Abstract
Vasculitis, characterized by inflammation of vessel walls, is comprised of heterogeneous clinicopathological entities, and thus poses a diagnostic challenge. The most widely used approach for classifying vasculitides is based on the International Chapel Hill Consensus Conference (CHCC) nomenclature system. Based on the recently revised CHCC 2012, we propose computed tomography (CT) features of vasculitides and a differential diagnosis based on location and morphological characteristics. Finally, vasculitis mimics should be differentiated, because erroneous application of immunosuppressive drugs on vasculitis mimics may be ineffective, even deteriorating. This article presents the utility of CT in the diagnosis and differential diagnosis of vasculitides.
Vasculitis is defined as inflammation of vessel walls. Diagnosis of vasculitis can be challenging because signs and symptoms of vasculitis are nonspecific and could mimic other conditions such as infection, malignancy, thrombotic disorders, and connective tissue diseases (12). Clinical symptoms reflect the affected vessels. Limb claudication, absent pulses, and unequal blood pressure are typical symptoms of large vessel vasculitis, while palpable purpura and proteinuria are typical symptoms of small vessel vasculitis. Medium vessels are defined as main visceral arteries and their initial branches, therefore various symptoms occur according to the involved vessels.
For the evaluation of vasculitis, detailed clinical history, physical examination and focused laboratory investigation are crucial. In addition, non-invasive imaging is helpful for evaluating the extent of the disease. Among them, computed tomography (CT) is commonly used as the initial imaging study for vasculitis, because it can allow evaluation of vascular wall change and localize the location and extent of the lesion with excellent spatial resolution (3). In this article, we describe the diagnosis and classification of vasculitis with CT findings on the basis of the revised International Chapel Hill Consensus Conference (CHCC) 2012.
Chapel Hill Consensus Conference is a nomenclature system (nosology) that does not specify the criteria by which patients are classified for clinical studies or are diagnosed for clinical care. The CHCC 1994 offered names and definitions for the most common forms of vasculitis (45), and categorized vasculitis by the size of the blood vessels affected (6). With advances in the understanding of the pathophysiology of vasculitis, the CHCC 2012 added four new categories: variable vessel vasculitis, single-organ vasculitis, vasculitis associated with systemic disease, and vasculitis associated with probable etiology (Table 1).
A non-invasive imaging approach is helpful for detecting and evaluating the extent of vasculitis. Among them, multidetector CT is a good imaging modality for assessing vasculitis, due to its excellent spatial resolution and accurate cross-sectional imaging in demonstrating vessel wall change. Table 2 summarizes the advantages and disadvantages of various non-invasive imaging modalities for assessment of vasculitis, including magnetic resonance imaging, positron emission tomography and ultrasonography (7).
In CHCC 2012 nomenclature, the first approach to identifying vasculitis is evaluation of the size of affected vessels (i.e., large, medium, and small vessel vasculitis) (4). If all sizes of vessels are present or additional veins are affected, variable vessel vasculitis, a new category in the CHCC 2012, can be suspected. Figure 1 shows a diagram of vasculitis, based on the affected vessel size and types.
By the CHCC 2012 definition, “large vessels” are the aorta and its major branches, except for the most distal branches.
Takayasu arteritis, which is also known as “aortic arch syndrome” and “pulseless disease”, is an idiopathic inflammatory disease that primarily affects large vessels such as the aorta, major branches of the aorta, and coronary and pulmonary arteries. Pathologically, TA is characterized by panarteritis affecting all three arterial layers (89), and subsequent scarring of media leading to luminal occlusion (10).
Takayasu arteritis occurs worldwide, but is much more common in Asia (1112). Women are predominantly affected compared to men, with a 7:1 ratio (13). The American College of Rheumatology states that TA can be suspected when patients have at least three of the following criteria; age < 40 years at disease onset, claudication of the extremities, decreased brachial artery pressure, blood pressure difference of more than 10 mm Hg in between the arms, bruit over the subclavian arteries or aorta, and abnormal arteriographic results (14).
Aortic involvement is common in TA; thoracic aorta and its branches are commonly affected, with a 65% prevalence (15). Pulmonary arteries and coronary arteries could also be affected, with prevalences of 63.3 and 44.4%, respectively (16).
CT findings of TA differ depending on the stage (1217). In the early phase (active stage), concentric mural thickening of the involved arteries is characteristic (18). On pre-contrast CT scanning, the mural thickening is of high attenuation compared with the lumen, while on the post-enhanced CT angiographic images, it exhibits a “double ring sign”. Double ring sign is typically shown in the venous phase, as an inner low attenuating wall surrounded by an outer high attenuating wall (Fig. 2) (1920). In the late phase (occlusive stage), arterial stenosis, occlusion, or aneurysmal dilatation may occur, associated with the mural thickening (21). Extensive vascular calcification and collateral vessels may be observed with luminal narrowing (Fig. 3).
Giant cell arteritis (GCA) is another form of large vessel vasculitis affecting the aorta and its large arterial branches. Characteristically, it commonly affects the branches of carotid arteries, vertebral arteries, and temporal arteries, leading to characteristic temporal headaches (10). A microscopic exam reveals vasculitis with mononuclear infiltrates or granulomas in the active stage and vessel wall fibrosis in the chronic stage (122).
Clinical differentiation between GCA and TA is sometimes difficult, but GCA predominantly affects female patients who are older than 50 years old, and is prevalent in the western countries, in contrast to TA. Pathologically, TA is commonly associated with extensive intimal and adventitial fibrosis with resultant luminal narrowing, whereas GCA is more commonly associated with extensive medial inflammation, necrosis, and formation of aortic aneurysm. “Skip lesion”, or aortic inflammation, is also more common in GCA.
Medium vessel vasculitis predominantly affects main visceral arteries and their initial branches (4).
Polyarteritis nodosa (PAN) is a necrotizing vasculitis of medium and small arteries without glomerulonephritis, and is unassociated with the anti-neutrophil cytoplasmic antibody (ANCA) (4). Absence of ANCA is a distinguishing point from microscopic polyangiitis (MPA), which is an ANCA-associated small vessel vasculitis that shares similar pathologic and radiologic findings with PAN.
Polyarteritis nodosa affects men twice as often as women in the 5–7th decades (23). Etiology is unclear, but the hepatitis B virus may play an important role in the development of the disease (24). Renal involvement is common with an incidence of 70–80%, resulting in proteinuria, hematuria, and hypertension, but not glomerulonephritis. Other involvements are the gastrointestinal tract and skin at an incidence rate of 50%; skeletal muscles and mesentery at an incidence rate of 30%; and the central nervous system at an incidence rate of 10% (23).
Kawasaki disease is a medium vessel vasculitis associated with mucocutaneous lymph node syndrome, and it frequently occurs in infants and children younger than 5 years. Although exact pathogenesis is unknown, it seems likely that infection or autoimmunity play a critical role (25). Approximately 20.8 in 100000 children under 5 years (0.02%) are affected, with the peak onset age in between 2 and 3 years (7). The incidence is especially high in Asian countries.
Coronary artery involvement is common in patients with KD (26), therefore myocardial ischemia or infarction frequently occur. CT findings include coronary artery aneurysm, ectasia, premature atherosclerosis and stenosis (25). A typical braid-like appearance of coronary arteries is one of the characteristics of KD (Fig. 5) (27). The size of the involved coronary arteries is an important prognostic factor of KD. Myocarditis, pericarditis with pericardial effusion, regional wall motion abnormality, valvulitis, and papillary muscle dysfunction are other cardiac manifestations.
Small vessel vasculitides mainly affect small intraparenchymal arteries, arterioles, capillaries, and venules (4). They are divided into two categories; ANCA-associated small vessel vasculitis and immune complex-associated small vessel vasculitis.
Anti-neutrophil cytoplasmic antibody is an antibody against intracellular antigens in neutrophils and monocytes. According to immunofluorescent staining pattern, there are cytoplasmic ANCA (c-ANCA) and perinuclear ANCA (p-ANCA) (2). Granulomatosis with polyangiitis (GPA) is a c-ANCA (+) vasculitis, whereas eosinophilic GPA (EGPA) or MPA are characteristic of p-ANCA (+) vasculitis (28). Although ANCA is an important characteristic of ANCA-associated vasculitis, its positive predictive value is only around 45% (28). This means that ANCA negativity does not exclude the possibility of ANCA-associated vasculitis (2).
Granulomatosis with polyangiitis is the replacement term for formerly known Wegener granulomatosis, because it appropriately reflects its classic histopathologic criteria including three major features: granulomatous inflammation involving the upper and lower respiratory tract, necrotizing vasculitis involving both arteries and veins, and glomerulonephritis.
The upper respiratory tract is affected in almost all patients with GPA, and lungs (90%) and kidneys (80%) are also frequently involved (229). Although clinical manifestations of GPA are usually limited to the upper and lower respiratory tract, presenting as oral or nasal inflammation, cough and hemoptysis (30), GPA can manifest with various symptoms according to the affected organs.
In GPA, pulmonary nodules or masses are the most common findings, which are usually multiple and bilateral (Fig. 6) (31). Central cavitation may be noted in up to 50% of nodules larger than 2 cm in diameter. Areas of consolidation or ground-glass opacity (GGO) are also noted in 30% of patients with active GPA, which may be the result of alveolar hemorrhage or mosaic perfusion secondary to small vessel vasculitis (32). When the tracheobronchial tree is involved, wall thickening of the airway is usually circumferential, smooth or nodular (29). Rarely, it can involve the cardiovascular system, presenting as pericarditis, coronary arteritis or valvular dysfunction (29).
Eosinophilic GPA is the disease that has been called Churg-Strauss syndrome. EGPA is diagnosed when there is asthma, increased blood eosinophils (> 1.5 G/L or > 10% leukocytes) and necrotizing vasculitis affecting small to medium vessels.
Clinical manifestations of EGPA can vary depending on its ANCA status, although almost all patients have a history of asthma, regardless of the ANCA status (33). ANCA-positive patients show a significantly higher frequency of ear, nose, and throat symptoms, peripheral neuropathy or renal manifestations, compared to ANCA-negative patients. On the other hand, ANCA-negative patients show cardiac manifestations more frequently.
Lungs are the most frequently involved organ in EGPA. CT typically shows subpleural GGO or consolidation of lobular distribution, centrilobular nodules, bronchial wall thickening, and interlobular septal thickening (34). Cardiac involvement is the leading cause of mortality and manifests as myocarditis (Fig. 7) or Loeffler's myocarditis, sometimes progressing to a fibrotic stage with thrombosis (3536). Coronary arteries or pericardium can also be involved, presenting as coronary arteritis, pericardial effusion or pericarditis.
Variable vessel vasculitis does not have any predominant type of vessel involvement, but instead, affects vessels of any size (small, medium, and large) and type (arteries, veins, and capillaries).
Behçet's disease is a chronic, relapsing autoinflammatory condition involving multiple organs. Clinically, Behçet's disease manifests as recurrent oral or genital aphthous ulcers, and inflammatory lesions of the cutaneous, ocular, articular, gastrointestinal, and central nervous system. It is most prevalent in the 3rd and 4th decades of life, and is more common in males.
Vascular involvement occurs in 5–30% of Behçet's disease cases, and it can be divided into three types: venous thrombosis, arterial aneurysm and aortic occlusion (37). Venous thrombosis is associated with a prevalence range of 10–30% (38). Deep vein thrombosis of the lower extremities is the most common finding, followed by superficial thrombophlebitis after venipuncture. Aneurysms, which are frequently seen in the aorta, pulmonary, subclavian, and popliteal arteries, can be the most serious complication of Behçet's disease. Saccular types are more frequent than fusiform aneurysms. In case of pulmonary involvement, pulmonary artery aneurysm is the most common finding with a prevalence of 1–10%, and tends to be multiple and bilateral (Fig. 8). Hughes-Stovin syndrome, which may be a variant of Behçet's disease, is also characterized by multiple pulmonary artery aneurysms and deep vein thrombosis, but it can be distinguished from Behçet's disease by the absence of mucocutaneous findings (39). Pseudoaneurysms of the sinus of Valsalva or aortic root can recur, even after an aortic root surgery (Fig. 9). Arterial occlusion or stenosis and mural enhancement, although less frequent compared to aneurysms, can also be recognized in any sized arteries. Incidence of cardiac involvement has been reported to be ranging from 1 to 5%. Main cardiac features include pericarditis, myocarditis, cardiomyopathy, valvular disease, coronary artery stenosis, aneurysm or right-side-predominant intracardiac thrombus (40).
Cogan syndrome is a rare inflammatory disorder characterized by ocular inflammatory lesions (interstitial keratitis, uveitis, and episcleritis) and audiovestibular dysfunctions (sensorineural hearing loss, vertigo, nausea, and tinnitus). In addition, various systemic manifestations of the cardiovascular, musculoskeletal, neurological, and gastrointestinal systems have been associated. Characteristic cardiovascular involvement in Cogan syndrome manifests as aortitis and aortic insufficiency which are similar to that of TA, occurring in about 10% of the patients (Fig. 10) (41).
The presence of known etiology or associated systemic disease can be helpful for the differential diagnosis of vasculitis. Systemic lupus erythematosus or rheumatoid arthritis are systemic inflammatory disorders, which may lead to inflammation of the cardiovascular system. IgG4-related sclerosing disease is characterized by idiopathic inflammatory lesions infiltrated with numerous IgG4-positive plasma cells (42). IgG4-related periarteritis predominantly involves the abdominal aorta, iliac vessels and thoracic aorta. CT findings include arterial wall thickening, homogeneous enhancement at the delayed phase of contrast enhanced CT, luminal change (mostly dilation and rarely stenosis), and exaggerated atherosclerotic change (42). An elevated serum IgG4 level and associated organ damage are essential in discerning this disease from other vasculitides (Fig. 11). A few cases of IgG4-related periarteritis affecting the coronary arteries have been reported, which appeared as soft tissue encasing the coronary artery with or without luminal narrowing, wall calcification, and aneurysm (Fig. 12) (27).
Several conditions without vascular inflammation could mimic vasculitis, and these should be differentiated, because immunosuppressive drugs may be ineffective or even deteriorating for the vasculitis mimics (Table 3) (434445). Fibromuscular dysplasia (FMD) mimics large or medium vessel vasculitis, showing similar imaging findings of PAN. At CT, FMD appears as an arterial narrowing, multifocal stenosis (string of beads appearance), aneurysm, or dissection (Fig. 13). Segmental arterial mediolysis is a rare non-atherosclerotic, non-hereditary vasculopathy with absence of an inflammatory component. It may mimic medium vessel vasculitis such as PAN. CT findings include a string of beads appearance of visceral arteries, aneurysms of small abdominal arteries, arterial wall thickening, and dissection (Fig. 14). Neurofibromatosis type I is also an autosomal dominant disorder that may affect large or medium vessels. The most commonly involved vessels are renal, cerebral and pulmonary arteries. CT shows various findings including aneurysm and stenosis.
Multidetector CT is a useful noninvasive imaging modality for the evaluation of vasculitis and vasculitis mimics because CT can provide the information of the vessel wall change and other accompanied findings. Although CT features of various vasculitis are often overlapping, CT features via consideration of the involved vessel type, location, morphology, and associated systemic disease, can be useful in narrowing down the differential diagnosis (Table 3, Fig. 15). Getting familiar with CT features will help radiologists to establish appropriate diagnosis for vasculitis.
Figures and Tables
Fig. 1
Diagram of vasculitides categorized based on frequently affected vessel size and types.
Drawing shows aorta, large artery, medium arteries, and small arteries or arterioles, capillaries, venules, and veins, in sequence from left to right. Modified from Jennette et al. Arthritis Rheum 2013;65:1-11 (4). ANCA = anti-neutrophil cytoplasmic antibody, Anti-GBM = anti-glomerular basement membrane, IgA = immunoglobulin A
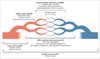
Fig. 2
Early phase Takayasu arteritis in 55-year-old woman with bilateral arm pain.
Axial CT scans obtained during delayed phase show typical concentric wall thickening with double rings (inner low and outer high enhancement) at ascending and descending aorta and ostia of both subclavian arteries (arrows).
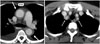
Fig. 3
Late phase Takayasu arteritis in 71-year-old woman with dizziness.
A. Volume rendered CT image shows massive patchy calcifications at aorta, indicating long-standing aortitis. B. Axial CT scan shows concentric wall calcification at ascending aorta (arrows). C. At level of arch vessels, total occlusion at left subclavian artery (arrows) was noted.

Fig. 4
Polyarteritis nodosa in 44-year-old man with hypertension.
Curved MPR image clearly demonstrates right renal artery aneurysm (arrow). Focal stenosis (arrowheads) at ostium of right renal artery is noted. MPR = multiplanar reformatted
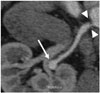
Fig. 5
Kawasaki disease in 18-year-old woman with chest pain.
A. Curved MPR image shows multifocal aneurysms (arrows) with thrombi (arrowheads) at left circumflex coronary artery. B. Invasive angiogram reveals aneurysms (arrows) with stenosis (arrowhead) at left circumflex coronary artery.
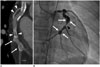
Fig. 6
Granulomatosis with polyangiitis in 58-year-old man with epistaxis.
A. CT scan demonstrates multiple non-cavitary nodules (arrows) in bilateral lungs, which are mainly distributed along bronchovascular bundles. B. PET scan shows multiple hypermetabolic pulmonary nodules (arrows; maximum SUV = 10.0). C. Bronchoscopy shows multiple whitish lesions at bilateral main bronchi. Biopsy reveals necrot

Fig. 7
Eosinophilic granulomatosis with polyangiitis in 53-year-old man with known asthma, generalized petechiae, and cardiac enzyme elevation.
A. CT scan shows uneven distribution of left ventricular (LV) hypertrophy (double-headed arrows) at midventricular LV wall. B. Delayed enhanced MR image shows multifocal patchy enhancement (arrows) at subendocardial and mid-layer of midventricular LV wall. C. Chest CT scan shows bronchovascular bundle thickening, and interlobular septal thickening with some peribronchial and subpleural ground-glass opacities in bilateral lungs.
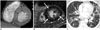
Fig. 8
Behçet's disease in 47-year-old man with hemoptysis.
A. Initial chest CT shows large pulmonary artery pseudoaneurysm (arrow) at right lower lobe, which is not resolved with pulmonary artery embolization. B. Follow up CT 6 months after initial scan shows another pulmonary artery pseudoaneurysm (arrows) at left lower lobe.
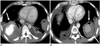
Fig. 9
Behçet's disease in 49-year-old man with history of aortic valve replacement.
Two months after aortic valve replacement, CT image shows paravalvular pseudoaneurysm (arrows) around aortic root.
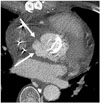
Fig. 10
Cogan syndrome in 47-year-old female with interstitial keratitis and sensory neural hearing loss.
A. Volume-rendered image shows tight stenosis (arrows) at ostia of right brachiocephalic, left common carotid, and left subclavian arteries. B. Sagittal reformatted CT image shows luminal irregularity (arrowheads) of abdominal aorta and tight stenosis (arrows) at ostia of celiac axis and superior mesenteric artery.
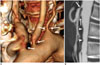
Fig. 11
IgG4-related sclerosing disease in 70-year-old man, with incidentally found abnormality of pancreas on routine ultrasound exam.
A. CT shows enlarged pancreas tail and peripancreatic low attenuation (arrowheads) indicative of autoimmune pancreatitis. Homogeneous enhancement of aorta (arrows) is also noted. B. Circumferential wall thickening with homogeneous enhancement around infrarenal abdominal aorta (arrows) are noted. C. PET scan demonstrates hypermetabolism around abdominal aorta (arrows).

Fig. 12
IgG4-related periarteritis in 67-year-old man with chest pain.
A. Coronary curved MPR image shows well-defined soft tissue encasement (arrows) along left anterior descending artery. B. Conventional angiography shows limitation in identifying coronary abnormalities in absence of any luminal abnormalities.

Fig. 13
Fibromuscular dysplasia in 20-year-old man with poorly controlled high blood pressure.
A. CT shows segmental narrowing (arrow) and luminal irregularity of right renal artery. B. Angiogram of right renal artery shows focal narrowing of right renal artery, with classic string of beads appearance (arrow).
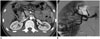
Fig. 14
Segmental arterial mediolysis in 40-year-old woman with abdominal pain
Serial axial CT scans show focal dissection (arrowhead) at ostium of celiac axis and diffuse severe stenosis with extensive concentric wall thickening and beaded appearance at common and proper hepatic arteries (arrows).
Fig. 15
Diagram of key findings and sites of predilection for various vasculitides
Section of vasculitis involving pulmonary artery is modified from Chung et al. Radiology 2010;255:322-341 (39). *Vasculitis mimics. SMA = superior mesenteric artery

Table 1
Various Vasculitides Based on 2012 International Chapel Hill Consensus Conference

Modified from Jennette et al. Arthritis Rheum 2013;65:1-11 (4). IgA = immunoglobulin A
Table 2
Advantages and Disadvantages of Each Imaging Modality for Evaluation of Vasculitis

Table 3
Clinical and Radiologic Characteristic Findings of Vasculitis Mimics

Notes
References
1. Restrepo CS, Ocazionez D, Suri R, Vargas D. Aortitis: imaging spectrum of the infectious and inflammatory conditions of the aorta. Radiographics. 2011; 31:435–451.
2. Castañer E, Alguersuari A, Gallardo X, Andreu M, Pallardó Y, Mata JM, et al. When to suspect pulmonary vasculitis: radiologic and clinical clues. Radiographics. 2010; 30:33–53.
3. Kim YJ, Yong HS, Kim SM, Kim JA, Yang DH, Hong YJ, et al. Korean guidelines for the appropriate use of cardiac CT. Korean J Radiol. 2015; 16:251–285.
4. Jennette JC, Falk RJ, Bacon PA, Basu N, Cid MC, Ferrario F, et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013; 65:1–11.
5. Jennette JC. Overview of the 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Clin Exp Nephrol. 2013; 17:603–606.
6. Kallenberg CG. Key advances in the clinical approach to ANCA-associated vasculitis. Nat Rev Rheumatol. 2014; 10:484–493.
7. Khanna G, Sargar K, Baszis KW. Pediatric vasculitis: recognizing multisystemic manifestations at body imaging. Radiographics. 2015; 35:849–865.
8. de Souza AW, de Carvalho JF. Diagnostic and classification criteria of Takayasu arteritis. J Autoimmun. 2014; 48-49:79–83.
9. Kang EJ, Kim SM, Choe YH, Lee GY, Lee KN, Kim DK. Takayasu arteritis: assessment of coronary arterial abnormalities with 128-section dual-source CT angiography of the coronary arteries and aorta. Radiology. 2014; 270:74–81.
10. Gornik HL, Creager MA. Aortitis. Circulation. 2008; 117:3039–3051.
11. Koide K. Takayasu arteritis in Japan. Heart Vessels Suppl. 1992; 7:48–54.
12. Park YB, Hong SK, Choi KJ, Sohn DW, Oh BH, Lee MM, et al. Takayasu arteritis in Korea: clinical and angiographic features. Heart Vessels Suppl. 1992; 7:55–59.
13. Lim AY, Lee GY, Jang SY, Gwag HB, Choi SH, Jeon ES, et al. Gender differences in clinical and angiographic findings of patients with Takayasu arteritis. Clin Exp Rheumatol. 2015; 33:2 Suppl 89. S-132–S-137.
14. Wolfe F, Smythe HA, Yunus MB, Bennett RM, Bombardier C, Goldenberg DL, et al. The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis Rheum. 1990; 33:160–172.
15. Hata A, Noda M, Moriwaki R, Numano F. Angiographic findings of Takayasu arteritis: new classification. Int J Cardiol. 1996; 54:S155–S163.
16. Zhu FP, Luo S, Wang ZJ, Jin ZY, Zhang LJ, Lu GM. Takayasu arteritis: imaging spectrum at multidetector CT angiography. Br J Radiol. 2012; 85:e1282–e1292.
17. Gotway MB, Araoz PA, Macedo TA, Stanson AW, Higgins CB, Ring EJ, et al. Imaging findings in Takayasu’s arteritis. AJR Am J Roentgenol. 2005; 184:1945–1950.
18. Mason JC. Takayasu arteritis--advances in diagnosis and management. Nat Rev Rheumatol. 2010; 6:406–415.
19. Park JH, Chung JW, Im JG, Kim SK, Park YB, Han MC. Takayasu arteritis: evaluation of mural changes in the aorta and pulmonary artery with CT angiography. Radiology. 1995; 196:89–93.
20. Khandelwal N, Kalra N, Garg MK, Kang M, Lal A, Jain S, et al. Multidetector CT angiography in Takayasu arteritis. Eur J Radiol. 2011; 77:369–374.
21. Yamato M, Lecky JW, Hiramatsu K, Kohda E. Takayasu arteritis: radiographic and angiographic findings in 59 patients. Radiology. 1986; 161:329–334.
22. Calvo-Romero JM. Giant cell arteritis. Postgrad Med J. 2003; 79:511–515.
23. Stanson AW, Friese JL, Johnson CM, McKusick MA, Breen JF, Sabater EA, et al. Polyarteritis nodosa: spectrum of angiographic findings. Radiographics. 2001; 21:151–159.
24. Guillevin L, Lhote F, Cohen P, Sauvaget F, Jarrousse B, Lortholary O, et al. Polyarteritis nodosa related to hepatitis B virus. A prospective study with long-term observation of 41 patients. Medicine (Baltimore). 1995; 74:238–253.
25. Díaz-Zamudio M, Bacilio-Pérez U, Herrera-Zarza MC, Meave-González A, Alexanderson-Rosas E, Zambrana-Balta GF, et al. Coronary artery aneurysms and ectasia: role of coronary CT angiography. Radiographics. 2009; 29:1939–1954.
26. Chung CJ, Stein L. Kawasaki disease: a review. Radiology. 1998; 208:25–33.
27. Jeon CH, Kim YK, Chun EJ, Kim JA, Yong HS, Doo KW, et al. Coronary artery vasculitis: assessment with cardiac multi-detector computed tomography. Int J Cardiovasc Imaging. 2015; 31:Suppl 1. 59–67.
28. Suresh E. Diagnostic approach to patients with suspected vasculitis. Postgrad Med J. 2006; 82:483–488.
29. Martinez F, Chung JH, Digumarthy SR, Kanne JP, Abbott GF, Shepard JA, et al. Common and uncommon manifestations of Wegener granulomatosis at chest CT: radiologic-pathologic correlation. Radiographics. 2012; 32:51–69.
30. Almouhawis HA, Leao JC, Fedele S, Porter SR. Wegener’s granulomatosis: a review of clinical features and an update in diagnosis and treatment. J Oral Pathol Med. 2013; 42:507–516.
31. Lohrmann C, Uhl M, Kotter E, Burger D, Ghanem N, Langer M. Pulmonary manifestations of Wegener granulomatosis: CT findings in 57 patients and a review of the literature. Eur J Radiol. 2005; 53:471–477.
32. Ananthakrishnan L, Sharma N, Kanne JP. Wegener’s granulomatosis in the chest: high-resolution CT findings. AJR Am J Roentgenol. 2009; 192:676–682.
33. Comarmond C, Pagnoux C, Khellaf M, Cordier JF, Hamidou M, Viallard JF, et al. Eosinophilic granulomatosis with polyangiitis (Churg-Strauss): clinical characteristics and long-term followup of the 383 patients enrolled in the French Vasculitis Study Group cohort. Arthritis Rheum. 2013; 65:270–281.
34. Worthy SA, Müller NL, Hansell DM, Flower CD. Churg-Strauss syndrome: the spectrum of pulmonary CT findings in 17 patients. AJR Am J Roentgenol. 1998; 170:297–300.
35. Knockaert DC. Cardiac involvement in systemic inflammatory diseases. Eur Heart J. 2007; 28:1797–1804.
36. Yune S, Choi DC, Lee BJ, Lee JY, Jeon ES, Kim SM, et al. Detecting cardiac involvement with magnetic resonance in patients with active eosinophilic granulomatosis with polyangiitis. Int J Cardiovasc Imaging. 2016; 32:Suppl 1. 155–162.
37. Chae EJ, Do KH, Seo JB, Park SH, Kang JW, Jang YM, et al. Radiologic and clinical findings of BehcXMLLink_XYZet disease: comprehensive review of multisystemic involvement. Radiographics. 2008; 28:e31.
38. Owlia MB, Mehrpoor G. Behcet’s disease: new concepts in cardiovascular involvements and future direction for treatment. ISRN Pharmacol. 2012; 2012:760484.
39. Chung MP, Yi CA, Lee HY, Han J, Lee KS. Imaging of pulmonary vasculitis. Radiology. 2010; 255:322–341.
40. Marzban M, Mandegar MH, Karimi A, Abbasi K, Movahedi N, Navabi MA, et al. Cardiac and great vessel involvement in “Behcet’s disease”. J Card Surg. 2008; 23:765–768.
41. Grasland A, Pouchot J, Hachulla E, Blétry O, Papo T, Vinceneux P. Study Group for Cogan's Syndrome. Typical and atypical Cogan’s syndrome: 32 cases and review of the literature. Rheumatology (Oxford). 2004; 43:1007–1015.
42. Inoue D, Zen Y, Abo H, Gabata T, Demachi H, Yoshikawa J, et al. Immunoglobulin G4-related periaortitis and periarteritis: CT findings in 17 patients. Radiology. 2011; 261:625–633.
43. Molloy ES, Langford CA. Vasculitis mimics. Curr Opin Rheumatol. 2008; 20:29–34.
44. Filippone EJ, Foy A, Galanis T, Pokuah M, Newman E, Lallas CD, et al. Segmental arterial mediolysis: report of 2 cases and review of the literature. Am J Kidney Dis. 2011; 58:981–987.
45. Baker-LePain JC, Stone DH, Mattis AN, Nakamura MC, Fye KH. Clinical diagnosis of segmental arterial mediolysis: differentiation from vasculitis and other mimics. Arthritis Care Res (Hoboken). 2010; 62:1655–1160.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download