Abstract
Eosinophilic gasteroenteritis is an uncommon disease with variable clinical features characterized by eosinophilic infiltration. Clinical manifestations range from non-specific gastrointestinal complaints such as nausea, vomiting, crampy abdominal pain, and diarrhea to specific findings such as malabsorption, protein loosing enteropathy, luminal obstruction, eosinophilic ascites and effusion. We report here on a case of eosinophilic gastroenteritis causing enterobiliary fistula which is an extremely unusual complication.
Eosinophilic gastroenteritis is a relatively uncommon disease that's characterized by eosinophilic infiltration of the gastrointestinal tract, peripheral eosinophilia and abnormal gastrointestinal function. Since it was first recognized by Kaijer in 1937, more than 400 cases of eosinophilic gastroenteritis have been reported in the literature (1). Most of the clinical signs and symptoms are related to the gastrointestinal tract, e.g., vomiting, abdominal pain, diarrhea, malabsorption, obstruction and ascites. Although various signs and symptoms have been previously reported, to the best of our knowledge, enterobiliary fistula as a complication of eosinophilic gastroenteritis has not yet been reported on. We report here on a case of enterobiliary fistula that was complicated with eosinophilic gastroenteritis, and this was demonstrated by an upper gastrointestinal series (UGI) and magnetic resonance cholangiopancreatography (MRCP).
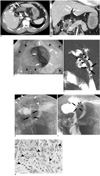
A 68-year-old man presented with a 3-week history of abdominal discomfort, nausea and skin eruption on his entire body. There was no past medial history of hepatolithiasis, recurrent peptic ulcer, abdominal operation, allergic disease, generalized skin eruption or food sensitivity. The laboratory investigations revealed a white blood cell count of 12.86 × 103/µl. There was peripheral eosinophilia ranging from 7% to 52% (normal: 0-7.2%). The total IgE was 593 IU/ml (normal: 0-170 IU/ml). Stool examination and an enzyme-linked immunosorbent assay (ELISA) test to detect parasite infestation showed negative results. The blood urea nitrogen (BUN) was 40 mg/dl (normal: 10-26 mg/dl) and the serum creatine was 2.1 mg/dl (normal: 0.6-1.3 mg/dl). An ultrasound scan for evaluation of acute renal failure yielded negative results except for pneumobilia. Because of the absence of prior sphincterotomy, a surgical bypass procedure or recent endoscopic retrograde cholangiopancreatography (ERCP), we inferred the existence of enterobiliary fistula. The acute renal failure was slightly improved and we then performed multidetector computed tomography (MDCT) for further evaluation. MDCT revealed diffuse submucosal thickening from the antrum of the stomach to the duodenal bulb and a bezoar at the first portion of duodenum. But enterobiliary fistula was not clearly detected (Figs. 1A, B). Gastrointestinal endoscopy showed large ulcers with a geographic pattern from the antrum of the stomach to the duodenal bulb (Fig. 1C). The duodenum revealed severe stricture involving the second portion and a soft bezoar at the first portion of the duodenum. MRCP demonstrated that there was pneumobilia in the gall bladder and common bile duct (CBD). In addition, cholecystoduodenal fistula and a suspected choledocoduodenal fistula were observed (Fig. 1D). The results of the UGI were similar to those of the MRCP, and the other findings were mucosal fold thickening and nodularity involving the antrum and severe stricture involving the second portion of the duodenum (Figs. 1E, F). Biopsy samples were obtained at the ulcer bed, and they showed marked eosinophilic infiltration (Fig. 1G). We adopted the criteria shown in Table 1 for evaluating the tissue eosinophilia; severe eosinophilia (over 70 eosinophils/× 400 field) was demonstrated in the lamina propria of the gastric mucosa. The diagnosis was enterobiliary fistula complicating eosinophilic gastroenteritis. After steroid therapy, there was interval improvement of both the ulcer and duodenal luminal narrowing on the follow up endoscopy.
Eosinophilic gastroenteritis is a rare heterogeneous disorder of an undetermined etiology. An allergic mechanism to food or other allergens has been suggested (2). Increased total IgE and food specific IgE levels have been detected in the majority of patients.
Eosinophilic gastroenteritis presents various clinical features depending on what layers of the bowel wall are infiltrated by the eosinophils (3). The mucosal form, which is the most common type, is characterized by infiltration of eosinophils predominantly in the mucosal layer, and this leads to diarrhea, blood loss in the stool, abdominal pain, vomiting, protein-losing enteropathy, malabsorption and iron-deficiency anemia. The muscularis form demonstrates thickening of the bowel wall, which might result in luminal narrowing or obstruction. This finding may simulate carcinoma, lymphoma or Crohn's disease. Because the gastric biopsy specimens in our case were obtained from the mucosal layer, eosinophilic infiltration of the muscle layer could not be confirmed. But considering these findings, that is, diffuse submucosal thickening from the antrum of the stomach to the duodenal bulb on CT and duodenal luminal narrowing on the UGI, our case was thought to be the muscularis form. The serosal form rarely demonstrates exudative ascites, pleural effusions with higher peripheral eosinophil counts and the best response to steroids, as compared with the other forms (4).
Enterobiliary fistulas represent a complication of cholelithiasis or choledocholithiasis in 90% of cases (5). The infrequent causes include peptic ulcer disease, malignancy and prior surgery. In our case, because there were no other reasons, we deduced that the ulcerations induced by the eosinophilic gastroenteritis caused the enterobiliary fistula. Biliary tract disease, as a rare manifestation of eosinophilic gastroenteritis, has been reported on (1). Similarly, enterobiliary fistula is considered to be a very rare complication.
The diagnosis of eosinophilic gastroenteritis requires the presence of three criteria: symptoms related to the gastrointestinal tract, biopsies showing eosinophilic infiltration of one or more areas from the esophagus to the rectum, and exclusion of other disorders that have similar features, such as malignancies, parasitic, allergic, autoimmune disorder, inflammatory bowel disease (Crohn's disease or ulcerative colitis) and hypereosinophilc syndrome (6). Other previous reports have noted that peripheral eosinophilia is not uniformly associated with eosinophilic gasteroenteritis (4). For that reason, the definite diagnosis of eosinophilic gastroenteritis requires the histologic findings of eosinophilic infiltration. The severe eosinophilia, as was shown in our case, should be demonstrated for a definitive diagnosis of eosinophilic gastroenteritis (7).
The UGI may demonstrate enlargement of the rugal folds, and narrowing and rigidity of the antrum (8). CT scans may show focal or diffuse thickening of the folds, especially at the antrum, and angulation and separation of the intestinal loops (9). Eosinophilic infiltration of the muscle layer results in thickening and rigidity of the bowel wall.
Once the correct diagnosis has been made, corticosteroid therapy usually results in rapid improvement (10). Yet the duration of treatment remains controversial and long-term follow up is required since recurrent symptoms commonly develop after an asymptomatic interval of several years (2). Surgery may be unavoidable when obstruction or perforation occurs or when a definitive diagnosis cannot be made (11).
Eosinophilic gastroenteritis shows diverse clinical manifestations. Although enterobiliary fistula complicating eosinophilic gastroenteritis is a very rare complication, radiologists should suspect this entity when pneumobilia with unexplained gastrointestinal symptoms and peripheral eosinophilia are demonstrated in a patient.
Figures and Tables
Fig. 1
Enterobiliary fistula in 68-year-old man.
A. Axial enhanced multidetector CT scan shows thickening of gastric wall (white arrows) and air in gall bladder (white arrowheads).
B. Coronal multiplanar reformatted multidetector CT image demonstrates pneumobilia (white arrows) and bezoar at duodenal bulb (white arrowheads).
C. Gastrointestinal endoscopy demonstrates bezoar (white arrows) and geographic ulceration at duodenal bulb (black arrowheads).
D. MR cholangiopancreatography coronal image of single-shot turbo spin-echo sequence shows linear high signal intensity from duodenal bulb to gall bladder, which is thought to be cholecystoduodenal fistula (black arrowheads). There is another area of faint linear high signal intensity, suggesting choledocoduodenal fistula (black arrows).
E. Upper GI reveals mucosal fold thickening and nodularity in antrum of stomach (white arrowheads) and severe narrowing involving second portion of duodenum (black arrows).
F. There are two linear structures (black arrows and black arrowheads) that originate from duodenal bulb. We can not trace definite connection to biliary system, but they are thought to be enterobiliary fistulas.
G. Microscopic finding shows marked eosinophilic infiltration (over 70 eosinophils/HPF) in lamina propria of gastric mucosa (black arrowheads) (Hematoxylin & Eosin staining, × 400).
References
1. Jimenez-Saenz M, Villar-Rodriguez JL, Torres Y, Carmona I, Salas-Herrero E, Gonzalez-Vilches J, et al. Biliary tract disease: a rare manifestation of eosinophilic gastroenteritis. Dig Dis Sci. 2003. 48:624–627.
2. Jaffe JS, James SP, Mullins GE, Braun-Elwert L, Lubensky I, Metcalfe DD. Evidence for an abnormal profile of interleukin-4 (IL-4), IL-5, and gamma-interferon (gamma-IFN) in peripheral blood T cells from patients with allergic eosinophilic gastroenteritis. J Clin Immunol. 1994. 14:299–309.
3. Klein NC, Hargrove RL, Sleisenger MH, Jeffries GH. Eosinophilc gastroenteritis. Medicine (Baltimore). 1970. 49:299–319.
4. Talley NJ, Shorter RG, Phillips SF, Zinsmeister AR. Eosinophilic gasteroenteritis. a clinicopathological study of patients with disease of the mucosa, muscle layer, and subserosal tissues. Gut. 1990. 31:54–58.
5. Inal M, Oguz M, Aksungur E, Soyupak S, Boruban S, Akgul E. Biliary-enteric fistulas: report of five cases and review of the literature. Eur Radiol. 1999. 9:1145–1151.
6. Khan S, Orenstein SR. Eosinophilic gastroenteritis: epidemiology, diagnosis and management. Paediatr Drugs. 2002. 4:563–570.
7. Whitington PF, Whitington GL. Eosinophilic gastroenteropathy in childhood. J Pediatr Gastroenterol Nutr. 1988. 7:379–385.
8. Culver GJ, Pirson HS, Montez M, Palanker HK. Eosinophilic gastritis. JAMA. 1967. 200:641–642.
9. Pringot J, Bodart P. Margulis AR, Burhenne HJ, editors. Inflammatory diseases. Alimentary tract radiology. 1989. 4th ed. St. Louis: Mosby;809–810.
10. Lee M, Hodges WG, Huggins TL, Lee EL. Eosinophilic gastroenteritis. South Med J. 1996. 89:189–194.
11. Naylor AR. Eosinophilic gastroenteritis. Scott Med J. 1990. 35:163–165.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download