Abstract
Objective
To describe the clinical and ultrasonographic (US) findings in patients with scleredema adultorum of Buschke, who presented with sclerotic skin on their posterior neck.
Materials and Methods
After obtaining IRB approval, eight patients with scleredema adultorum of Buschke were enrolled. They underwent US examination of their posterior neck. The diagnoses were confirmed pathologically. The clinical history and US images were evaluated retrospectively. Dermal thickness was compared between the patient group and the age- and sex-matched control group.
Results
The patients included seven males and one female with a mean age of 51.5 years. All patients presented with thickening of the skin and/or a palpable mass on the posterior neck. Five (62.5%) of the eight patients showed erythematous discoloration. Six patients (75.0%) had a history of diabetes. The Hemoglobin A1c level was found to be increased in all patients. US images did not show any evidence of a soft tissue mass or infection. The mean dermal thickness in patients (7.01 ± 1.95 mm) was significantly greater than that in the control group (3.08 ± 0.87 mm) (p = 0.001). Multiple strong echogenic spots in the dermis were seen in all patients. Seven patients (87.5%) showed posterior shadowing in the lower dermis.
Conclusion
When a patient with a history of diabetes presents with a palpable mass or erythematous discoloration of the posterior neck and US shows the following imaging features: 1) no evidence of a soft tissue mass or infection, 2) thickening of the dermis, 3) multiple strong echogenic spots and/or posterior shadowing in the dermis, scleredema adultorum of Buschke should be considered in the differential diagnosis.
When a patient presents with a palpable lesion on the posterior neck, ultrasonography (US) is a useful diagnostic tool for the evaluation of a soft tissue mass such as subcutaneous lipoma, epidermal inclusion cyst, chronic abscess, hematoma, etc. However, a lot of patients showed skin thickening without any soft tissue mass. Among them, patients with scleredema, also known as scleredema adultorum of Buschke, have the characteristic pattern of skin involvement and clinical features (12). It typically begins in the posterior neck skin (34). Hands and feet are characteristically spared. Raynaud's phenomenon or internal organ involvement is infrequent (3567). Scleredema may occur secondarily to diabetes or independently (2489).
Although dermatologists have paid attention to this disease entity, specialists at other departments or radiologists are not familiar with it. Furthermore, US findings have been rarely published in the English literature (10). There is a possibility of misdiagnosing scleredema, and eventually it can lead to improper treatment. The purpose of this study was to describe the clinical and US findings in patients with scleredema adultorum of Buschke, who presented with sclerotic skin on their posterior neck.
This study was approved by our Institutional Review Board. The requirement for obtaining informed written consent was waived for the patient study owing to the retrospective nature of the study, but it was obtained from all volunteers for the comparison study. After a computerized search of medical and US study records, we initially recruited 13 patients who had sclerotic skin thickening without any soft tissue mass on the posterior neck on US examination. For confirming the final diagnosis, medical records and histopathologic examinations were reviewed. Five patients were excluded. Two patients had an uncertain diagnosis. Three patients were diagnosed with scleredema adultorum of Buschke on a clinical basis, but histopathologic studies were not performed. Finally, we enrolled 8 patients (seven males and one female with a mean age of 51.5 years [range, 36–70 years]) with scleredema adultorum of Buschke who underwent US examination of their posterior neck and pathological examinations.
Ultrasonography examination was performed by an experienced musculoskeletal radiologist (with 15 years of experience) in the prone position owing to the anatomic location of the lesion (Fig. 1A). An ultrasound system (iU22; Philips, Seattle, WA, USA) with a 12-5 MHz high-frequency linear array was used.
Imaging findings were reviewed by two musculoskeletal radiologists, and an interpretation was achieved by consensus. Initially, the presence of a hidden soft tissue mass such as iso-echogenic lipoma was considered. Some patients presented with skin discoloration or redness. Therefore, we also evaluated the evidence of soft tissue infection such as subcutaneous fat edema, focal abscess or increased vascularity either on US and color Doppler imaging.
The thickness of the dermis, presence of multiple echogenic spots, and posterior shadowing were evaluated in the patient group. The thickness of the dermis was measured at the most thickened area. The distance from the epidermal reflection to subcutaneous fat was considered as the dermal thickness, because a very thin hypoechoic epidermis was not always clearly separated (Fig. 1B). The presence of multiple echogenic spots was considered if strong echogenic foci without posterior shadowing were detected in the dermis (Fig. 1C). When posterior structures were partially (Fig. 1B) or completely (Fig. 2A) invisible due to signal voids, the presence of posterior shadowing was considered. Five of the eight patients had erythematous skin swelling, and therefore, vascularity of the lesion was assessed with color Doppler imaging.
In a previous study by Cole et al. (10), US showed a difference in dermal thickness between the patient group and the control group. Also, the dermal thickness can change, depending on the difference in the anatomical site, age, and sex. Therefore, we enrolled eight age- and sex-matched normal volunteers (age range: 42–58 years, mean age: 48.4 years, seven males and one female) and compared the dermal thickness between the patient group and the control group. The control group did not have any history of diabetes and connective tissue disorders.
The mean values of the dermal thickness between the patient and control groups were compared using the Mann-Whitney U-test. A p value less than 0.05 was considered statistically significant. The statistical analysis was performed by using SPSS software (version 23.0; IBM Corp., Armonk, NY, USA).
All patients presented with thickening of the skin or a palpable mass on the posterior neck. Five (62.5%) of the eight patients showed erythematous discoloration.
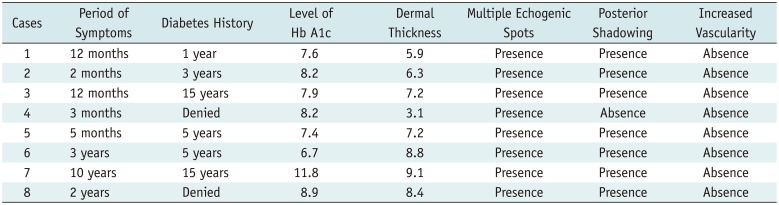
Other clinical findings of these eight patients are described in Table 1. The period of symptoms varied, ranging from 2 months to 10 years. Six (75.0%) of the eight patients had a history of diabetes; two patients denied any history of diabetes. Nevertheless, all patients showed increased Hb A1c level (6.7–11.8%).
Ultrasonography and color Doppler images did not show any evidence of hidden masses or soft tissue infection. Other US findings, including dermal thickness, presence of multiple echogenic spots, and posterior shadowing in the dermis, are shown in Table 1. The thickness of the dermis in patients ranged from 3.1 mm to 9.1 mm. In all patients, multiple echogenic spots in the dermis were observed. Seven (87.5%) of the eight patients showed posterior shadowing, especially in the lower dermis.
The mean dermal thickness in the patient group (7.01 ± 1.95 mm) was found to be significantly increased, compared to that in the control group (3.08 ± 0.87 mm) (p = 0.001). This result was similar to that in a previous study by Cole et al. (10).
Scleredema is a rare connective tissue disease of unknown etiology that belongs to a group of scleroderma-like disorders (1711). It is characterized by symmetric and non-pitting induration of the skin due to thickening of the dermis and deposition of collagen and mucin (34). It is typically begins on the posterior and lateral aspects of the neck, and then, it gradually extends to the upper back, shoulders and face.
Patients with sclerodema or other scleroderma-like disorders (pseudoscleroderma) also present with sclerosis of the skin (1211). Scleroderma, also called systemic sclerosis, is characterized by fibrosis of the skin and internal organs and vasculopathy. Raynaud's phenomenon is a common and cardinal manifestation (12). The list of scleroderma-like disorders includes localized (circumscribed) forms of scleroderma (morphea), stiff skin syndrome, eosinophilic fasciitis, scleredema adultorum of Buschke, diabetic scleredema, scleromyxedema, dialysis-associated scleromyxedema, pseudosclerodermas induced by various drugs, environmental and occupational factors, etc. (11). Differential diagnosis is based on clinical, laboratory, and histologic criteria defined by experts (2).
The pattern of cutaneous involvement can be useful for the differential diagnosis (12). For instance, scleroderma (systemic sclerosis) has a predilection for the hands and perioral region before skin thickening becomes more generalized and widespread, whereas localized forms of scleroderma (morphea) involve the trunk, extremities and the face (1113). However, scleroderma initially appears on the skin of the posterior neck. In this study, eleven of the thirteen patients with posterior neck skin thickening on US were diagnosed with scleredema clinically or pathologically. In eight patients, the diagnosis was confirmed by histopathologic examination.
Graft et al. have described three types of scleredema (167). Type 1 is the “classic type,” with preceding febrile illness. Most cases resolve completely over a period of several months to two years. Type 2 tends to follow a slow and progressive course, with an increased risk of developing para-proteinemia including multiple myeloma. Type 3 is a slow and progressive type that occurs in diabetic patients. Krakowski et al. (9) have described the clinical features of diabetic scleredema, which has been considered as Type 3 (8). Rho et al. (4) reported that diabetes was observed in about 50% of the documented cases of scleredema. However, nowadays, there is a tendency to distinguish diabetic scleredema from scleredema adultorum of Buschke (1415). In this study, in six of the eight patients, diagnoses were confirmed by histopathologic examinations, and they had a documented history of diabetes. The level of Hb A1c was found to be increased in all patients. Therefore, it appears that our cases of scleredema closely correlate with diabetes.
It was our impression that US findings might correlate with certain pathologic features. The pathologic features of scleredema include thickened dermis and deposition of mucin and collagen in the dermis. The epidermis is usually thin and unaffected (Fig. 2B, C). In our study, prominent dermal thickness was measured. The mean dermal thickness in the patient group was two-fold compared to that in the control group. Pathologic studies showed multifocal mucin depositions between collagen fibers and Alcian blue staining, especially in the deep dermis (Fig. 2D) (367814). We suspected that mucin deposition might be attributable to strong echogenic spots on US imaging (Figs. 1C, 2D). In the thickened dermis, collagen bundles were enlarged and swollen, and clear spaces between collagen bundles were observed due to split appearance. This was prominent in the lower dermis. On US study, seven of the eight patients (87.5%) showed posterior shadowing, especially in the lower dermis. We thought that these pathologic changes (enlarged and swollen collagen bundles and presence of clear spaces) could serve to produce posterior shadowing.
We did not determine whether these imaging findings (dermal thickening, echogenic spots in the dermis and posterior shadowing) were specific to scleredema. Other sclerotic dermal disorders (sclerodema or other scleroderma-like disorders) may show similar US findings. Also, when patients with sclerotic dermal disorders present with a palpable mass or induration, the primary purpose of the US study is to confirm or rule out the presence or absence of a subcutaneous mass or soft tissue infection. However, scleredema has a propensity to affect the skin of the posterior neck, and it frequently affects the individuals who suffer from diabetes. This clinical history may be useful in diagnosing scleredema with US.
In our study, five of the eight patients had erythematous skin swelling. The skin and subcutaneous fat infections such as dermatitis, panniculitis, cellulitis, and abscess should be excluded. US appearance of cellulitis ranges from diffuse swelling and increased echogenicity of the skin and subcutaneous fat to a variable cobblestone appearance depending on the amount of perifascial fluid (16). Cutaneous panniculitis-like T cell lymphoma can show a similar appearance to cellulitis (17). Cellulitis and cutaneous panniculitis-like T cell lymphoma can involve the skin and subcutaneous fat, but prominent appearance was observed in the subcutaneous fat. US can help differentiate sclerotic dermal disorders from these disorders.
In our study, dermal echogenicity of the lesion except for posterior shadowing was iso or slight hyperechogenic in comparison with the adjacent dermis. If posterior shadowing is not prominent, the lesion may not be clearly distinguished from the adjacent dermis (Fig. 1).
Numerous therapies have been tried with contradictory results including corticosteroid therapy, immunosuppressant therapy, bath PUVA, UVA-1 phototherapy, etc. (1
2). General clinicians or radiologists are not familiar with this disease entity. There is a possibility that scleredema might be misdiagnosed as cellulitis or subcutaneous lipoma. Therefore, familiarity with this disease entity and diagnosis by using US can potentially prevent inappropriate treatment.
This study has several limitations. First, the small sample size limited the evaluation of more proper US imaging features. Further comparison study between scleredema and other sclerotic skin disorders is needed. In addition, because this was a retrospective study, laboratory tests were not performed completely to rule out other collagen vascular diseases.
In conclusion, when a patient with a history of diabetes presents with a palpable mass or erythematous discoloration of the posterior neck and US shows the following imaging features: 1) no evidence of a focal mass or soft tissue infection, 2) thickening of the dermis, 3) multiple echogenic spots and/or posterior shadowing, scleredema should be considered in the differential diagnosis.
References
1. Foti R, Leonardi R, Rondinone R, Di Gangi M, Leonetti C, Canova M, et al. Scleredema-like disorders. Autoimmun Rev. 2008; 7:331–339. PMID: 18295739.
2. Fabri M, Hunzelmann N. [Differential diagnosis of scleredema and pseudoscleredema]. J Dtsch Dermatol Ges. 2007; 5:977–984. PMID: 17976138.
3. Meguerditchian C, Jacquet P, Béliard S, Benderitter T, Valéro R, Carsuzza F, et al. Scleredema adultorum of Buschke: an under recognized skin complication of diabetes. Diabetes Metab. 2006; 32(5 Pt 1):481–484. PMID: 17110904.

4. Rho YW, Suhr KB, Lee JH, Park JK. A clinical observation of scleredema adultorum and its relationship to diabetes. J Dermatol. 1998; 25:103–107. PMID: 9563277.

5. Ioannidou DI, Krasagakis K, Stefanidou MP, Karampekios S, Panayiotidis J, Tosca AD. Scleredema adultorum of Buschke presenting as periorbital edema: a diagnostic challenge. J Am Acad Dermatol. 2005; 52(2 Suppl 1):41–44. PMID: 15692512.

6. Venencie PY, Powell FC, Su WP, Perry HO. Scleredema: a review of thirty-three cases. J Am Acad Dermatol. 1984; 11:128–134. PMID: 6736348.

7. Beers WH, Ince A, Moore TL. Scleredema adultorum of Buschke: a case report and review of the literature. Semin Arthritis Rheum. 2006; 35:355–359. PMID: 16765712.

8. Toyota T, Umezu M, Oikawa N, Sanoyama R, Suzuki S, Suzuki H, et al. Diabetic scleredema. Tohoku J Exp Med. 1983; 141:457–461. PMID: 6670101.

9. Krakowski A, Covo J, Berlin C. Diabetic scleredema. Dermatologica. 1973; 146:193–198. PMID: 4717483.

10. Cole GW, Handler SJ, Burnett K. The ultrasonic evaluation of skin thickness in scleredema. J Clin Ultrasound. 1981; 9:501–503. PMID: 6796610.

11. Haustein UF. Scleredema and pseudo-scleredema: uncommon presentations. Clin Dermatol. 2005; 23:480–490. PMID: 16179182.
13. Gupta RA, Fiorentino D. Localized scleredema and systemic sclerosis: is there a connection? Best Pract Res Clin Rheumatol. 2007; 21:1025–1036. PMID: 18068859.
14. Kurihara Y, Kokuba H, Furue M. Case of diabetic scleredema: diagnostic value of magnetic resonance imaging. J Dermatol. 2011; 38:693–696. PMID: 21729146.

15. Sattar MA, Diab S, Sugathan TN, Sivanandasingham P, Fenech FF. Scleroedema diabeticorum: a minor but often unrecognized complication of diabetes mellitus. Diabet Med. 1988; 5:465–468. PMID: 2970922.

16. Chau CL, Griffith JF. Musculoskeletal infections: ultrasound appearances. Clin Radiol. 2005; 60:149–159. PMID: 15664569.

17. Kang BS, Choi SH, Cha HJ, Jung YK, Lee JH, Jeong AK, et al. Subcutaneous panniculitis-like T-cell lymphoma: US and CT findings in three patients. Skeletal Radiol. 2007; 36 Suppl 1:S67–S71. PMID: 16909278.

Fig. 1
44-year-old man suffering from skin thickening and palpable mass on posterior neck for 1 year.
A. Photograph showing swelling and erythematous change of skin. B. Extended-field-of-view scanning image showing thickening of dermis (double-headed arrow), compared to adjacent control side (black arrow). Partial posterior acoustic shadowing (white arrow) is noted. There is no focal mass or subcutaneous fat edema. C. Multiple echogenic spots (arrows) are observed in thickened dermis (doubleheaded arrow) on ultrasonographic image.
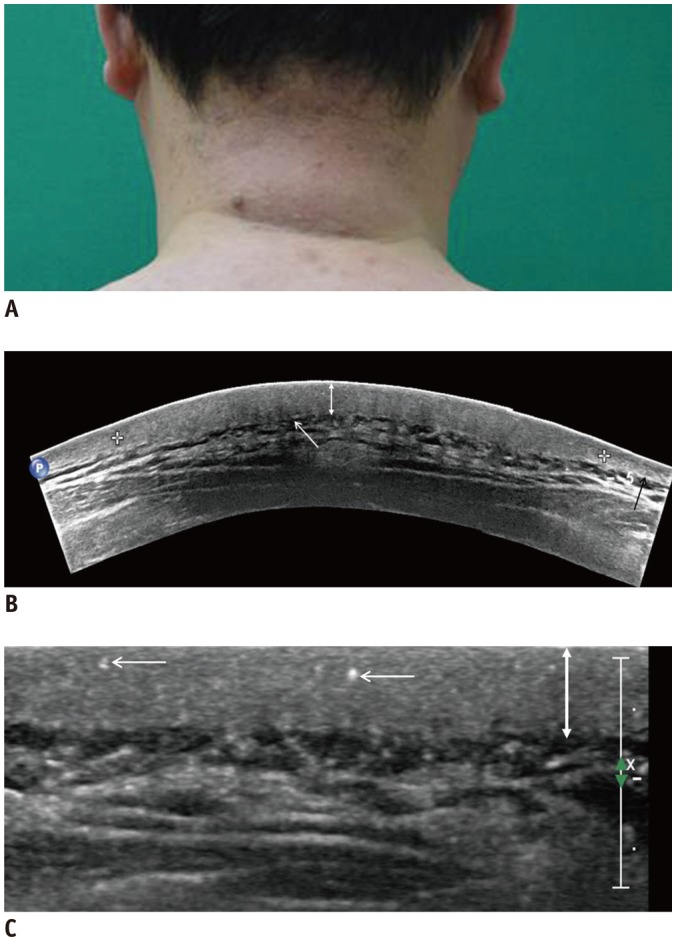
Fig. 2
46-year-old man suffering from diffuse nuchal skin thickening for 5 months.
He was diagnosed with diabetes 5 years ago. A. Ultrasonographic image showing thickened dermis and marked posterior acoustic shadowing in lower dermis area. There is no vascularity on color Doppler imaging. B. Hematoxylin-eosin stain image with original magnification (× 10) showing thin and unaffected epidermis (arrow) and thickened dermis (double-headed arrow). C. Higher magnification (Hematoxylin eosin stain, × 400) image of lower portion of dermis. Collagen fibers appear swollen and separated by wide spaces. D. Alcian blue stain (× 400) demonstrating multifocal mucin depositions (arrows) between swollen collagen fibers.
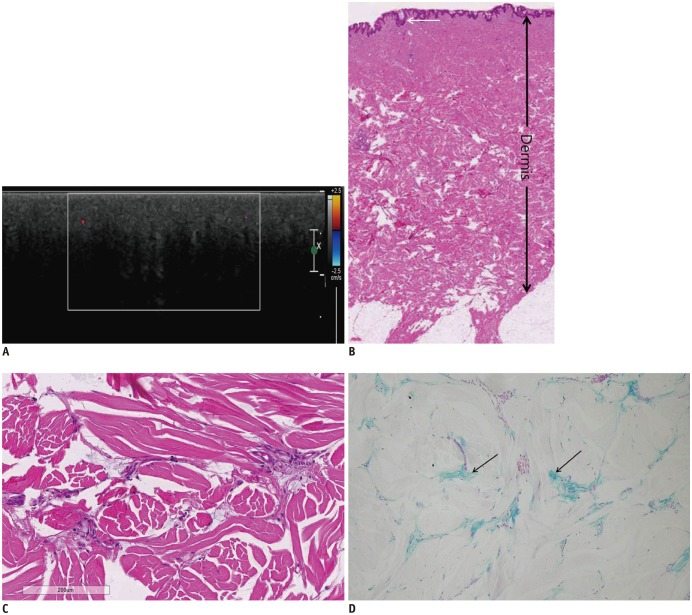
Table 1
Clinical and Ultrasonographic Findings of Patients with Scleredema




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download