Abstract
Skin, as the outermost organ in the human body, continuously confronts the external environment and serves as a primary defense system. The protective functions of skin include UV-protection, anti-oxidant and antimicrobial functions. In addition to these protections, skin also acts as a sensory organ and the primary regulator of body temperature. Within these important functions, the epidermal permeability barrier, which controls the transcutaneous movement of water and other electrolytes, is probably the most important. This permeability barrier resides in the stratum corneum, a resilient layer composed of corneocytes and stratum corneum intercellular lipids. Since the first realization of the structural and biochemical diversities involved in the stratum corneum, a tremendous amount of work has been performed to elucidate its roles and functions in the skin, and in humans in general. The perturbation of the epidermal permeability barrier, previously speculated to be just a symptom involved in skin diseases, is currently considered to be a primary pathophysiologic factor for many skin diseases. In addition, much of the evidence provides support for the idea that various protective functions in the skin are closely related or even co-regulated. In this review, the recent achievements of skin researchers focusing on the functions of the epidermal permeability barrier and their importance in skin disease, such as atopic dermatitis and psoriasis, are introduced.
The skin, as an interface between the organism and the external environment, plays a major role in protecting and supporting the life it encloses. The functions of human skin include the maintenance of body temperature, recognition of the outer environment, defense against microorganisms and protection from harmful materials in the external environment. Among these important functions, defensive functions can be further classified as physical, thermal, immune, ultra- violet, oxidant, racial, antimicrobial and the permeability barrier.
The epidermis is the outermost viable layer of the skin and expresses various proteins and other molecules that perform the above mentioned protective functions. Inflammatory mediators such as prostaglandins, eicosanoids, leukotrienes, histamines and cytokines are synthesized and secreted from keratinocytes and regulate the skin's immune responses.1 UV-absorbing molecules, including melanin, trans-urocanic acid, vitamin D and C metabolites, and heat-shock proteins are also expressed in keratinocytes and play important roles in thermal and UV-barrier functions.2-5 The antimicrobial systems in skin are primarily mediated through the surface lipids, skin surface acidification, iron-binding proteins and antimicrobial peptides.6,7 The permeability barrier function, which impedes the transcutaneous movement of water and other important electrolytes, is the most important defensive functions for terrestrial life.
The outermost layer of the skin, the stratum corneum (SC), primarily mediates this permeability barrier function (Fig. 1). SC is composed of two different structural components: the corneocytes and intercorneocyte lipids. Both components are derived from keratinocytes through the terminal differentiation process. Considerable efforts have been made to elucidate the structure, function and biochemistry of the SC and, about two decades ago, Elias proposed the 'brick and mortar model', in which the SC is composed of flat cells (bricks) surrounded by a lipid matrix (mortar).8 Since then, many studies have been performed to elucidate the role of the SC at the organismal, biochemical and molecular biological levels. The corneocytes, as terminal differentiation forms of keratinocytes, provide structural supports for the SC and act as hydrating reservoirs for adequate enzymatic processes in the SC. The cornified envelope (CE), which encapsulates the corneocytes in SC, is a 15-20 nm-thick structure comprising defined structural proteins.9 A 5nm-thick structure of specialized lipids, identified as the cornified lipid envelope (CLE), encloses the CE and this membrane-bound lipid monolayer provides hydrophobic interfaces between the hydrophilic surface of the CE and the highly hydrophobic lipid lamellae.10 Recently, a detailed elucidation of the component proteins of the CE and its formation has been reported.11
In addition to these major structural domains, the corneodesmosome(CD), which corresponds to desmosome in the epidermis, is another important components in the SC. Generally, the integrity of the SC is maintained by these intercellular proteins which connect to adjacent corneocytes, both in the plane of the SC layer and in adjacent layers. In the SC, the CD structures represent the primary cohesive force and they are directly related the desquamation process. These structures are composed of certain proteins, including desmocollin and desmoglein, and special protein-degrading enzymes which are presented in the SC and play a crucial role in the desquamation process.12 Water activity and pH control the activity of the protease in the SC. The cholesterol sulfate/cholesterol ratio and protease inhibitors in the SC are also important regulators of the protease activity.13,14
In this review, we introduce recent achievements in the research of skin barrier functions and their significances in treating atopic dermatitis and psoriasis.
In addition to the permeability barrier function, the SC also plays an important role in other critical functions such as antimicrobial defense, the hydration of viable epidermis, UV defense, antioxidant defense and the formation of a mechanical barrier. Due to structural heterogeneity, either corneocytes or the SC intercellular lipids mediate the defensive functions of the SC: the extracellular lipids act as permeability, anti-oxidant, and antimicrobial barriers, and the corneocytes act as UV and mechanical barriers. Recent reports, however, show that these functions are closely interrelated and that the change of any function could result in the modulation of other functions. These defensive functions are interrelated by co-localization, biochemical link-age or through common regulatory mechanisms.
As an antimicrobial barrier against invading microorganisms, skin acts as a physical barrier and produces a number of antimicrobial peptides and proteins, including human defensins and cathelicidins. These antimicrobial peptides (AMPs), which play a major role in the host's innate defenses, are generally small, cationic polypeptides and have a capacity to inhibit the growth of microbes. In addition to the direct antimicrobial activities against various bacteria, viruses and fungi, AMPs also activate cellular and adaptive immune responses. They also play important roles as mediators of inflammation with effects on epithelial and inflammatory cells, influencing cell proliferation, wound healing, cytokine and chemokine production and chemotaxis. While cathelicidins and defensins comprise the major families of AMPs in the skin, other molecules such as protease inhibitors, chemokines and neuropeptides, also exhibit antimicrobial activity. In addition to those molecules, free fatty acids, glucosylceramides and sphingosine, which are major constituents for SC intercellular lipids, and hydrolytic products of ceramide also provide an antimicrobial barrier in the skin.
While AMPs are generally synthesized in the upper stratum spinosum (SS) and stratum granulosum, the active site for these molecules is the SC where they are delivered. Recently, it has been shown that human β-defensin 2 (HBD-2) is localized in the lamellar bodies (LBs)15 and cathelicidin also resides in LBs.16 Since the LB is an essential micro-organelle for epidermal barrier formation, it can be postulated that the epidermal permeability barrier function is closely related to the antimicrobial function of skin. In several skin diseases showing disturbed skin barrier function, such as psoriasis and atopic dermatitis (AD), abnormal expression of AMPs was reported. For example, LL-37, hBD-2 and hBD-3 are up regulated in psoriatic skin lesions.17,18 The expression of LL-37 and hBD-2 is relatively down regulated in atopic dermatitis lesions compared to psoriatic skin,19 which correlates with the high susceptibility of AD skin to bacterial and viral infections. Recently, we have reported that epidermal expression of CRAMP, a murine analogue of human LL-37, is significantly increased after UVB exposure.20 Since phototherapy using UVB is frequently used in severe AD, increases in AMPs can give a supplemental explanation for the beneficial effect of UV in AD.
The formation of corneocytes is considered to be a result of finely regulated differentiation processes. During the terminal differentiation process, structural change of the keratinocyte is associated with the sequential formation of differentiation-marker proteins that are unique to keratinocytes. Keratins such as K1 and K10, as early marker proteins of terminal differentiation, are expressed in the spinous layer. Late differentiation marker proteins, including involucrin, loricrin and filaggrin, appear in the granular layer. Complete degradation of intracellular micro-organelles such as nucleus and mitochondria and the formation of rigid CE complete the differentiation process. Keratinocyte-specific transglutaminase-1 catalyzes the cross-linking reaction between keratins and other structural proteins to form the chemically resistant cornified envelope structures of corneocytes.11
An in vitro cell culture system has been extensively used to identify the crucial factors influencing keratinocyte differentiation. Through these intensive studies, it has been shown that the epidermal differentiation process is regulated by the concentration of extracellular calcium ions. Keratinocytes cultured in low calcium concentrations (0.04 mM) show an undifferentiated, basal cell-like phenotype. Raising calcium concentrations up to 0.14 mM in the medium results in a terminal differentiation process, which is almost identical to the in vivo process. The early differentiation markers, K1 and K10, are observed within 8-24 hrs, and the late markers, loricrin and filaggrin, are shown after 24-48 hrs. A difference not found in vitro is the down-regulation of K14 expression, which takes place in differentiating keratinocytes in vivo but is constitutively expressed in cultured keratinocytes. A relationship between elevated calcium ion concentration and keratinocyte differentiation is observed both in vivo and in vitro, where a calcium gradient has been well documented in human and murine epidermis. Higher calcium levels occur in the differentiated granular and spinous layers as compared to the undifferentiated basal layer. From these findings, the calcium-sensitive signaling pathway, involved in the regulation of keratinocyte differentiation, was elucidated.
The epidermal calcium gradient, however, not only regulates the epidermal differentiation process, but also regulates the homeostasis of the epidermal permeability barrier. Several signals have been suggested for mediating the homeostatic responses after skin barrier disruption. Occlusion of barrier-disrupted skin with water vapor-impermeable membrane could block the lipid synthesis responses to barrier disruption, which also suggests that TEWL is a regulatory signal in barrier homeostasis. When the barrier disrupted skin was immediately exposed to isoosmolar, hyperosmolar or hypoosmolar external solutions, however, the barrier recovery responses were normalized, which suggests that water movement, itself, might not be considered as a major signal in barrier homeostasis. The epidermal calcium gradient, with its highest level in the granular layer and its lowest level in the basal layer, disappears after barrier disruption, and reappears in parallel with barrier restitution. In a previous study, it was shown that immersion of barrier-disrupted skin into a calcium containing solution significantly delayed barrier recovery. The central role of calcium ions in the skin barrier was also demonstrated by manipulating LB secretion by changing the epidermal calcium gradient without changing TEWL, using iontophoresis or sonophoresis. These studies suggest that changes in calcium ion concentration in the stratum granulosum (SG) can directly induce the homeostatic signals for barrier repair, even without a change in permeability barrier function. The precise mechanism for the change of intracellular calcium levels in response to the change of extracellular calcium, however, is still not clear.
Protein kinase C (PKC) comprises a family of serine/threonine kinases that play central roles in the regulation of various cellular processes in numerous cell types. Currently, eleven PKC isotypes have been identified, which can be classified according to their activation factors.21 Human epidermal keratinocytes express several PKC isoforms, such as PKC α, δ, ε and ξ. Other PKC isoforms (PKC β, γ, µ) were found in various other keratinocytes. Previous studies suggest a crucial role for PKC in the regulation of the proliferation and differentiation of keratinocytes. From these studies, PKC expression and activity, which was represented by the change of subcellular localization, was seen to alter with calcium induced keratinocyte differentiation. It was also shown that inhibition of PKC activity with a selective PKC inhibitor suppressed the expression of late differentiation marker proteins, such as involucrin and filaggrin, and terminal differentiation markers, such as keratinocyte-specific transglutaminase-1, in HaCaT keratinocytes. Phorbol 12-myristate 13-acetate (PMA), an activator of PKC, increased the expressions of involucrin, filaggrin, and transglutaminase-1.22 In addition to its crucial role in keratinocyte differentiation, PKC might play some important roles in epidermal permeability barrier function. Previously, Murata et al. reported that the activity of serine palmitoyl transferase and glucosylceramide synthase, both essential for ceramide production, was unregulated by a PKC-dependent mechanism in cultured human keratinocytes.23 Recently, we have also shown that the PKC-δ specific inhibitor, rottlerin, delayed barrier recovery rate by inhibiting the intracellular calcium responses to the loss of the extracellular calcium gradient.24 Although the precise mechanism of PKC involvement in permeability barrier homeostasis is still unknown, it seems that some PKC isozymes are responsible for the regulation of intracellular calcium concentration during the permeability barrier recovery process.
The lamellar body (LB), also known as a lamellar granule, a keratinosome, a membrane-coating granule (MCG), and an Odland body, is a specialized secretory granule in the epidermal keratinocyte and considered to be essential in epidermal permeability barrier formation, as well as desquamation.25 LBs are tubular or ovoid-shaped, membrane-bound organelles unique to mammalian epidermis. The lipid components for SC intercellular lipids, such as glucosylceramide, cholesterol and phospholipids, are delivered through the LBs and their respective extracellular processing enzymes, including β-glucocerebrosidase. Acidic sphingomyelinase and secretory phospholipase A2 (sPLA2) are also packed in LBs.26 Recently, it was shown that other materials as well as lipid precursors, such as antimicrobial peptides, proteins, protein-degrading enzymes and protease inhibitors, are also packed in LBs. Using confocal laser scanning microscopy and immunoelectron microscopy, Ishida-Yanamoto et al. showed that cathepsin D and kallikrein (KLK) 7 are also packed in LBs. They also showed that each LB has its own separate molecules.27 More recently, they also showed that KLK5 and LEKTI (lymphoepithelial Kazal-type-related inhibitor), a putative serine protease inhibitor, is also localized in the LBs.28 The heterogeneous content of the LBs suggests that they are important for not only epidermal permeability barrier functions, but also the other defensive functions exerted by the SC.
While the epidermal LBs play a basic and crucial role in the formation, maintenance and repair of the epidermal permeability barrier, the exact mechanism underlying LB formation in the SG and its extrusion into the SC-SG interface is not yet fully understood. In a previous report, Madison and Howard showed that glucosylceramide, a major component of LB and the precursor of SC ceramides, was produced through the Golgi apparatus and delivered to the plasma membrane, which suggested that the Golgi apparatus is the origin of LB.29 Recently, the role of caveolin proteins in LB formation and secretion has been suggested. The caveolins are cholesterol-binding scaffolding proteins that facilitate the assembly of cholesterol and sphingolipid-enriched membrane domains, known as caveolae. Previously, Sando et al. reported morphological evidence of co-localization of the caveolin and LBs in the SG, which suggests that caveolins may play a role in LB assembly and function.30 Caveolae are a subset of lipid raft domains, which are characterized as flask-shaped invaginations in the plasma membrane of 50 to 100 nm in diameter and, interestingly, Elias et al. observed a structural similarity between the caveolae and the LB secretion site.31 Since the lipid raft provides platforms for preserving the specificity and efficiency of cell signal transduction processes,32 it can be postulated that the LB secretory response to barrier disruption is important not only for replenishing the lipids to the SC intercorneocyte domains, but also for creating higher-order signaling complexes to amplify cellular signaling.33 Recently, numerous studies using caveolin knockout (KO) mice have been reported. Interestingly, it was reported that cavolin-1 KO mice showed no abnormalities in cutaneous phenotype34 and barrier function recovery rate was accelerated after acute permeability barrier disruption,35 which also suggests a close relationship between the caveolin proteins and LB formation and its secretion.
The SC intercellular lipid matrix constitutes about 15% of the dry weight of the SC and the only continuous phase of the skin barrier. The unique lamellar, bilayer organization of this lipid matrix provides the structural basis for the extraordinary lower permeability to water and other electrolytes through the SC.36 The major lipid species of the SC are ceramides, fatty acids, and cholesterol.37 Small amounts of cholesterol esters and cholesterol sulfates are also present in SC and both lipids play a critical role in proper structural organization of SC lipids and, therefore, in normal barrier function.38 The chemical structures of SC lipids species are illustrated in Fig. 2.
The majority of SC lipids are derived from LBs and synthesized in the keratinocyte of the upper stratum spinosum (SS) and SG. At the SC-SG junction, phospholipids, sphingolipids, and other precursor molecules for SC lipids are extruded from the LB to the SC intercellular spaces. Extracellular processing of these lipid precursors provides the SC intercellular lipid matrix. When the epidermal permeability barrier function is compromised, several homeostatic responses follow. The first and most important response is immediate secretion of preformed LBs to the SC-SG interface. Increased synthesis of LBs in the granulocytes and continuing secretion to SG-SC interface result in a restoration of the perturbed barrier function. Synthesis of cholesterol, free fatty acids, and sphingolipids, which are the major constituents for SC intercellular lamellae, is also increased in the epidermis to provide the lipid content for nascent LBs. In addition to the increase of lipid synthesis in the epidermis, the epidermal expression of several cytokines and DNA synthesis is also in creased after acute disruption of barrier function, which could be an additional support for barrier recovery. In murine epidermis, mRNA expression of tumor necrosis factor (TNF)-α, interleukin (IL)-1α, IL-1β and granulocyte macrophage-colony-stimulating factor (GM-CSF) are elevated after barrier disruption (Fig. 3).
For a complete recovery of epidermal permeability barrier function, both the corneocytes of the SC intercellular lipids need to be replenished into the barrier-damaged skin site. While a great deal of studies have been reported on the regulation of lipid synthesis in the stratum granulosum after barrier disruption, relatively little is known about the regulation of keratinocyte differentiation after barrier disruption and its role in barrier homeostasis. Peroxisome proliferator- activated receptor (PPAR) and liver X receptor (LXR) belong to a subset of the nuclear hormone receptor superfamily (class II receptors), which need retinoid X receptor (RXR) as a heterodimerizing factor for their activation.39 PPAR and LXR are ligand-activated transcriptional factors that regulate the expression of target genes involved in many cellular functions including cell proliferation, differentiation and immune/inflammation responses. Along with the fibrate hypolipidemic agents, a large number of endogenous fatty acids and fatty acid metabolites can activate PPAR. Several oxysterols, including cholesterol, can activate LXR.40 Activation of PPAR and LXR in skin, induced various cellular responses including the promotion of keratinocyte differentiation and alleviation of epidermal hyperproliferation, as well as the reduction of epidermal inflammation.41 Recently, it was also shown that topical application of PPAR or LAX activator on barrier-disrupted skin improves epidermal permeability barrier recovery, inducing an increase of epidermal lipid synthesis and LB secretion.42 Since endogenous lipids produced in the keratinocyte can activate PPAR or LXR, and, in turn, regulate lipid metabolism of the keratinocyte, these receptors are currently referred to as 'liposensors' in keratinocytes, and considered major players connecting lipid metabolism and keratinocyte differentiation.
In 1985, Menon et al. reported that an epidermal calcium gradient with low calcium concentrations in the basal, proliferating layers, and a progressively higher concentration as one proceeds to the outer differentiated layers, i.e. the SG.43 Several years later, it was suggested that this epidermal calcium gradient plays a crucial role in skin barrier function.44 Acute barrier disruption with either acetone treatment or repeated tape stripping induced an immediate, marked decrease of intracellular calcium concentrations in the SG. The calcium levels in the SG then progressively normalized in parallel with barrier recovery over 24 hours.45 Recently, Elias et al. reported the relationship of barrier function and the reappearance of the epidermal calcium gradient after acute barrier disruption.46 Immediate application of a vapor-permeable membrane on the barrier-disrupted skin provided an artificial permeability barrier and accelerated epidermal calcium gradient formation. While the exposure of barrier-disrupted skin to a cold environment retarded both the barrier recovery and calcium gradient formation, vapor-permeable membrane application with cold exposure resulted in significant calcium gradient formation after three hours, without barrier recovery. With these results, it can be postulated that barrier status dominantly regulates the formation of the epidermal calcium gradient and calcium gradient formation or maintenance is achieved though passive, ATP-independent processes.
Many studies exploring the relationship between permeability barrier homeostasis and epidermal calcium ions have been reported. Sonophoresis and iontophoresis are both used for increasing the delivery of drugs and other bioactive materials across the SC and were shown to decrease the calcium concentrations in the upper epidermis. While the sonophoresis or iontophoresis-treated skin showed no changes in TEWL, increased secretion and synthesis of LB were observed in both skin layers.47-49 Since the secretion of LB from the SG and consequent accumulation of lamellar materials at the SC-SG junction is a hallmark for barrier homeostatic responses after barrier disruption, these results showed that the regulation of LB secretion is mediated by calcium ions in the SG. Recently, it has been reported that several agonists and antagonists for G-protein-coupled receptors (GPCRs) expressed in keratinocytes, such as the γ-aminobutyric acid receptor (GABA),50 the P2X purinergic receptor,51 the NMDA type glutamate receptor,52 the β2-adrenergic receptor53 and the histamine receptor,54 have accelerating or delaying effects on barrier recovery after acute barrier disruption. Since the major signaling molecules for GPCRs include intracellular calcium ions, it can be concluded that the change of intracellular calcium ion concentration is a major signal for permeability barrier homeostasis. In addition to the GPCR activation-related calcium modulation, we have also shown that topical application of calcium chelating agents onto normal skin induced the barrier homeostatic responses, including loss of the epidermal calcium gradient and LB secretion.55 Topical application of glycolic acid, which does not induce any changes in TEWL in normal murine skin, significantly increased LB secretion56,57 and an in vitro study using cultured keratinocytes suggests that glycolic acid could lower the calcium ion concentration, at least in part, through its chelating effects on cations, such as calcium ions.
It is well known that skin surface pH, which ranges from pH 4.5 to pH 5.5 in humans, is slightly acidic compared to the normal physiologic pH.58 SC acidity, or the "acid mantle" is currently considered to be crucial for establishing the epidermal permeability barrier, as well as for producing the epidermal antimicrobial barrier and controlling SC integrity and cohesion. Various endogenous and/or exogenous pathways are currently suggested for acidifying the pH of the SC. Endogenous factors influencing SC acidification include the generation of free fatty acids from phospholipid hydrolysis catalyzed by secretory phospholipase A2 (sPLA2) and the sodium-proton antiporter-1, NHE1, which is expressed in the outer, nucleated layers of the epidermis.59 Exogenous mechanisms include the generation of free fatty acids by bacterial lipase, free fatty acids derived from sebum60 and eccrine gland-derived products, such as lactic acid (Fig. 4).61
In atopic eczematous lesions, the epidermal permeability barrier is perturbed and the skin pH is also significantly elevated.62 In addition to the perturbation of lipid metabolism and their molecular organization, increased skin pH also induces bacterial growth (e.g. Staphylococcus aureus).
Diverse functions for this acid mantle have been suggested, one of the major ones being antimicrobial action. A few studies relating surface pH and its role in supporting the growth of normal microflora as well as inhibiting skin pathogen growth have been previously reported.63 A typical example where the impaired antimicrobial barrier is correlated with elevated skin surface pH is diaper dermatitis. Due to chronic exposure to urine and feces, diapered, neonatal skin shows a more neutral pH than uncovered skin.64 In addition, pathogens that grow at neutral pH worsen diaper dermatitis,65 resulting in a vicious cycle where neutral pH-enhanced pathogen growth and inflammatory cytokine release combine to produce an inflamed, colonized skin with impaired antimicrobial barriers.66
Another important function of the acid mantle is related to the extracellular processing of LB-derived lipid precursors, which comprise the SC intercellular lipid domain. Among the various important enzymes in SC are β-glucocerebrosidase, acid sphingomyelinase and secreted phospholipase A2 (sPLA2). Each of these functions optimally in an acidic environment and contributes to the generation of the ceramide molecules and free fatty acids required for epidermal permeability barrier homeostasis. Acute disruption of the permeability barrier results in a slight but significant increase in skin surface pH. Along with the barrier recovery, the acid mantle reappears. Recent reports using super-acid or super-base, which could modulate skin surface pH without affecting viable epidermis, also showed that skin surface pH regulated the epidermal barrier homeostasis.67 In addition, maintenance of intact murine SC to a neutral pH produced significant abnormalities in SC integrity and cohesiveness.68 Using neonatal hairless mice, Fluhr et al. showed that topical application of liver X receptor (LXR) activators accelerated SC acidification, as well as stimulated keratinocyte differentiation and improved permeability barrier homeostasis.69 Among the proposed acidification pathways, LXR activators induced the increase of secretory phospholipase A2 (sPLA2) activity in the SC. The above described roles of the SC acid mantle in epidermal permeability barrier function suggest that both barriers are closely related and maintenance of skin surface pH in the normal acidic range is crucial for not only the antimicrobial barrier, but epidermal permeability barrier function as well.
Skin surface pH also affects the desquamation processes. A lot of protein degrading enzymes and proteases exist in the SC and each protease has its optimal pH, respectively (Fig. 5).70,71 Since each protease has its own substrate in the corneodesmosome components, changes in pH induce abnormal desquamation and results in a scaly skin. A typical example of abnormal desquamation induced by alkaline pH is soap-induced xerotic skin.72
In addition, skin pH is also related to the activation of the protease-activated receptor-2 (PAR-2) in skin. The protease-activated receptor (PAR) is a seven transmembrane G-protein-coupled receptor, distinguished by a unique mechanism of self-activation following specific proteolytic cleavage of its extracellular domains.73 It was reported that PAR2 agonist peptides significantly delay epidermal permeability barrier recovery after acute barrier disruption in murine skin,74 possibly due to the increase of intracellular calcium concentration in epidermal keratinocytes. Previous studies also showed that serine protease activity was increased in atopic dermatitis, and it has been postulated that increased PAR-2 activity could be a pathophysiologic factor for atopic dermatitis.75 Recently, we have reported the effects of PAR-2 activators on epidermal barrier functions, as well as on epidermal calcium gradients. Cockroach allergens and house dust mite allergens, both known to be associated with the development of asthma and atopic dermatitis, could activate PAR-2 and thereby induce various cellular responses.76,77 Topically applied PAR-2 activators delayed the barrier recovery, which might be due to the change of intracellular calcium concentration. Since the protease in cockroach and house dust mite allergens have their highest activity at neutral pH, maintaining an acidic pH after barrier disruption significantly prevented the inhibitory effects of both allergens on epidermal permeability barrier recovery.78
Currently, several skin diseases are known to show a defect in epidermal permeability barrier function as a major or minor pathophysiologic factor. While the most representative ones include atopic dermatitis and psoriasis, other skin diseases caused by genetic defects in lipid metabolism are also accompanied by barrier defects. Recessive X-linked lamellar ichthyosis (RXLI) is one such disease. It is caused by a deficiency in the enzyme steroid sulfatase and leads to an accumulation of cholesterol sulfate and a reduction of cholesterol. The abnormality of cholesterol sulfate/cholesterol balance results in an altered lipid organization and consequent abnormality in the structural organization of the intercorneocyte lipid lamellae.79 Cholesterol sulfate is also known to affect other biological processes such as protease activity in the SC, which results in abnormal desquamation in the RXLI.13 In addition to the inhibitory effects on protease in the SC, cholesterol sulfate also acts as an activating factor for the eta isoform of protein kinase C (PKC eta). PKC eta is a subtype of novel PKC and, in the keratinocyte, is closely associated with cell cycle arrest and differentiation.80 It can, therefore, be postulated that cholesterol sulfate is also related to the differentiation of epidermal keratinocytes. Other genetic skin diseases, including Netherton syndrome,81 characterized by defects in proteolysis, and type 2 Gaucher disease, exhibiting defects in β-glucocerebrosidase, involve skin barrier abnormalities as a pathologic outcomes.
While various pathogenic factors are suggested for AD, impaired barrier function is currently considered an important one. Atopic dry skin displays impaired barrier function, indicated by an increased transepidermal water loss and lowered water-binding capacities. Significant decreases in SC ceramide,82 especially in ceramide 1 (CER EOS) levels is reported in AD skin.83 Bouwstra et al. suggested that the ceramide 1 in SC intercellular lipids plays a dominant role in the formation of proper molecular organization.84 In addition, Pilgram et al. reported that the abnormal lipid organization in the SC of AD patients resulted from the decrease of ceramide 1 and suggested that the impaired barrier function of AD skin was, at least in part, due to this structural change.85 Recently, we have reported a structural property of a physiologic lipid mixture containing a pseudoceramide, which showed a close similarity to that of SC intercellular lipids.36 Since the structural property of lipids can affect the epidermal permeability barrier function, it might be beneficial to barrier function to use the physiologic lipid mixture on the barrier-disrupted skin.
The decrease of ceramides in AD patients is linked to an increased expression of sphingomyelin deacylase, which converts the sphingomyelin to free fatty acids and sphingosylphosphorylcholine.86 In normal skin, epidermal sphingomyelin is catalyzed by sphingomyelinase to produce ceramide, but in AD skin, extraordinary sphigomyelin deacylase activity is presented. Recently, the existence of another novel enzyme, termed as glucosylceramide deacylase, has been reported in AD skin lesions. In normal skin, glucosylceramide, secreted into the SC through LBs, is catalyzed by β-glucocerebrosidase to produce ceramide. In AD skin, however, glucosylceramide is catalyzed by this novel enzyme to produce glucosylsphingosine.87 With this enzymatic abnormality, ceramide level is down regulated in AD skin, and permeability barrier function is incompetent (Fig. 6). Altered ceramide metabolism in AD skin also induces a decrease in sphingosine, which is produced from ceramide by ceramidase in the SC. Since sphingosine is known to have potent antimicrobial activities on Staphylococcus aureus at physiologic levels, down regulation of sphingosine is one of the reasons for the vulnerability of AD patients to S. aureus colonization.88
In addition to the abnormalities in lipid processing metabolism, genomic defects can also affect the barrier abnormalities in AD. Recently, it was reported that there is a lack of differentiation-specific gene expression in the keratinocytes in AD skin lesions. Using a large-scale DNA microarray Sugiura et al. showed a significant decrease of mRNA expression of filaggrin and loricrin in AD skin.89 More recently, loss-of-function mutations for keratinocyte-differentiation related-genes encoding filaggrin are observed in AD patients and suggested as an important risk factor in AD.90 As seen in another recent report showing filaggrin gene mutations in several families of ichthyosis vulgaris,91 defects in adequate filaggrin formation in terminal differentiation of epidermal keratinocytes results in abnormal keratinization of skin. Filaggrins are crucial proteins for terminal differentiation of the epidermal keratinocytes, which form a skin barrier with the SC intercellular lipids. In addition, pyrrolidone carboxylic acid (PCA), as a major component of the natural moisturizing factors (NMF) in skin, is primarily produced from filaggrin proteins in the SC and plays a vital role in maintaining the hydration of the SC. It was also previously reported that filaggrin mutation is associated with the dry skin phenotype, which is characterized by a decrease of the NMF components in skin.92 Mutations of filaggrin genes result in abnormal corneocyte structure and impaired barrier function, leading to skin that is more vulnerable to the penetration of exogenous allergens than normal skin. Along with the abnormalities in lipid metabolisms, these findings suggest that both the bricks and the mortar have defects in AD skin, and consequent impairment of epidermal barrier function is an important pathophysiologic factor for AD.93
Another important skin disease with impaired barrier function is psoriasis. In this condition, TEWL are significantly increased, according to the severity of the lesions.94 Alterations in ceramide content and abnormal lipid structures are also reported in psoriasis.95,96 These changes include an increase in ceramide with non-hydroxyl fatty acids and sphingosine (CerNS), ceramide with 6-hydroxy-4-shingenine and omega-hydroxy fatty acid ester linked to linoleic acid (CerEOH) and decreases in ceramide with alpha-hydroxy fatty acids and sphingosine (CerAS). In addition, altered cholesterol and fatty acid levels are also observed in psoriatic lesions. This abnormality in lipid composition and components results in not only the alterations in epidermal permeability barrier function, but also alterations in corneocyte adhesion and desquamation. The cornified lipid envelope (CLE) structure and its components are also changed in psoriasis. While it needs to be clarified whether the abnormal permeability barrier is primary pathophysiologic factors for these skin diseases or not, many studies have now shown that improvements in barrier function resulted in significant amelioration of disease severity.
Epidermal permeability barrier function has the crucial role of maintaining skin homeostasis and the perturbation of barrier function has significant effects on overall skin quality. Recently, many studies have suggested that the defects in permeability barrier function are not secondary consequences, but critical factors for various skin diseases. Therefore, understanding the molecular basis of barrier function and its homeostatic responses can provide not only a more rational therapy of barrier-disrupted skin diseases, but also improve the specificity and efficacy of treatments for human skin with abnormal SC structure and function.
Figures and Tables
 | Fig. 1The localization of various protective functions in the stratum corneum. While the corneocytes (bricks) serve as UV and mechanical barriers as well as playing a hydrating role in the stratum corneum, the intercellular lipids perform the functions of antimicrobial barrier, anti-oxidant barrier and permeability barrier. It should be noted, however, that regulation of these protective functions are closely related and modulation of one function can affect other functions. |
 | Fig. 2The structure of the major components of the stratum corneum intercellular lipids. Numbers 1 to 8 are ceramides and represent a thin layer with chromatographic mobility, with ceramide 1 being least polar and ceramide 8 the most polar. The letters in square brackets are the structural classifications of the ceramide as suggested by Motta et al. (Biochim Biophys Acta 1993;1182:145-51). |
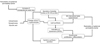 | Fig. 3The epidermal permeability barrier homeostasis. When barrier function is perturbed, immediate secretion of preformed lamellar bodies occurs, followed by an increased synthesis of lamellar bodies and lipid precursors. Increased expression of inflammatory cytokines is observed after barrier disruption. Keratinocyte differentiation and proliferation are also increased after barrier disruption. The epidermal permeability barrier is restored through the above described homeostatic responses. Iontophoresis, sonophoresis and topical glycolic acid can also induce the down-stream homeostatic responses without changing the transepidermal water loss. |
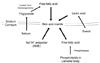 | Fig. 4The endogenous and exogenous factors affecting stratum corneum acidification. Exogenous, free fatty acids are either derived from sweat glands or catalyzed from sebaceous gland-originated triglycerol by microorganism-secreted lipases. Endogenous free fatty acids are derived from phospholipids by phospholipase A2 (PLA2), both of which are secreted by lamellar bodies at the stratum corneum-stratum granulosum junction. The Na+/H+ antiporter is also involved in maintaining the skin acid mantle. |
 | Fig. 5The activation of corneodesmosome degrading protease in the stratum corneum. SCCE/KLK7 and KLK17 are activated by the tryptic cleavage of a short amino-terminal domain of Pro-SCCE and pro-KLK14 by SCTE/KLK5. Pro-SCTE/KLK7 is autoactivated to yield active SCTE/KLK5. The cathepsin activator is unknown. The proteolytic activities of each protease are regulated by their chemical and biochemical environment, including the pH and the protease inhibitors in the stratum corneum. SCTE, stratum corneum tryptic enzyme; SCCE, stratum corneum chymotryptic enzyme; LEKTI, lymphepithelial Kazal-type 5 serine protease inhibitor; SKALP, skin-derived-antileukoprotease; ALP, antileukoproteinase; SLPI, secretory leukocyte protease inhibitor. |
References
1. Albanesi C, Scarponi C, Giustizieri ML, Girolomoni G. Keratinocytes in inflammatory skin diseases. Curr Drug Targets Inflamm Allergy. 2005. 4:329–334.
2. Hearing VJ. Biogenensis of pigment granules: a sensitive way to regulate melanocyte function. J Dermatol Sci. 2005. 37:3–14.
3. Lehmann B, Querings K, Reichrath J. Vitamin D and skin: new aspects for dermatology. Exp Dermatol. 2004. 13:Suppl 4. 11–15.
4. Garssen J, Vandebriel RJ, van Loveren H. Molecular aspects of UVB-induced immunosuppression. Arch Toxicol Suppl. 1997. 19:97–109.
5. Ghoreishi M. Heat shock proteins in the pathogenesis of inflammatory skin diseases. J Med Dent Sci. 2000. 47:143–150.
6. Bowdish DM, Davidson DJ, Hancock RE. A re-evaluation of the role of host defense peptides in mammalian immunity. Curr Protein Pept Sci. 2005. 6:35–51.
7. Braff MH, Bardan A, Nizet V, Gallo RL. Cutaneous defense mechanisms by antimicrobial peptides. J Invest Dermatol. 2005. 125:9–13.
8. Elias PM. Epidermal lipids, barrier function and desquamation. J Invest Dermatol. 1983. 80:Suppl. 44–49.
9. Jarnik M, Simon MN, Steven AC. Cornified cell envelope assembly: a model based on electron microscopic determinations of thickness and projected density. J Cell Sci. 1998. 111:1051–1060.
10. Swartzendruber DC, Wertz PW, Madison KC, Downing DT. Evidence that the corneocytes has a chemically bound lipid envelope. J Invest Dermatol. 1987. 88:709–713.
11. Kalinin AE, Kajava AV, Steinert PM. Epithelial barrier function: assembly and structural features of the cornified cell envelope. Bioessays. 2002. 24:789–800.
12. Komatsu N, Saijoh K, Sidiropoulos M, Tsai B, Levesque MA, Elliott MB, et al. Quantification of human tissue kallikreins in the stratum corneum: dependence on age and gender. J Invest Dermatol. 2005. 125:1182–1189.
13. Elias PM, Crumrine D, Rassner U, Hachem JP, Menon GK, Man W, et al. Basis for abnormal desquamation and permeability barrier dysfunction in RXLI. J Invest Dermatol. 2004. 122:314–319.
14. Schechter NM, Choi EJ, Wang ZM, Hanakawa Y, Stanley JR, Kang Y, et al. Inhibition of human kallikreins 5 and 7 by the serine protease inhibitor lymphoepithelial Kazal-type inhibitor (LEKTI). Biol Chem. 2005. 386:1173–1184.
15. Oren A, Ganz T, Liu L, Meerloo T. In human epidermis, β-defensin 2 is packaged in lamellar bodies. Exp Mol Pathol. 2003. 74:180–182.
16. Braff MH, Di Nardo A, Gallo RL. Keratinocytes stores the antimicrobial peptide cathelicidin in lamellar bodies. J Invest Dermatol. 2005. 124:394–400.
17. Frohm M, Agerberth B, Ahangari G, Stahle-Backdahl M, Liden S, Wigzell H, et al. The expression of the gene coding for the antimicrobial peptide LL-37 is induced in human keratinocytes during inflammatory disorders. J Biol Chem. 1997. 272:15258–15263.
18. Nomura I, Gao B, Boguniewicz M, Darst MA, Travers JB, Leung DY. Distinct patterns of gene expression in the skin lesions of atopic dermatitis and psoriasis: a gene microarray analysis. J Allergy Clin Immunol. 2003. 112:1195–1202.
19. de Jongh GJ, Zeeuwen PL, Kucharekova M, Pfundt R, van der Valk PG, Blokx W, et al. High expression levels of keratinocyte antimicrobial proteins in psoriasis compared with atopic dermatitis. J Invest Dermatol. 2005. 125:1163–1173.
20. Kim M, Jeon H, Goo J, Ahn S, Park Y, Lee S, et al. Expression of anti-microbial peptides in murine epidermis in increased by UVB exposure. J Invest Dermatol. 2006. 126:A66.
21. Parker PJ, Murray-Rust J. PKC at a glance. J Cell Sci. 2004. 117:131–132.
22. Papp H, Czifra G, Lazar J, Gonczi M, Csernoch L, Kovacs L, et al. Protein kinase C isozymes regulate proliferation and high cell density-mediated differentiation in HaCaT keratinocytes. Exp Dermatol. 2003. 12:811–824.
23. Murata S, Uchida Y, Ichikawa S, Hirabayashi Y, Holleran WM. Regulation of glucosylceramide synthase expression in cultured human keratinocyte. J Invest Dermatol. 2000. 114:795.
24. Ahn BK, Jeong SK, Kim HS, Choi KJ, Seo JT, Choi EH, et al. Rottlerin, a specific inhibitor of protein kinase C-delta, impedes barrier repair responses by increasing intracellular free calcium. J Invest Dermatol. 2006. 126:1348–1355.
25. Odland GF, Holbrook A. The lamellar granules of epidermis. Curr Probl Derm. 1981. 9:29–49.
26. Menon GK, Ghadially R, Williams ML, Elias PM. Lamellar bodies as delivery systems of hydrolytic enzymes: implications for normal and abnormal desquamation. Br J Dermatol. 1992. 126:337–345.
27. Ishida-Yamamoto A, Simon M, Kishibe M, Miyauchi Y, Takahashi H, Yoshida S, et al. Epidermal lamellar granules transport different cargoes as distinct aggregates. J Invest Dermatol. 2004. 122:1137–1144.
28. Ishida-Yamamoto A, Deraison C, Bonnart C, Bitoun E, Robinson R, O'Brien TJ, et al. LEKTI is localized in lamellar granules, separated from KLK5 and KLK7, and is secreted in the extracellular spaces of the superficial stratum granulosum. J Invest Dermatol. 2005. 124:360–366.
29. Madison KC, Howard EJ. Ceramides are transported through the Golgi apparatus in human keratinocytes in vitro. J Invest Dermatol. 1996. 106:1030–1035.
30. Sando GN, Zhu H, Wies JM, Richman JT, Wertz PW, Madison KC. Caveolin expression and localization in human keratinocytes suggest a role in lamellar granule biogenesis. J Invest Dermatol. 2003. 120:531–541.
31. Elias PM, Cullander C, Mauro T, Rassner U, Komuves L, Brown BE, et al. The secretory granular cell: The outermost granular cells as a specialized secretory cells. J Investig Dermatol Symp Proc. 1998. 3:87–100.
32. Guest AFG, Leyton L, Parraga M. Caveolins, caveolae, and lipid rafts in cellular transport, signalingm and disease. Biochem Cell Biol. 2004. 82:129–144.
33. Menon GK. Caveolins in epidermal lamellar bodies: skin is an interactive interface, not an inflexible barrier. J Invest Dermatol. 2003. 120:15–16.
34. Capozza F, Williams TM, Schubert W, McClain S, Bouzahzah B, Sotgia F, et al. Absence of caveolin-1 sensitized mouse skin to carcinogen-induced epidermal hyperplasia and tumor formation. Am J Pathol. 2003. 162:2029–2039.
35. Reolandt T, Huth M, Houben E, Crumrine D, Kutsumo A, Giddelo G, et al. Caveolin-1 incorporation into lipid rafts in the outer stratum granulosum suppresses lamellar body secretion in parallel with initiation of terminal differentiation. J Invest Dermatol. 2006. 126:A71.
36. Park BD, Youm JK, Jeong SK, Choi EH, Ahn SK, Lee SH. The characterization of molecular organization of multilmellar emulsions containing pseudoceramide and type III synthetic ceramide. J Invest Dermatol. 2003. 121:794–801.
37. Elias PM. Epidermal barrier function: intercellular lamellar lipid structures, origin, composition and metabolism. J Control Release. 1991. 15:199–262.
38. Bouwstra JA, Gooris GS, Weerheim A, Ponec M. pH, cholesterol sulfate and fatty acids affects the stratum corneum lipid organization. J Invest Dermatol Symp Proc. 1998. 3:69–74.
39. Berger J, Moller DE. The mechanisms of action of PPARs. Annu Rev Med. 2002. 53:409–435.
40. Krey G, Braissant O, L'Horset F, Kalkhoven E, Perroud M, Parker MG, et al. Fatty acids, eicosanoids, and hypolipidemic agents identified as ligands for peroxisome proliferators-activated receptors by coactivator-dependent receptor ligand assay. Mol Endocrinol. 1997. 11:779–791.
41. Elias PM. Stratum corneum defensive functions: an integrated view. J Invest Dermatol. 2005. 125:183–200.
42. Man M-Q, Choi EH, Schmuth M, Crumrine D, Uchida Y, Elias PM, et al. Basis for improved permeability barrier homeostasis induced by PPAR and LXR activators: liposensors stimulate lipid synthesis, lamellar body secretion, and post-secretory lipid processing. J Invest Dermatol. 2006. 126:386–392.
43. Menon GK, Grayson S, Elias PM. Ionic calcium reservoirs in mammalian epidermis: ultrastructural localization by ion-capture cytochemistry. J Invest Dermatol. 1985. 84:508–512.
44. Lee SH, Elias PM, Proksch E, Menon GK, Mao-Quiang M, Feingold KR. Calcium and potassium are important regulators of barrier homeostasis in murine epidermis. J Clin Invest. 1992. 89:530–538.
45. Menon GK, Elias PM, Lee SH, Feingold KR. Localization of calcium in murine epidermis following disruption and repair of the permeability barrier. Cell Tissue Rep. 1992. 270:503–512.
46. Elias PM, Ahn SK, Brown BE, Crumrine D, Feingold KR. Origin of epidermal calcium gradient: regulation of barrier status and role of active vs passive mechanisms. J Invest Dermatol. 2002. 119:1269–1274.
47. Menon GK, Price LF, Bommannan B, Elias PM, Feingold KR. Selective obliteration of the epidermal calcium gradient leads to enhanced lamellar body secretion. J Invest Dermatol. 1994. 102:789–795.
48. Lee SH, Choi EH, Feingold KR, Jiang S, Ahn SK. Iontophoresis itself on hairless mouse skin induces the loss of the epidermal calcium gradient without skin barrier impairment. J Invest Dermatol. 1998. 111:39–43.
49. Choi EH, Kim MJ, Yeh BI, Ahn SK, Lee SH. Iontophoresis and sonophoresis stimulate epidermal cytokine expression at energies that do not provoke a barrier abnormality: lamellar body secretion and cytokine expression are linked to altered epidermal calcium levels. J Invest Dermatol. 2003. 121:1138–1144.
50. Denda M, Inoue K, Inomata S, Denda S. γ-aminobutyric acid (A) receptor agonists accelerate cutaneous barrier recovery and prevent epidermal hyperplasia induced by barrier disruption. J Invest Dermatol. 2002. 119:1041–1047.
51. Denda M, Inoue K, Fuziwara S, Denda S. P2X purinergic receptor antagonist accelerates skin barrier repair and prevents epidermal hyperplasia induced by skin barrier disruption. J Invest Dermatol. 2002. 119:1034–1040.
52. Fuziwara S, Inoue K, Denda M. NMDA-type glutamate receptor is associated with cutaneous barrier homeostasis. J Invest Dermatol. 2003. 120:1023–1029.
53. Denda M, Fuziwara S, Inoue K. β2-adrenergic receptor antagonist accelerates skin barrier recovery and reduces epidermal hyperplasia induced by barrier disruption. J Invest Dermatol. 2003. 121:142–148.
54. Ashida Y, Denda M, Hirao T. Histamine H1 and H2 receptor antagonists accelerate skin barrier repair and prevent epidermal hyperplasia induced by barrier disruption in a dry environment. J Invest Dermatol. 2001. 116:261–265.
55. Jeong SK, Ko JY, Seo JT, Ahn SK, Lee CW, Lee SH. Stimulation of epidermal calcium gradient loss and increase in TNF-α and IL-1α expressions by glycolic acid in murine epidermis. Exp Dermatol. 2005. 4:571–579.
56. Kim TH, Choi EH, Kang YC, Lee SH, Ahn SK. The effects of topical α-hydroxyacids on the normal skin barrier of hairless mice. Br J Dermatol. 2001. 144:267–273.
57. Jeong SK, Kim S, Lee EH, Choi EH, Ahn SK, Lee SH. Comparison of the effect of various chemical peeling agents on the skin barrier. Korean J Dermatol. 2002. 40:1181–1187.
58. Parra JL, Paye M. EEMCO Group. EEMCO guidance for the in vivo assessment of skin surface pH. Skin Pharmacol Appl Skin Physiol. 2003. 16:188–202.
59. Fluhr JW, Behne MJ, Brown BE, Moskowitz DG, Seldel C, Man MQ, et al. Stratum corneym acidification in neonatal skin: secretory phospholipase A2 and the sodium/hydrogen antiproter-1 acidify neonatal rat stratum corneum. J Invest Dermatol. 2004. 122:320–329.
60. Bibel DJ, Miller SJ, Brown BE, Pandey BB, Elias PM, Shinefield HR, et al. Antimicrobial activity of stratum corneum lipids from normal and essential fatty acid-deficiency mice. J Invest Dermatol. 1989. 92:632–638.
61. Thueson DO, Chan EK, Oechsli LM, Hahn GS. The roles of pH and concentration in lactic acid-induced stimulation of epidermal turnover. Dermatol Surg. 1998. 24:641–645.
62. Rippke F, Schreiner V, Doering T, Maibach HI. Stratum corneum pH in atopic dermatitis: impact on skin barrier function and colonization with Staphylococcus aureus. Am J Clin Dermatol. 2004. 5:217–223.
63. Korting HC, Hubner K, Greiner K, Hamm G, Barun-Falco O. Differences in the skin surface pH and bacterial microflora due to long-term application of synthetic detergent preparations of pH 5.5 and pH 7.0. Results of a crossover trial in healthy volunteers. Acta Derm Venereol. 1990. 70:429–431.
64. Berg RW, Milligan MC, Sarbaugh FC. Association of skin wetness and pH with diaper dermatitis. Pediatr Dermatol. 1994. 11:18–20.
65. Ferrazzini G, Kaiser RR, Hirsig Cheng SK, Wehrli M, Della Casa V, Pohlig G, et al. Microbiological aspects of diaper dermatitis. Dermatology. 2003. 206:136–141.
66. Mauro TM. Eilas PM, Feingold KR, editors. SC pH: measurement, origins, and functions. Skin Barrier. 2006. New York: Taylor & Francis Group;223–230.
67. Hachem JP, Behne M, Aronchik I, Demerjian M, Feingold KR, Elias PM, et al. Extracellular pH controls NHE1 expression in epidermis and keratinocytes: implications for barrier repair. J Invest Dermatol. 2005. 125:790–797.
68. Fluhr JW, Kao J, Jain M, Ahn SK, Feingold KR, Elias PM. Generation of free fatty acids from phospholipids regulates stratum corneum acidification and integrity. J Invest Dermatol. 2001. 117:44–51.
69. Fluhr JW, Crumrine D, Man MQ, Moskowitz DG, Elias PM, Feingold KR. Topical liver X receptor activators accelerate postnatal acidification of stratum corneum and improve function in the neonate. J Invest Dermatol. 2005. 125:1206–1214.
70. Caubet C, Jonca N, Brattsand M, Guerrin M, Bernard D, Schmidt R, et al. Degradation of corneodesmosome proteins by two serine protease of the kallikrein family, SCTE/KLK5/hK5 and SCCE/KLK7/hK7. J Invest Dermatol. 2004. 122:1235–1244.
71. Brattsand M, Stefansson K, Lunhd C, Haasum Y, Egelrud T. A proteolytic cascade of kallikreins in the stratum corneum. J Invest Dermatol. 2005. 124:198–203.
72. Barel AO, Lambrecht R, Clarys P, Morrison BM, Paye M. A comparative study of the effects on the skin of a classical bar soap and a syndet cleansing bar in normal use conditions and in the soap chamber test. Skin Res Technol. 2001. 7:98–104.
73. Macfarlane SR, Seatter MJ, Kanke T, Hunter GD, Plevin R. Proteinase-activated receptors. Phamacol Rev. 2001. 53:245–282.
74. Hachem J, Uchida Y, Crumrine D, Choi E, Houben E, Brown BE, et al. Serine protease-induced alterations in permeability barrier homeostasis are mediated by protease-activated receptor 2. J Invest Dermatol. 2005. 124:A58.
75. Steinhoff M, Neisius U, Ikoma A, Fartasch M, Heyer G, Skov PS, et al. Proteinase-activated receptor-2 mediates itch: A novel pathway for pruritus in human skin. J Neurosci. 2003. 23:6176–6180.
76. Hong JH, Lee SI, Kim KE, Yong TA, Seo JT, Sohn MH, et al. German cockroach extract activates protease-activated receptor 2 in human airway epithelial cells. J Allergy Clin Immunol. 2004. 113:315–319.
77. Asokananthan N, Graham PT, Stewart DJ, Bakker AJ, Eidne KA, Thompson PJ, et al. House dust mite allergens induce proinflammatory cytokines from respiratory epithelial cells: the cysteine protease allergen, Der p 1, activates protease-activated receptor (PAR)-2 and inactivates PAR-1. J Immunol. 2002. 169:4572–4578.
78. Jeong SK, Jeon JE, Kim HJ, Ahn SK, Choi EH, Cho IS, et al. Aeroallergens, as a protease activated receptor-2 activator, delayed epidermal permeability barrier recovery. (under submission).
79. Bouwstra JA, Honeywell-Nguyen L, Gooris GS, Ponec M. Structure of the skin barrier and its modulation by vesicular formulations. Progress Lipid Res. 2003. 42:1–36.
80. Kashiwaga M, Ohba M, Chida K, Kuroki T. Protein kinase C eta (PKC eta): its involvement in keratinocyte differentiation. J Biochem (Tokyo). 2002. 132:853–857.
81. Chavanas S, Bodemar T, Rochat A, Hamel-Teillac D, Ali M, Irvine AD, et al. Mutations in SPINK5, encoding a serine protease inhibitorm cause Netherton syndrome. Nat Genet. 2000. 25:141–142.
82. Imokawa G, Abe A, Jin K, Higari Y, Kawashima M, Hidano A. Decreased levels of ceremides in stratum corneum of atopic dermatitis: an etiological factor in atopic dermatitis. J Invest Dermatol. 1991. 96:523–526.
83. Yamamoto A, Serizawa S, Ito M, Sato Y. Stratum corneum lipid abnormalities in atopic dermatitis. Arch Dermatol Res. 1991. 283:219–223.
84. Bouwstra JA, Gooris GS, Dubbelaar FER, Weerheim AM, Ijzerman AP, Ponec M. Role of ceramide 1 in the molecular organization of the stratum corneum lipids. J Lipids Res. 1998. 39:186–196.
85. Pilgram GSK, Vissers DCJ, van der Meulen H, Pavel S, Lavijsen SPM, Bouwstra JA, et al. Aberrant lipid organization in stratum corneum of parients with atopic dermatitis and lamellar ichthyosis. J Invest Dermatol. 2001. 117:710–717.
86. Hara J, Higuchi K, Okamoto R, Kawashima M, Imokawa G. High-expression of sphigomyelin deacylase is an important determinant of ceramide deficiency leading to barrier disruption in atopic dermatitis. J Invest Dermatol. 2002. 115:406–413.
87. Ishibashi M, Arikawa J, Okamoto R, Kawashima M, Takagi Y, Ohguchi K, et al. Abnormal expression of the novel epidermal enzyme, glucosylceramide deacylase, and the accumulation of its enzymatic reaction product, glucosylsphingosine, in the skin of patients with atopic dermatitis. Lab Invest. 2003. 83:397–408.
88. Arikawa J, Ishibashi M, Kawashima M, Takagi Y, Ichikawa Y, Imokawa G. Decreased levels of sphigosine, a natural antimicrobial agent, may be associated with vulnerability of the stratum corneum from patients with atopic dermatitis to colonization by Staphylococcus aureus. J Invest Dermatol. 2002. 119:433–439.
89. Sugiura H, Ebise H, Tazawa T, Tanaka K, Sugiura Y, Uehara M, et al. Large-scale DNA microarray analysis of atopic skin lesions shows overexpression of an epidermal differentiation gene cluster in the alternative pathway and lack of protective gene expression in the cornified envelope. Br J Dermatol. 2005. 152:146–149.
90. Palmer CN, Irvine AD, Terron-Kwiatkowski A, Zhao Y, Liao H, Lee SP, et al. Common loss-of-function variants of the epidermal barrier protein filaggrin are a major predisposing factor for atopic dermatitis. Nat Genet. 2006. 38:441–446.
91. Smith FJ, Irvine AD, Terron-Kwiatkowski A, Sandilands A, Campbell LE, Zhao Y, et al. Loss-of-function mutations in the gene encoding filaggrin cause ichthyosis vulgaris. Nat Genet. 2006. 38:337–342.
92. Ginger RS, Blachford S, Rowland J, Rowson M, Harding CR. Filaggrin repeat number polymorphism is associated with a dry skin phenotype. Arch Dermatol Res. 2005. 297:235–241.
93. Hudson T. Skin barrier function and allergic risk. Nat Genet. 2006. 38:399–400.
94. Tagami H, Yoshikuni K. Interrelationship between water barrier and reservoir function in pathologic stratum corneum. Arch Dermatol. 1985. 121:642–645.
95. Motta S, Monti M, Sesana S, Mellesi L, Ghidoni R, Caputo R. Abnormality in water barrier function in psoriasis: role of ceramide fraction. Arch Dermatol Res. 1994. 130:452–456.
96. Ghadially R, Reed JT, Elias PM. Stratum corneum structure and function correlates with phenotype in psoriasis. J Invest Dermatol. 1996. 107:558–564.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download