Abstract
Objective
The aim of our study was to describe and compare the radiologic findings of esophageal leiomyomas.
Materials and Methods
The chest radiographic (n = 12), esophagographic (n = 12), CT (n = 12), and MR (n = 1) findings of surgically proven esophageal leiomyomas in 12 consecutive patients [ten men and two women aged 34 - 47 (mean, 39) years] were retrospectively reviewed.
Results
The tumors, surgical specimens of which ranged from 9 to 90 mm in diameter, were located in the upper (n = 1), middle (n = 5), or lower esophagus (n = 6). In ten of the 12 patients, chest radiography revealed the tumors as mediastinal masses. Esophagography showed them as eccentric, smoothly elevated filling defects in 11 patients and a multilobulated encircling filling defect in one. In 11 of the 12 patients, enhanced CT scans revealed a smooth (n = 9) or lobulated (n = 2) tumor margin, and attenuation was homogeneously low (n = 7) or iso (n = 4). In one patient, the tumor signal seen on T2-weighted MR images was slightly high.
Although it is the most common benign esophageal tumor, esophageal leiomyoma is relatively rare, its overall incidence being 8-43 per 10,000 autopsy series (1, 2).
The reported radiographic findings of esophageal leiomyoma describe it as a smoothly marginated, round or lobulated mass projecting to one or both sides of the mediastinum along the course of the esophagus (3, 4). Esophagography reveals it as a localized intramural mass contrasting with the surrounding air column (4, 5).
The CT findings of esophageal leiomyoma have been described in several case reports (4, 6) and in one major paper (7). According to these articles, it is seen as a homogeneous intramural mass or esophageal wall thickening. No CT reports, however, have detailed the findings in a relatively large series of esophageal leiomyomas. Our study aims to describe and compare the chest radiographic, esophagographic, CT and MR findings of esophageal leiomyoma in 12 patients.
A search of a pathologic database detailing cases occurring between January 1995 and March 2000 revealed 18 cases of esophageal leiomyoma. In six of these, only endoscopic biopsy without surgery was performed and imaging studies had thus not been undertaken. The current study includes the 12 cases in which the tumor was surgically removed; twelve patients were involved, namely ten men and two women aged between 34 and 47 (mean, 39) years. Seven of the 12, in whom the tumor was detected incidentally on routine chest radiographic examinations, were asymptomatic. The remaining five complained of dysphagia (n = 3), substernal pain (n = 2), and epigastric discomfort (n = 2). Esophagography indicated that tumor diameter ranged from 9 to 90 (mean, 47.2) mm.
Initial chest radiographs (n = 12), esophagograms (n = 12), CT scans (n = 12), and MR images (n = 1) were available. Chest radiographs were obtained using a standard posteroanterior projection with a high-kilovoltage technique (125 kVp, 5 mAs, 10-12:1 grid ratio), while for esophagography routine double contrast and mucosal relief study with 200-250% thick barium suspension was used.
For most CT scanning, a GE 9800 or HiSpeed Advantage scanner (General Electric Medical Systems, Milwaukee, Wis., U.S.A) was used; enhanced scans (n = 12), after the injection of 30 g of iodinated contrast medium (Iopamidol, Bracco, Milan, Italy), and unenhanced (n = 10) CT scans from the lung apices to the middle portion of both kidneys were obtained, with 7-10 mm collimation. Before acquiring conventional CT images, canography was used to obtain additional high-resolution (1-mm collimation, 5-mm interval) CT scans at the lesion sites of five patients. In one of the five, in whom the initial CT diagnosis was bronchogenic cyst, enhanced and unenhanced T1- and T2-weighted MR images were also obtained.
The findings of chest radiography, esophagography, CT and MRI were retrospectively analyzed by two thoracic radiologists and one gastrointestinal radiologist working as a team, and decisions on the findings were reached by consensus. The analysis of chest radiographs included determination of the presence or absence of a tumor and of the obliteration or displacement of mediastinal interfaces and lines. In analyzing esophagograms, determination of the location and shape of the main tumor was included. Location was subcategorized as the upper, middle, or lower esophagus. A tumor was presumed to be in the upper esophagus if located proximal to the aortic arch, in the middle esophagus if between the aortic arch and inferior pulmonary vein, and in the lower esophagus if distal to the right inferior pulmonary vein (8). Analysis of the CT findings included a tumor's size, shape, margin, homogeneity, attenuation value and degree of enhancement. Its longest diameter, where it appeared largest on axial images, was measured, and it was classified as smooth or lobulated. Tumor attenuation was compared to that of chest wall muscle before and after the injection of contrast medium. The presence or absence of intratumoral necrosis or calcification and of adjacent fat obliteration, peritumoral invasion, pleural effusion, and lymph node enlargement was also noted. The MRI findings were analyzed in terms of signal intensity and tumor enhancement.
Chest radiography detected tumors in 10 of 12 patients, with normal findings in two. The tumors were seen as soft tissue masses with obliteration or displacement of the azygo-esophageal recess interface (n = 8) (Fig. 1), the aortopulmonary window interface (n = 2), the left paraspinal line (n = 2), the descending aortic interface (n = 2), the superior vena cava interface (n = 1), or the posterior junctional line (n = 1).
Esophagography demonstrated the presence of all tumors. They were located in the upper (n = 1), middle (n = 5), or lower (n = 6) esophagus and were seen as intramural, eccentric, smoothly elevated, sessile lesions of various sizes in 11 patients (Figs. 1 and 2) and in the other, in whom CT showed the lesions to be circumferential, as a multilobulated, encircling, soft tissue mass with passage disturbance and mild mural rigidity (Fig. 3). Neither mucosal destruction nor constricting luminal narrowing was observed in any patient.
One tumor, found at surgery to be 9 mm in its longest diameter, was not revealed by CT. The longest diameter of the remaining 11 ranged from 10 to 90 mm; nine were exophytic and eccentric (Figs. 1 and 3) and two were circumferential (Figs. 2 and 3). They had either a smooth (n = 9) (Figs. 1 and 4) or a lobulated (n = 2) (Figs. 2 and 3) margin. The tumors seen in nine of ten unenhanced scans were homogeneous and showed iso (n = 5) or low attenuation (n = 4) (Figs. 2-4). In five of these cases, the attenuation value ranged from 21 to 39 (mean, 28) HU. Eleven of 12 enhanced scans showed homogeneous enhancement, and compared to chest wall muscle, attenuation was slightly lower (n = 7) or iso (n = 4) (Figs. 1, 2, and 4). In six such patients, attenuation values ranged from 25 to 51 (mean, 37) HU. Multinodular calcification was seen in one patient but central necrosis, adjacent fat obliteration or peritumoral invasion was not noted and there was neither mediastinal nor hilar lymph node enlargement.
In one patient, unenhanced T1-weighted axial MR images demonstrated slight hyperintensity, while enhanced T1-weighted MR images showed slight homogeneous enhancement. On T2-weighted MR images, the lesion was slightly hyperintense (Fig. 1).
Grossly, leiomyomas form well-defined masses in the esophageal wall and their cut surface has a solid, grayish-white appearance. If their growth is intraluminal, they encroach on the mucosa and appear as sessile or pedunculated polyps. Ulceration of the overlying mucosa is rare, in contrast to its common occurrence in gastric leiomyomas. Esophageal leiomyomas can encircle the entire esophagus in its lower third, and microscopically, have the usual characteristics of a benign smooth muscle tumor. Local resection or enucleation is usually successful (9, 10).
Esophageal leiomyomas are usually solitary, ranging from 2 to 6 cm in diameter. Especially when large, they may be recognized as posterior mediastinal masses with a mass effect on an adjacent organ (3, 4). Multiple esophageal leiomyomas are rare (11-13), and though have been reported in a patient with Alport syndrome (13). Esophagography reveals them, in profile, as smoothly round or crescentic filling defects. When viewed en face, they appear as round or lobulated filling defects sharply outlined by the barium flowing around each side. Sharp demarcation between their edges and the involved esophagus is characteristic. The overlying mucosal folds are normal, showing no evidence of infiltration, ulceration, or undercutting at the tumor margin (4, 5).
According to the reported CT findings, esophageal leiomyomas are smoothly marginated, round or ovoid masses of muscle attenuation, lying intramurally or eccentrically within the esophageal wall. Surrounding mediastinal fat is not usually disrupted (4, 6). In our study, enhanced scans showed that tumor attenuation was homogeneously low or iso and, characteristically, the same even when contrast medium was administered. These findings of homogeneity and slight enhancement contrast with those of uterine leiomyomas, which show recognizable enhancement and heterogeneity owing to calcification, hyaline and cystic degeneration, infection or necrosis (14). This difference in the degree of enhancement is presumed to be due to the relatively small vascular supply of the esophageal tumors. Esophageal leiomyomatosis, occurring in children and young adults, may appear as marked wall thickening at CT (12), and the distal third or half of the esophagus and occasionally the proximal stomach are involved. In our study, a circumferential esophageal mass was revealed by CT in two patients. Its lobulated margin and uneven thickness of the mass were, however, different from the findings observed in leiomyomatosis. Furthermore, the tumors appeared as eccentric or multilobulated encircling filling defects. In esophageal leiomyomatosis, esophagography reveals smooth, tapered narrowing of the distal esophagus (12). In our study, multinodular calcification was observed in one (8%) of 12 patients, a frequency similar to that mentioned in a previous report (1/9, 11%) (6).
There have been few published accounts of the MR findings of esophageal leiomyomas. On T2-wighted images their signal is usually iso intense, while in esophageal cancer a high-intensity signal is normally observed (15). In our study, T2-wighted images showed that the lesion was slightly more hyperintense than chest wall muscle.
Although leiomyoma of the esophagus is relatively rare, it is the most common benign tumor involving this structure. In a series of 7,459 autopsies, Moersch and Harrington (2) detected only 44 benign tumors of the esophagus. Thirty-two (73%) of these, however, were leiomyomas. In a series of 103 benign esophageal tumors obtained from surgical and autopsied patients, Totten et al. (16) found 46 (45%) of 103 tumors to be leiomyomas.
Approximately 90 percent of esophageal leiomyomas occur in patients between 20 and 60 years old (3, 4); in our study, patient age ranged from 34 to 47 (mean, 39) years. Surgical incidence peaks during the third and fourth decades, indicating that symptomatic tumors are confined primarily to early adult and middle life. The tumors have a slight predilection for the male sex, with some series reporting a male to female ratio as high as 2:1. In our study, this ratio was 5:1 (10 men and 2 women).
Approximately 60% of leiomyomas occur in the lower third of the esophagus, 33% in the middle third and 7% in the upper third (17). Their relative rarity in the upper esophagus reflects the smaller amount of smooth muscle in that region. In our study, one, five and six tumors occurred in the upper, middle and lower esophagus, respectively.
About 50 percent of patients remain asymptomatic, while varying degrees of dysphagia and substernal pain are the most common presenting symptoms. A tumor's size and growth characteristics are the principal factors governing the appearance of symptoms. Reviews of surgically treated cases suggest the average size of symptomatic tumors is 6-8 cm, though, tumors twice as large as this are common (3, 4, 17). Esophageal leiomyomas grow slowly and usually produce intermittent, insidiously progressive symptoms. Occasionally, they encircle the esophagus in a serpentine or U-shaped fashion and cause obstructive symptoms.
Recourse to surgery depends on a tumor's size and location, as well as mucosal fixation, stomach involvement, and its adherence to contiguous structures (18). Enucleation without entering the esophageal lumen, or esophageal resection, is successful even with a large tumor (19). In our study, all patients underwent enucleation, regardless of tumor size. There is no debate as to whether symptomatic esophageal leiomyomas should be removed, but there is no consensus as to whether asymptomatic small leiomyomas should be resected. Because both small esophageal cancer and leiomyoma do not usually cause symptoms, and because imaging studies alone do not provide a sound basis for differential diagnosis, enucleation or surgical biopsy may be performed even where esophageal leiomyomas are small, as in our cases.
In summary, esophageal leiomyoma, usually located in the middle or distal esophagus, appears on chest radiographs as a posterior mediastinal mass. Esophagography, in which the tumor is seen as an intramural, eccentric, smoothly elevated, sessile lesion, varying in size, is the most reliable form of diagnosis. Even where contrast medium is administered, CT reveals a lesion which demonstrates homogeneous low or iso attenuation, while T2-weighted MRI indicates the presence of one which is slightly hyperintense.
References
1. Seremetis MG, Lyons WS, Deguzman VC, Peabody JW. Leiomyomata of the esophagus. Cancer. 1976; 38:2166–2175. PMID: 991129.
2. Moersch HJ, Harrington SW. Benign tumor of the esophagus. Ann Otol. 1944; 53:800–817.
3. Barreiro F, Seco JL, Molina J, et al. Giant esophageal leiomyoma with secondary megaesophagus. Surgery. 1976; 79:436–439. PMID: 1257906.
4. Gallinger S, Steinhardt MI, Goldger M. Giant leiomyoma of the esophagus. Am J Gastroenterol. 1983; 78:708–711. PMID: 6637958.
5. Montesi A, Pesaresi A, Graziani L, Salmistraro D, Dini L, Bearzi I. Small benign tumors of the esophagus: radiological diagnosis with double-contrast examination. Gastrointest Radiol. 1983; 8:207–212. PMID: 6618085.

6. Megibow AJ, Balthazar EJ, Hulnick DH, Naidich DP, Bosniak MA. CT evaluation of gastrointestinal leiomyomas and leiomyosarcomas. AJR. 1985; 144:727–731. PMID: 3872029.

7. Massari M, Lattuada E, Zappa MA, et al. Evaluation of leiomyoma of the esophagus with endoscopic ultrasonography. Hepatogastroenterology. 1997; 44:727–731. PMID: 9222681.
8. Pelot D. Haurich WS, Schaffner F, Berk JE, editors. Anatomy, anomalies, and physiology of the esophagus. Bockus Gastroenterology. 1995. 5th ed. Philadelphia: Saunders;p. 397–411.
9. Evans HL. Smooth muscle tumors of the gastrointestinal tract. A study of 56 cases followed for a minimum 10 years. Cancer. 1985; 56:2242–2250. PMID: 4052969.
10. Lewis B, Maxfield RG. Leiomyoma of the esophagus: Case report and review of the literature. Int Abstr Surg. 1954; 99:105–128. PMID: 13187207.
12. Levine MS, Buck JL, Pantongrag-Brown L, Buetow PC, Lowry MA, Sobin LH. Esophageal leiomyomatosis. Radiology. 1996; 199:533–536. PMID: 8668807.

13. Rabushka LS, Fishman EK, Kulman JE, Hurban RH. Diffuse esophageal leiomyomatosis in a patient with Alport syndrome: CT demonstration. Radiology. 1991; 179:176–178. PMID: 2006273.

14. Casillas J, Joseph RC, Guerra JJ. CT appearance of uterine leiomyomas. RadioGraphics. 1990; 10:999–1007. PMID: 2259770.

15. Schnall MD. Gore RM, Levine MS, editors. Magnetic resonance imaging of the hollow viscera. Textbook of gastrointestinal radiology. 2000. 2nd ed. Philadelphia: Saunders;p. 86–97.
16. Totten RS, Stout AP, Humphreys GH, Moore RL. Benign tumors and cysts of the esophagus. J Thorac Surg. 1953; 25:606–622. PMID: 13062346.

17. Storey CF, Adams WC. Leiomyoma of the esophagus: report of four cases and a review of the surgical literature. Am J Surg. 1956; 91:3–23. PMID: 13268774.
18. Seremetis MG, Lyons WS, de Guzman VC, et al. Leiomyomata of the esophagus: an analysis of 838 cases. Cancer. 1976; 38:2166–2177. PMID: 991129.
19. Seremetis MG, de Guzman VC, Lyons WS, et al. Leiomyoma of the esophagus: a report of 19 surgical cases. Ann Thorac Surg. 1973; 16:308–316. PMID: 4728612.
Fig. 1
Esophageal leiomyoma in a 35-year-old man who had suffered substernal pain for two months.
A. Chest radiography reveals a right retrocardiac soft-tissue mass (arrows), with obliteration of the azygoesophageal recess interface.
B. Esophagography indicates the presence of a large, smoothly elevated filling defect in the distal esophagus. Also note the extraluminal component of the mass (arrows).
C. Enhanced CT scan (10-mm collimation) obtained at the ventricular level shows a homogeneous, iso-attenuated, pear-shaped mass, 75×45 mm in diameter, in the right azygoesophageal recess. Note the presence of gastrograffin-filled esophageal lumen (arrow).
D. Enhanced coronal T1-weighted MR image reveals a slightly hyperintense lesion in the azygoesophageal recess area.
E. T2-weighted MR image obtained at a level similar to that in C reveals a lesion slightly more hyperintense than chest wall muscle.
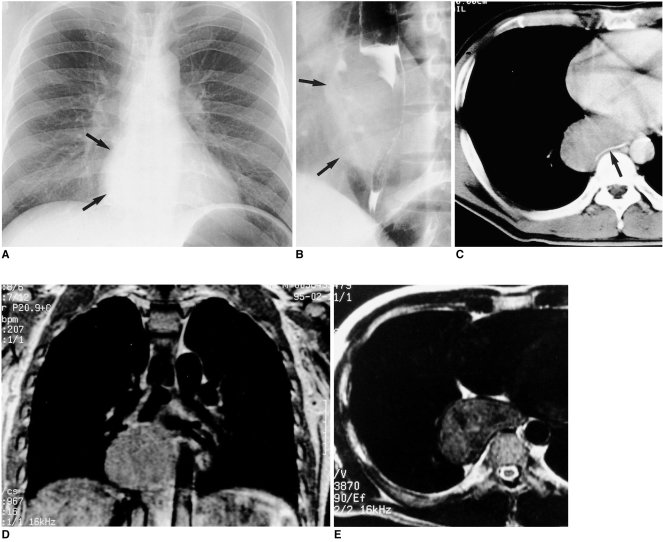
Fig. 2
Esophageal leiomyoma in a 34-year-old man who for one month had complained of abdominal fullness.
A. Esophagography demonstrates a crescentic filling defect in the distal esophagus due to the presence of a smoothly elevated mass lesion. Also note the extraluminal component of the mass (arrows).
B, C. Enhanced (10-mm collimation) CT scans obtained at the level of the liver dome reveal a circumferential, homogeneous, iso-attenuated mass measuring 60×45 mm in the distal esophagus.
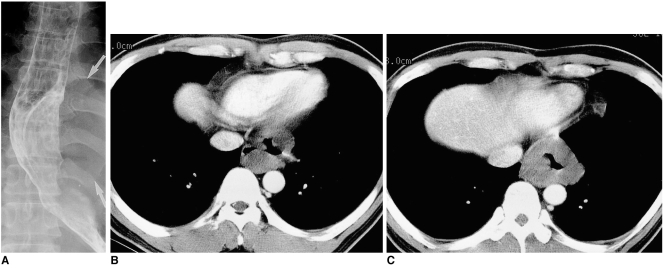
Fig. 3
Esophageal leiomyoma in a 37-year-old man with mild dysphagia.
A. Esophagography shows a soft tissue mass (arrows) surrounding the opacified esophageal lumen. Note that there is mild passage disturbance and mucosal rigidity in the distal esophagus.
B. Unenhanced (10-mm collimation) CT scan obtained at another hospital at the level of the liver dome reveals a soft tissue mass measuring 80×40 mm (arrows) surrounding the gas-filled esophageal lumen.
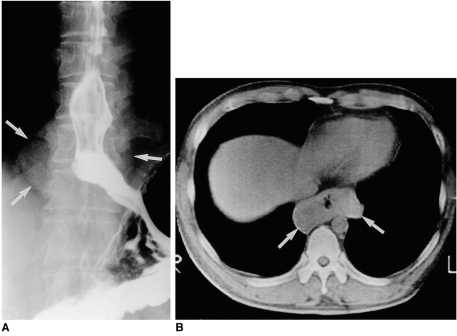
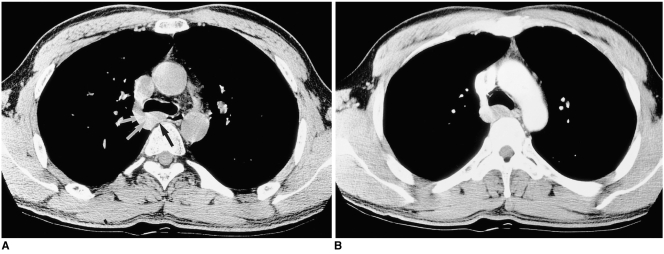
Fig. 4
Esophageal leiomyoma in a 47-year-old man with no subjective symptoms.
A. Thin-section (1-mm collimation) CT scan obtained at the level of the azygos arch shows a homogeneously iso-attenuated lesion (arrows) measuring 26×13 mm (arrows) between the azygos arch and the esophageal lumen, posterior to the carina.
B. Enhanced CT scan (7-mm collimation) demonstrates slight homogeneous enhancement.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download