Abstract
Objective
To determine whether the appearance of a spiculated mass on a mammogram is associated with luminal A subtype breast cancer and the factors that may influence the presence or absence of the spiculated mass.
Materials and Methods
Three hundred seventeen (317) patients who underwent image-guided or surgical biopsy between December 2014 and April 2015 were included in the study. Radiologists conducted retrospective assessments of the presence of spiculated masses according to the criteria of Breast Imaging Reporting and Data System. We used combinations of estrogen receptor (ER), progesterone receptor (PR), human epithelial growth factor receptor 2 (HER2), and Ki67 as surrogate markers to identify molecular subtypes of breast cancer. Pearson chi-square test was employed to measure statistical significance of correlations. Furthermore, we built a bi-variate logistic regression model to quantify the relative contribution of the factors that may influence the presence or absence of the spiculated mass.
Results
Seventy-one percent (71%) of the spiculated masses were classified as luminal A. Masses classified as luminal A were 10.3 times more likely to be presented as spiculated mass on a mammogram than all other subtypes. Patients with low Ki67 index (< 14%) and HER2 negative were most likely to present with a spiculated mass on their mammograms (p <0.001) than others. The hormone receptor status (ER and PR), pathology grade, overall breast composition, were all associated with the presence of a spiculated mass, but with less weight in contribution than Ki67 and HER2.
Invasive breast cancer is a molecularly heterogeneous disease that appears to include at least four major tumor subtypes (12). But for economic reasons, formal gene expression analysis is replaced by clinicopathological criteria in clinical practice. However, the concordance for molecular subtype classification between immunohistochemical surrogates and formal genetic analysis ranges from 41% to 100%, and the use of surrogates has been shown to be less robustly predictive of outcomes (3). As a result of the limitations, there is a demand for additional alternative methods that can allow differentiation of breast cancer into molecular subtypes.
Previous studies indicate that the mammographic appearance could be correlated with molecular subtype and provide a new perspective to understand the biologic behavior of the breast tumor (456). In particular, several investigators recently reported that masses with a spiculated margin were significantly more common in patients with luminal A breast cancer than in those with other subtypes and may be an independent, good prognostic factor for the disease (46). However, questions regarding the mechanism and significant factors affecting the presence of a spiculated mass remain unanswered.
The purpose of our study was to determine the relationship between the presence of a spiculated mass and luminal A subtype of invasive breast cancer using receptors as surrogate markers, and the factors which may influence the presence or absence of a spiculated mass.
Our Institutional Review Board approved the study protocol and waived the requirement for informed consent. This study retrospectively analyzes a consecutive series of primary invasive carcinoma of the breast.
In our cohort, we included three hundred seventeen (317) Chinese patients with primary invasive breast cancer who were diagnosed at the Breast Center of the Affiliated Hospital of Qingdao University (AHQU) between December 2014 and April 2015.
The inclusion criteria were as follows: 1) adequate excision of the patient's tumors (mastectomy or lumpectomy with negative margins), 2) histological confirmation of invasive breast carcinoma and pathology information regarding receptor status were available, and 3) a pre-operative mammogram examination. All 317 patients in the cohort met the inclusion criteria. Exclusion criteria were: 1) male patient sex (n = 1), 2) no diagnostic mammogram performed at the institution (n = 7), 3) prior history of breast injury or percutaneous core biopsy before mammogram examination (n = 2), and 4) neo-adjuvant therapy received prior to surgery (n = 31). Patients were excluded from the study if they met any of the above-mentioned exclusion criteria. Consequently, 41 patients were excluded.
All mammograms were performed with Selenia Dimensions 3D Digital Mammography Tomosynthesis System (Hologic, Bedford, MA, USA) and reviewed on high-resolution picture archiving and communication system workstations at the imaging department of the breast center of AHQU. Craniocaudal view and mediolateral view were performed routinely for all patients, and mediolateral oblique view was obtained when required.
All the mammograms were reviewed by two radiologists (with 12 and 3 years of experience in the interpretation of breast images, respectively) according to the analytic criteria of Breast Imaging Reporting and Data System, and the mammographic features were recorded respectively (7). Since the doctor was blinded to biopsy results, it was apparent that the molecular subtypes will not have any effect on the evaluation of the lesions.
In this study, patients with a mass on the mammogram were divided into groups of spiculated and non-spiculated according to the margin status of the mass, respectively. Spiculated masses are defined as masses with lines radiating from their margins (Fig. 1). Lesions classified as non-spiculated were circumscribed, microlobulated, obscured or indistinct (Figs. 2, 3). The non-spiculated category also included cases without a mass and cases with extremely dense breast that made it impossible to note any abnormal findings. If the findings of spiculation were difficult to be determined precisely (for example, possibly related to overlapping structures), the case was classified as non-spiculated. When a mass could not be clearly visualized and spiculations were seen, the lesion was considered to be an architectural distortion, and was classified as non-spiculated.
Pathologic assessment of all surgical cases was performed by a breast pathologist with more than 8 years of experience. Pathology type, grade, tumor diameter, estrogen receptor (ER), progesterone receptor (PR), human epithelial growth factor receptor 2 (HER2) status and Ki67 were recorded. Tumors were considered positive for ER or PR if immunostaining was observed in more than 1% percent of tumor nuclei. HER2 positivity was defined as overexpression by FISH analysis.
We used combinations of ER, PR, HER2, and Ki67 as surrogate markers to identify molecular subtypes of breast cancer. To avoid confounding of HER2 effects, luminal B breast cancers were divided into two subgroups, luminal B for HER2-negative and luminal HER2 for HER2-positive. In this study, we defined the luminal A subtype as being ER and/or PR-positive, HER2-negative, and Ki67-low (< 14%); the luminal B subtype was defined as being ER and/or PR-positive, HER2-negative, and Ki67-high (≥ 14%); the luminal HER2 subtype was defined as being ER and/or PR-positive, HER2 positive, any Ki67; the HER2 subtype was defined as ER and PR negative with HER2 overexpression; and the triple-negative breast cancer (TNBC) subtype was defined as ER-negative, PR-negative, and HER2-negative.
We performed univariate analyses with Pearson chi-square tests and multivariate analyses with logistic regression for factors correlating to mammographic and molecular subtypes. A p value of less than 0.05 was considered to indicate a statistically significant difference. We performed all statistical analyses with SPSS 21.0 for Windows (SPSS Inc., Chicago, IL, USA).
We captured complete data on receptor status and mammographic appearance from the entire cohort. The median age of the patients at diagnosis was 49 years (range; 20 to 81 years). Mean tumor size for the study group was 20 mm. Ninety-two percent (292/317) of patients presented with an invasive ductal carcinoma, while the remaining eight percent (25/317) was composed of invasive lobular carcinoma (n = 7), invasive papillary carcinoma (n = 6), mucinous carcinoma (n = 5), medullary carcinoma (n = 3), tubular carcinoma (n = 2), invasive carcinoma with endocrine differentiation (n = 1), and invasive apocrine carcinoma (n = 1). Twenty-two percent (69/317) of the patients presented with a spiculated mass during their mammography, while the remaining seventy-eight percent (248/317) of the patients did not.
Table 1 shows the association of demographic and tumor characteristics with molecular subtype. Age, grade, and tumor size were associated with molecular subtype as classified by receptor combination. There were no statistically significant associations between the histologic types and molecular expression. Luminal cancers were mainly composed of low and middle pathology grade, and were more likely to occur in young patients.
Table 2 shows the associations of the presence of a spiculated mass with molecular subtype. There was a statistically significant difference in the presence of a spiculated mass across molecular subtype. Patients with the Luminal A subtype had 10.3 times greater odds of having presented with a spiculated mass on their mammograms when compared to the other subtypes (p < 0.001).
Table 3 shows the associations between the presence of a spiculated mass and clinicopathologic variables. Univariate analysis revealed significant association of breast composition, pathology grade, hormone receptor status (ER and PR), Ki67 index and HER2 status with the presence of spiculation. In order to stratify the contribution of the covariates to the presence of spiculation, multivariate analyses were performed. The patients with low Ki67 index (< 14%) and HER2 negative were most likely than the others to present with a spiculated mass on their mammograms (p < 0.001). The hormone receptor status (ER and PR), pathology grade, overall breast composition, were all associated with the presence of a spiculated mass, but with less weight in contribution than Ki67 and HER2.
Our study shows that the presence of a spiculated mass is associated with the luminal A subtype of invasive breast cancer, and the status of Ki67 and HER2 may perhaps be the most significant factors affecting the visualization of a spiculated mass.
Spiculation is a characteristic appearance of invasive breast cancer at mammography and a well-known criterion in the diagnosis of the disease. The characteristics of spiculated masses continue to attract interest of several researchers (4589101112). Several investigators recently reported that masses with a spiculated margin were significantly more common in patients with the luminal A breast cancer than in those with other subtypes (45). Several factors, including higher ER-positive or PR-positive rates, HER2 negativity and lower Ki67 index, were believed to contribute to the visualization of spiculation (412). However, questions regarding the most significant factor affecting the presence of a spiculated mass remain unanswered.
We performed our study in several different ways to answer these questions. First, we divided luminal B cancers into two subgroups to stratify the effect of HER2 status for the presence of a spiculated mass. Second, we used a logistic regression model, which considered a large number of confounders of the relationship between molecular subtype and the presence of a spiculated mass.
The logistic regression model shows the relative importance of the association between the luminal A subtype and the presence of a spiculated mass. The luminal A subtype is the most important factor for the presence of a spiculated mass when compared to the luminal B subtype (with or without HER2 expression), HER2 overexpression and TNBC. In addition, we observed that HER2 overexpressing breast cancers never presented a spiculated mass, though the exact reason remained unclear.
We further explored the correlation between spiculation and clinicopathological characteristics by building a logistic regression model. The results of the present study demonstrated that the presence of a spiculated mass on mammography was strongly associated with HER2 negativity and the lower proliferative activity as shown by Ki67 labeling, followed by positive expression of hormone receptors with less weight. Our results are somewhat in accordance with the immunohistochemistry criteria of the luminal A subtype. We also observed a statistically significant association between low and intermediate histological grade tumors and the presence of a spiculated mass. Several investigators have reported that a low histologic tumor grade is one of the reasons for spiculation (412). Our findings are consistent with the reported data, and may explain the notation from previous reports that spiculated masses can be an excellent prognostic factor in disease (589). In addition, we found that patients with low-fibroglandular-density breasts had a greater likelihood of presenting with a spiculated mass as compared to patients with high-fibroglandular-density breasts. It is unclear whether spiculated tumors are more common in women with fatty breasts or if the lesions are more frequently characterized as spiculated in non-dense breasts. The reason for a spiculated mass was more common seen in low-fibroglandular-density breasts was most likely due to the greater sensitivity in detecting mass margin in breasts that have less fibroglandular density (10).
In general, the formation of a spiculated mass is thought to represent the in-pulling of normal Cooper's ligaments into a tumor or the invasion of tumor cells into the surrounding tissue (111314). It is unclear whether either of the two processes dominates in the visualization of a spiculated lesion on mammography. One previous analysis showed that low grade or luminal A breast cancer seems to exhibit much more interaction with stromal tissue; furthermore, all patients without adipose tissue invasion had nonspiculated lesions (11). Moreover, it was believed that spicular formation is a result of two time-dependent steps of low-grade cancer (11). The destructive invasion of cancer cells with desmoplastic stromal reactions occurs initially, followed by adipose tissue invasion and/or involvement of Cooper ligaments by carcinoma tissue (11). Therefore, visualization of a spiculated mass on mammography has at least two-fold meanings. Firstly, it is a predictor for low grade luminal A subtype of breast cancer. Secondly, it is a flag for late stage as a result of adipose tissue invasion by carcinoma tissue.
It is hypothesized that improved understanding of the correlation between the presence of a spiculated mass and luminal A breast cancer may provide information about underlying tumor biology and help guide clinical decision making. When put into context with clinical information, additional important descriptors could help radiologists make decisions regarding workup and treatment before receptors are available. For example, referral of the patient to a medical oncology professional for endocrinotherapy will allow for early detection of a spiculated mass (56). Additionally, all laboratory tests have an incidence of false negative and false positive results, and the appearance of a spiculated mass on a mammogram could signal a possible discrepancy. For example, if a cancer is presented with a spiculated mass on mammogram and found to be HER2 overexpressing, a flag can be raised to indicate a discordant result. As HER2 overexpressing cancers almost never present as spiculated masses, it might be worthwhile to repeat pathology testing of the receptors (615).
There were limitations to our study. First, the study was retrospective in nature and was performed in a single institute. Second, our study evaluated only masses identified on diagnostic mammography, which limits the generalizability of our results to the larger screening population.
In conclusion, we found that the presence of a spiculated mass on a mammogram is associated with the luminal A subtype of breast cancer, and lower Ki67 index and HER2 negativity may be the most significant factors in the presence of a spiculated mass.
Figures and Tables
Fig. 1
Luminal A breast cancer (Ki67 = 10%, HER2 negative) presenting as mass with representative spiculated margin on mammogram.
HER2 = human epithelial growth factor receptor 2
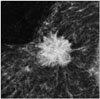
Fig. 2
Triple negative breast cancer (Ki67 = 60%, HER2 negative) presenting as mass with circumscribed margin on mammogram.
HER2 = human epithelial growth factor receptor 2
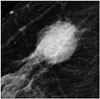
Fig. 3
HER2 breast cancer (Ki67 = 40%, HER2 positive) presenting as mass with indistinct margin on mammogram.
HER2 = human epithelial growth factor receptor 2
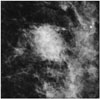
Table 1
Associations of Demographic and Tumor Characteristics to Molecular Subtype

Table 2
Associations of Presence of Spiculated Mass to Molecular Subtype

Table 3
Associations between Presence of Spiculated Mass and Clinicopathologic Variables
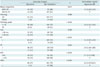
Unless otherwise specified, data are numbers of masses, with percentages in parentheses. *Almost entirely fat or scattered fibroglandular densities, †Heterogeneously dense or extremely dense, ‡Data in parentheses are 95% confidence intervals, §Reference level in OR calculation. ACR = categories of American College of Radiology in breast composition, ER = estrogen receptor, HER2 = human epithelial growth factor receptor 2, IDC = invasive ductal carcinoma, OR = odds ratio, PR = progesterone receptor
References
1. Huber KE, Carey LA, Wazer DE. Breast cancer molecular subtypes in patients with locally advanced disease: impact on prognosis, patterns of recurrence, and response to therapy. Semin Radiat Oncol. 2009; 19:204–210.
2. Lam SW, Jimenez CR, Boven E. Breast cancer classification by proteomic technologies: current state of knowledge. Cancer Treat Rev. 2014; 40:129–138.
3. Guiu S, Michiels S, André F, Cortes J, Denkert C, Di Leo A, et al. Molecular subclasses of breast cancer: how do we define them? The IMPAKT 2012 Working Group Statement. Ann Oncol. 2012; 23:2997–3006.
4. Jiang L, Ma T, Moran MS, Kong X, Li X, Haffty BG, et al. Mammographic features are associated with clinicopathological characteristics in invasive breast cancer. Anticancer Res. 2011; 31:2327–2334.
5. Tamaki K, Ishida T, Miyashita M, Amari M, Ohuchi N, Tamaki N, et al. Correlation between mammographic findings and corresponding histopathology: potential predictors for biological characteristics of breast diseases. Cancer Sci. 2011; 102:2179–2185.
6. Killelea BK, Chagpar AB, Bishop J, Horowitz NR, Christy C, Tsangaris T, et al. Is there a correlation between breast cancer molecular subtype using receptors as surrogates and mammographic appearance? Ann Surg Oncol. 2013; 20:3247–3253.
7. American College of Radiology. Breast imaging reporting and data system (BI-RADS). 4th ed. Reston, VA: American College of Radiology;2003.
8. Alexander MC, Yankaskas BC, Biesemier KW. Association of stellate mammographic pattern with survival in small invasive breast tumors. AJR Am J Roentgenol. 2006; 187:29–37.
9. Evans AJ, Pinder SE, James JJ, Ellis IO, Cornford E. Is mammographic spiculation an independent, good prognostic factor in screening-detected invasive breast cancer? AJR Am J Roentgenol. 2006; 187:1377–1380.
10. Porter GJ, Evans AJ, Cornford EJ, Burrell HC, James JJ, Lee AH, et al. Influence of mammographic parenchymal pattern in screening-detected and interval invasive breast cancers on pathologic features, mammographic features, and patient survival. AJR Am J Roentgenol. 2007; 188:676–683.
11. Yamaguchi J, Ohtani H, Nakamura K, Shimokawa I, Kanematsu T. Prognostic impact of marginal adipose tissue invasion in ductal carcinoma of the breast. Am J Clin Pathol. 2008; 130:382–388.
12. Moriuchi H, Yamaguchi J, Hayashi H, Ohtani H, Shimokawa I, Abiru H, et al. Cancer cell interaction with adipose tissue: correlation with the finding of spiculation at mammography. Radiology. 2016; 279:56–64.
13. Tchou J, Kossenkov AV, Chang L, Satija C, Herlyn M, Showe LC, et al. Human breast cancer associated fibroblasts exhibit subtype specific gene expression profiles. BMC Med Genomics. 2012; 5:39.
14. Park SY, Kim HM, Koo JS. Differential expression of cancer-associated fibroblast-related proteins according to molecular subtype and stromal histology in breast cancer. Breast Cancer Res Treat. 2015; 149:727–741.
15. Baré M, Torà N, Salas D, Sentís M, Ferrer J, Ibáñez J, et al. Mammographic and clinical characteristics of different phenotypes of screen-detected and interval breast cancers in a nationwide screening program. Breast Cancer Res Treat. 2015; 154:403–415.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download