EZH2 promotes proliferation and invasion of prostate cancer cells.12 Recently, Takeshita et al. showed that the EZH2 small interfering RNA, which was selected as candidate targets for inhibition of bone metastasis, resulted in an efficient inhibition of metastatic tumor growth in bone tissues.13 In this study, we analyzed EZH2 mRNA in circulating tumor cells (CTCs) in peripheral blood as a biomarker in patients with metastatic prostate cancer in order to more clearly elucidate their metastatic potential.
Blood samples were collected from 20 patients each with metastatic prostate cancer and localized prostate cancer, and 10 healthy volunteers. Metastatic diseases were confirmed by chest X-ray, whole body bone scan, or computed tomography. Blood samples of patients with localized prostate cancer were obtained before radical prostatectomy. Healthy volunteers were men with normal serum PSA and normal digital rectal examination. All patients provided written informed consent. Characteristics of the subjects are summarized in Table 1.
Five mL of venous peripheral blood were collected in a heparinized tube and put on ice immediately. Whole blood was mixed with 10 mL of phosphate buffered saline (PBS) and the mononuclear cell layer was isolated using Accuspin™ System-Histopaque®-1077 (Sigma-Aldrich®, St. Louis, USA) according to the manufacturer's protocol. The cell pellet was resuspended in 1 mL of PBS-2% fetal calf serum (FCS; Invitrogen Corporation, Gibco™) and then incubated with 20 million of prewashed Ber-EP4 monoclonal antibodies for 30 minutes at 4℃ with gentle tilting and rotation (Dynal Biotech®, Oslo, Norway). Dynabeads to purify epithelial cells are superparamagnetic polystyrene beads coated with anti-human Ber-EP4 antibody, directed against the human epithelial antigen. The rosetted cells were isolated by placing the tube in a Dynal magnetic particle concentrator (MPC). The harvested epithelial cells were then washed five times with PBS-2% FCS.
The Dynabeads mRNA Direct™ Micro kit (Dynal Biotech®) was used to isolate mRNA from epithelial cells. The rosetted cells were lysed using 100 µL Lysis-binding buffer (Dynal Biotech®). The supernatant was then transferred into a new tube containing 20 µL of pre-washed oligo-dT Dynabeads and incubated for 5 minutes on a roller at room temperature. The Dynabeads for oligo-dT are superparamagnetic beads with covalently attached oligo-dT. The tube was then placed in the Dynal MPC, and the supernatant was discarded. The washing step was repeated five times according tothe manufacturer's recommendations. The final suspension was kept on ice prior to RT-PCR amplification.
Ten µL of mRNA were reverse transcribed into cDNA, using a reverse transcription system (Promega Co., WI, USA) following the supplier's recommendations. The 50 µL PCR reaction mixture included 20 µL of cDNA, 25 µL Taq PCR Master Mix Kit (Qiagen, Germany), 5 µL of sense primer, and 5 µL of anti-sense primer. The EZH2 primers used in this study were described by Varambally et al.11 Thermocycling conditions included 35 cycles of 94℃ for 1 minute, 60℃ for 45 sec, and 72℃ for 2 minutes. The entire RT-PCR product was run on a 2% ethidium bromide-stained agarose gel, and the image density of EZH2 mRNA expression was measured by Quantity One® software version 4 (Bio-Rad laboratories, CA, USA).
PC3 cells, derived from a patient with hormone refractory prostate cancer, were used to define the sensitivity of the assay. These prostatic tumor cells were mixed with 5 mL of PBS in 10-fold serial dilutions of 10,000, 1,000, 100, 10, and 1 PC3 cells in 5 mL of PBS. These mixtures were subjected to the experimental steps previously described.
Immunomagnetic separation and RT-PCR for EZH2 mRNA was highly sensitive and could detect 10 tumor cells in 5 mL of PBS (Fig. 1). EZH2 mRNA expression density increased in proportion to the number of PC3 cells present.
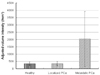
EZH2 mRNA expression was obtained from peripheral blood samples of healthy volunteers and patients with localized prostate cancer or metastatic prostate cancer (Fig. 2). The mean expression density was 345.7 ± 131.8 intensity/mm2 in healthy volunteers, 349.4 ± 156.7 intensity/mm2 in patients with localized prostate cancer, and 2040.5 ± 1881.3 ntensity/mm2 in patients with metastatic prostate cancer (Fig. 3).
EZH2 mRNA expression density in the metastatic prostate cancer group was significantly higher than in the control (p = 0.023) and localized prostate cancer groups (p = 0.019). There was no difference between the control and localized prostate cancer groups (p > 0.05) (Fig. 3).
Current methods for detecting micrometastases in prostate cancer patients are not satisfactory. For example, 15% to 40% of men who seem to have localized disease based on imaging studies and clinical parameters will instead develop recurrent disease.14-16 For this reason, identification of CTCs in peripheral blood is expected to have a therapeutic or prognostic significance.
CTCs can be detected by PCR amplification of tumor-specific DNA and mRNA abnormalities that are present in malignant cells. PCR is more sensitive than conventional techniques for the detection of tumor cells and micrometastases, allowing the identification of 1 tumor cell among 106-107 normal cells.17 In 1992, Moreno et al first used RT-PCR to detect the presence of PSA mRNA in peripheral blood specimens of patients with metastatic prostate cancer.18 Since then, there have been numerous reports using this technology to detect the presence of occult prostate cells in blood, with CTCs detected in 0 to 72% in patients with clinically organ-confined disease and 25 to 100% in patients with distant metastases.1-9 However, the major question of the specificity of the RT-PCR approach remains unanswered due to the potential for illegitimate transcripts in lymphocytes or other blood cells.
Interestingly, Ellis et al showed that epithelial cells express PSA in the bone marrow of 54% of men with clinically localized disease before radical prostatectomy.2 This finding is much higher than the 10% to 30% of patients expected to develop recurrence after radical prostatectomy. Such approaches to detect PSA-expressing epithelial cells have only limited value, because dissemination of cells is an early event in prostate cancer that may or may not lead to the development of metastases.
To overcome these limitations and enhance the clinical significance of detection of CTCs, new approaches to enumerate CTCs have been tried. Moreno et al demonstrated that CTC counts can be reproducibly measured in patients with hormone refractory prostate cancer without apparent diurnal variation, and that the changes in the CTC levels mirror disease progression.19 Chen et al further demonstrated that the number of CTCs reflects current disease status and correlates significantly with clinical disease indicators.20 On the other hand, a more specific marker for tumor progression that is not a prostate-specific may be more attractive. For example, HER-2/neu in CTCs of patients with prostate cancer is more frequent in those with metastases than those with localized disease.21
To our knowledge, EZH2 has not been used for detecting CTCs from prostate cancer. In the present study, we combined immunomagnetic separation of CTCs with RT-PCR detection of the EZH2 transcripts to increase the specificity and better characterize the molecular profile of CTCs. Gene expression profiling of hormone refractory metastatic prostate cancer cases has shown that a high level of expression of the Polycomb Group gene EZH2 is positively correlated with the progression of this malignancy in humans.11 Amplification of the EZH2 gene occurs in a significant proportion of advanced hormone refractory prostate tumors compared to earlier untreated tumor samples, suggesting a specific role for EZH2 in disease progression.22 Moreover, patients with clinically localized prostate cancer that express high levels of EZH2 are associated with a poorer prognosis following radical prostatectomy.11,23 The role of EZH2 in prostate cancer is still poorly understood, but a recent study suggests that early dysregulation of EZH2 in a subset of prostate cancer cases might promote progression of this disease from an indolent to an aggressive and advance phenotype.12
In this preliminary study, we demonstrate a promising approach to investigate CTCs. Studies are ongoing to determine how the genotype and phenotype of isolated disseminated cells differs in tumors with different metastatic potentials. Nevertheless, our results have some limitations. First, a more sensitive technique is needed to detect CTCs less than 10 in number because tumor cells in peripheral blood are very rare. Although EZH2 mRNA was most strongly expressed in patients with metastatic prostate cancer, it was also identified in the control and localized prostate cancer groups. This result may be explained by mononuclear cell contamination and illegitimate transcription of EZH2, necessitating more specific techniques to avoid such contamination. In addition, further experiments such as real time RT-PCR are needed for more clear quantification of EZH2 mRNA expression. Meanwhile, the clinical significance of EZH2 mRNA expression in CTCs remains uncertain, and studies with a larger sample size and long-term follow-up are needed to fully examine the diagnostic and prognostic possibilities explored in this study.
In conclusion, EZH2 mRNA in circulating tumor cells was over-expressed in patients with metastatic prostate cancer. EZH2 mRNA could be a promising marker for detecting early metastasis in prostate cancer, but more specific and sensitive techniques for the detection of circulating tumor cells are needed to avoid mononuclear cell contamination.
Figures and Tables
Fig. 1
Sensitivity of reverse transcriptase-polymerase chain reaction for enhancer of zeste homolog 2. The numbers 1-10,000 indicate the number of PC3 cells in 5 mL of phosphate buffered saline.

Fig. 2
Representative results of reverse transcriptase-polymerase chain reaction for enhancer of zeste homolog 2 in peripheral blood samples obtained from healthy volunteers and patients with localized prostate cancer or metastatic prostate cancer.

Fig. 3
Expression density of reverse transcriptase-polymerase chain reaction for enhancer of zeste homolog 2 in peripheral blood samples obtained from subjects as measured by the Quantity One® program (*p < 0.05 by Mann-Whitney U test, compared to the healthy group or localized prostate cancer group).
References
1. Corey E, Arfman EW, Oswin MM, Melchior SW, Tindall DJ, Young CY, et al. Detection of circulating prostate cells by reverse transcriptase-polymerase chain reaction of human glandular kallikrein (hK2) and prostate-specific antigen (PSA) messages. Urology. 1997. 50:184–188.
2. Ellis WJ, Pfitzenmaier J, Colli J, Arfman E, Lange PH, Vessella RL. Detection and isolation of prostate cancer cells from peripheral blood and bone marrow. Urology. 2003. 61:277–281.
3. Ghossein RA, Scher HI, Gerald WL, Kelly WK, Curley T, Amsterdam A, et al. Detection of circulating tumor cells in patients with localized and metastatic prostatic carcinoma: clinical implications. J Clin Oncol. 1995. 13:1195–1200.
4. Hara N, Kasahara T, Kawasaki T, Bilim V, Obara K, Takahashi K, et al. Reverse transcription-polymerase chain reaction detection of prostate-specific antigen, prostate-specific membrane antigen, and prostate stem cell antigen in one milliliter of peripheral blood: value for the staging of prostate cancer. Clin Cancer Res. 2002. 8:1794–1799.
5. Israeli RS, Miller WH Jr, Su SL, Powell CT, Fair WR, Samadi DS, et al. Sensitive nested reverse transcription polymerase chain reaction detection of circulating prostatic tumor cells: comparison of prostate-specific membrane antigen and prostate-specific antigen-based assays. Cancer Res. 1994. 54:6306–6310.
6. Katz AE, Olsson CA, Raffo AJ, Cama C, Perlman H, Seaman E, et al. Molecular staging of prostate cancer with the use of an enhanced reverse transcriptase-PCR assay. Urology. 1994. 43:765–775.
7. Mejean A, Vona G, Nalpas B, Damotte D, Brousse N, Chretien Y, et al. Detection of circulating prostate derived cells in patients with prostate adenocarcinoma is an independent risk factor for tumor recurrence. J Urol. 2000. 163:2022–2029.
8. Seiden MV, Kantoff PW, Krithivas K, Propert K, Bryant M, Haltom E, et al. Detection of circulating tumor cells in men with localized prostate cancer. J Clin Oncol. 1994. 12:2634–2639.
9. Sokoloff MH, Tso CL, Kaboo R, Nelson S, Ko J, Dorey F, et al. Quantitative polymerase chain reaction does not improve preoperative prostate cancer staging: a clinicopathological molecular analysis of 121 patients. J Urol. 1996. 156:1560–1566.
10. Wood DP Jr, Banerjee M. Presence of circulating prostate cells in the bone marrow of patients undergoing radical prostatectomy is predictive of disease-free survival. J Clin Oncol. 1997. 15:3451–3457.
11. Varambally S, Dhanasekaran SM, Zhou M, Barrette TR, Kumar-Sinha C, Sanda MG, et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature. 2002. 419:624–629.
12. Bryant RJ, Cross NA, Eaton CL, Hamdy FC, Cunliffe VT. EZH2 promotes proliferation and invasiveness of prostate cancer cells. Prostate. 2007. 67:547–556.
13. Takeshita F, Minakuchi Y, Nagahara S, Honma K, Sasaki H, Hirai K, et al. Efficient delivery of small interfering RNA to bone-metastatic tumors by using atelocollagen in vivo. Proc Natl Acad Sci U S A. 2005. 102:12177–12182.
14. Hull GW, Rabbani F, Abbas F, Wheeler TM, Kattan MW, Scardino PT. Cancer control with radical prostatectomy alone in 1,000 consecutive patients. J Urol. 2002. 167:528–534.
15. Han M, Partin AW, Piantadosi S, Epstein JI, Walsh PC. Era specific biochemical recurrence-free survival following radical prostatectomy for clinically localized prostate cancer. J Urol. 2001. 166:416–419.
16. Amling CL, Blute ML, Bergstralh EJ, Seay TM, Slezak J, Zincke H. Long-term hazard of progression after radical prostatectomy for clinically localized prostate cancer: continued risk of biochemical failure after 5 years. J Urol. 2000. 164:101–105.
17. Ghossein RA, Rosai J. Polymerase chain reaction in the detection of micrometastases and circulating tumor cells. Cancer. 1996. 78:10–16.
18. Moreno JG, Croce CM, Fischer R, Monne M, Vihko P, Mulholland SG, et al. Detection of hematogenous micrometastasis in patients with prostate cancer. Cancer Res. 1992. 52:6110–6112.
19. Moreno JG, O'Hara SM, Gross S, Doyle G, Fritsche H, Gomella LG, et al. Changes in circulating carcinoma cells in patients with metastatic prostate cancer correlate with disease status. Urology. 2001. 58:386–392.
20. Chen BT, Loberg RD, Neeley CK, O'Hara SM, Gross S, Doyle G, et al. Preliminary study of immunomagnetic quantification of circulating tumor cells in patients with advanced disease. Urology. 2005. 65:616–621.
21. Ady N, Morat L, Fizazi K, Soria JC, Mathieu MC, Prapotnich D, et al. Detection of HER-2/neu-positive circulating epithelial cells in prostate cancer patients. Br J Cancer. 2004. 90:443–448.
22. Saramäki OR, Tammela TL, Martikainen PM, Vessella RL, Visakorpi T. The gene for polycomb group protein enhancer of zeste homolog 2 (EZH2) is amplified in late-stage prostate cancer. Genes Chromosomes Cancer. 2006. 45:639–645.
23. Rhodes DR, Sanda MG, Otte AP, Chinnaiyan AM, Rubin MA. Multiplex biomarker approach for determining risk of prostate-specific antigen-defined recurrence of prostate cancer. J Natl Cancer Inst. 2003. 95:661–668.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download