This article has been corrected. See "Erratum: Multidetector CT Findings and Differential Diagnoses of Malignant Pleural Mesothelioma and Metastatic Pleural Diseases in Korea" in Volume 17 on page 825.
Abstract
Objective
To compare the multidetector CT (MDCT) features of malignant pleural mesothelioma (MPM) and metastatic pleural disease (MPD).
Materials and Methods
The authors reviewed the MDCT images of 167 patients, 103 patients with MPM and 64 patients with MPD. All 167 cases were pathologically confirmed by sonography-guided needle biopsy of pleura, thoracoscopic pleural biopsy, or open thoracotomy. CT features were evaluated with respect to pleural effusion, pleural thickening, invasion of other organs, lung abnormality, lymphadenopathy, mediastinal shifting, thoracic volume decrease, asbestosis, and the presence of pleural plaque.
Results
Pleural thickening was the most common CT finding in MPM (96.1%) and MPD (93.8%). Circumferential pleural thickening (31.1% vs. 10.9%, odds ratio [OR] 3.670), thickening of fissural pleura (83.5% vs. 67.2%, OR 2.471), thickening of diaphragmatic pleura (90.3% vs. 73.4%, OR 3.364), pleural mass (38.8% vs. 23.4%, OR 2.074), pericardial involvement (56.3% vs. 20.3%, OR 5.056), and pleural plaque (66.0% vs. 21.9%, OR 6.939) were more frequently seen in MPM than in MPD. On the other hand, nodular pleural thickening (59.2% vs. 76.6%, OR 0.445), hilar lymph node metastasis (5.8% vs. 20.3%, OR 0.243), mediastinal lymph node metastasis (10.7% vs. 37.5%, OR 0.199), and hematogenous lung metastasis (9.7% vs. 29.2%, OR 0.261) were less frequent in MPM than in MPD. When we analyzed MPD from extrathoracic malignancy (EMPD) separately and compared them to MPM, circumferential pleural thickening, thickening of interlobar fissure, pericardial involvement and presence of pleural plaque were significant findings indicating MPM than EMPD. MPM had significantly lower occurrence of hematogenous lung metastasis, as compared with EMPD.
Malignant pleural mesothelioma (MPM) is the most common primary tumor of pleura and its association with asbestos exposure has been well established. Its incidence has been estimated at 2200–2500 cases per year in the United States (12). Although environmental regulations and a subsequent decrease in exposure to asbestos have led to a decline in its incidence in developed countries, its incidence has yet to peak in developing countries because disease onset lags exposure by approximately two decades (3).
In 2011, the South Korean government enacted and enforced the Asbestos Damage Relief Act and compensated MPM patients. A definitive diagnosis of MPM usually requires histologic sampling and hematoxyline and eosine staining, but nevertheless, its differential diagnosis may be problematic. In particular, the most frequently encountered diagnostic problem is the differentiation of MPM and adenocarcinoma. Accordingly, additional immunohistochemical studies are often needed to confirm a diagnosis of MPM (3). Sometimes radiologic differential diagnosis may be required for the judgment of compensation, if the pathologic diagnosis is uncertain or the patient is incapable of receiving a biopsy.
Imaging plays an important role in the diagnosis, treatment planning, and post-treatment assessment of MPM. Although CT is the first-line imaging modality for the evaluation of MPM, it has its limitations. A variety of benign and malignant pleural diseases may cause pleural abnormalities that resemble MPM, and thus, early diagnosis is often difficult. The most common causes of misdiagnosis are metastatic pleural disease (MPD), tuberculous pleurisy, and empyema (45). The CT features that aid in the differential diagnosis of malignant and benign pleural diseases are; 1) circumferential pleural thickening, 2) nodular pleural thickening, 3) pleural thickening > 10 mm, and 4) mediastinal pleural involvement (45). Though these findings are frequently seen in MPM, they are not characteristic of MPM. In one study, CT findings of rind-like pleural involvement, mediastinal pleural involvement, and a pleural thickness exceeding 1 cm were suggested to distinguish between MPM and MPD (5); however, CT scans of 10 mm slice thickness were used. Most reported studies for CT findings of MPM were performed by the use of thick-slice images or only evaluation of transverse images (45678910).
In the present study, we evaluated multidetector row CT (MDCT) images (slice thickness ≤ 5 mm) with coronal or sagittal reformatted images of MPM patients and compared them with MPD patients.
The Institutional Review Boards of participating hospitals approved the study and waived the requirement for informed consent due to its retrospective nature.
This study was performed as a part of a project entitled "Diagnostic guidelines for asbestos-related diseases caused by environmental exposure in Korea". The Korean mesothelioma surveillance system was initiated in 2001 by the Cardiopulmonary Study Group of the Korean Society of Pathologists (11). The records of more than 500 MPM cases treated at hospitals nationwide were archived in the database. For comparison purposes, we enrolled MPD patients treated at eight different tertiary hospitals. The inclusion criteria were; 1) pathologic confirmation based on examinations of pleural tissue samples obtained by sonography-guided needle biopsy of pleura, thoracoscopic pleural biopsy, or open thoracotomy, and 2) available CT images obtained by MDCT (with more than 4 detector rows) with coronal and sagittal reformation, a slice thickness of ≤ 5 mm, and contrast enhancement. Lung pathologists from the Cardiopulmonary Study Group of the Korean Society of Pathologists reviewed slides to confirm diagnoses. Immunohistochemical staining was also conducted when a diagnosis was indefinite by hematoxyline and eosine staining. Confused cases or cases without available tissue samples were excluded. Accordingly, only patients with a definite histopathological diagnosis of MPM or MPD were included. Finally, 167 patients (103 with MPM and 64 with MPD) were enrolled in the study.
CT scans were evaluated retrospectively by two thoracic radiologists who had 21 and 12 years of experience of interpreting chest CT images. Both were unaware of histopathologic diagnoses. Conclusions were reached by consensus. Images were reviewed with respect to disease laterality, pleural effusion, pleural thickening, lymph node enlargement, mediastinal shift, thoracic volume decrease, invasion of other organs, lung abnormalities, and the presence of asbestos-related abnormalities such as asbestosis and pleural plaque. Transverse images were evaluated for general review and coronal and sagittal reformatted images were used to evaluate fissures, diaphragms, pericardia, and lung parenchyma.
Pleural effusion was classified as small, moderate, or large; defined as occupying < one-third, between one-third and two-thirds, and > two-thirds of the hemithorax, respectively. Pleural thickening was classified as thin (thickest point on axial images < 10 mm), nodular (focal pleural thickening with a short diameter between 10 and 30 mm), circumferential (continuous pleural thickening occupying > three-quarters of the hemithorax on axial images), or as a mass (focal pleura-based lesion with a short diameter of > 30 mm). For the evaluation of lymph nodes, internal mammary nodes were considered abnormal when larger than their accompanying vessels. Supraclavicular and diaphragmatic nodes were considered abnormal when their short-axis diameters were > 5 mm in the transverse plane (8). Mediastinal and hilar nodes were considered pathologically enlarged when their short-axis diameters were > 10 mm in the transverse plane. Hematogenous lung metastasis was defined as the presence of > 5 non-calcified, circumscribed nodules of > 0.5 cm with random distribution in both lungs. If there was a solitary nodule > 2 cm or dominant mass circumscribed by lung parenchyma or attached to the pleura with an acute angle, the lesion was considered as primary lung tumor. Mediastinal shifting was defined as the dislocation of mediastinal structures due to pleural lesions. Thoracic volume decrease was defined as decreased volume of hemithorax in the involved side compared with opposite side, and assessed by analyzing thoracic cage configurations and the positions of mediastina and diaphragms. In addition, asbestos-related lung and pleural abnormalities, such as, asbestosis and pleural plaques, were recorded. Asbestosis was defined as the presence of the CT findings such as subpleural dot-like or branching opacities, subpleural curvilinear lines, intralobular interstitial thickening, interlobular septal thickening, parenchymal bands, and honeycombing (1213). Pleural plaque was defined as variable-size localized pleural thickenings of soft tissue, or calcific densities attached along the pleura of the chest wall, diaphragm, and mediastinum (14). In the involved side of pleural disease, only calcified plaque was recorded, and noncalcified pleural plaque was evaluated only in the opposite hemithorax. Plaque extent was estimated according to the involvement of the circumference of the lung, by combined maximum lengths of plaques on axial image at the mid-thoracic level; grade 1 < 1/4, grade 2 = 1/4–1/2, and grade 3 > 1/2 of the hemithorax (15).
Statistical analysis was performed using SPSS ver. 21 (SPSS Inc., Chicago, IL, USA). Demographic data and the frequencies of CT findings in the MPM and MPD groups were compared using the chi-square test and Fisher's exact test. p values of < 0.05 were considered statistically significant. The importance of findings was assessed using p values, odds ratios (ORs), and 95% confidence intervals.
Patient demographics were summarized in Table 1. Pathologic subtypes of MPM were epithelioid in 52 (50.5%), biphasic in 8 (7.8%), sarcomatoid in 2 (1.9%), desmoplastic in 6 (5.8%), and undetermined in 35 (34%). Of the 64 patients with MPD, 40 (62.5%) had lung cancer (22 adenocarcinomas, 5 non-small cell lung cancers, 5 small cell lung cancers, 3 large cell neuroendocrine carcinomas, 3 squamous cell carcinomas, and others), 9 had carcinoma from different extrathoracic sites, 4 had breast cancer, 3 had renal cell carcinoma, 3 had thymic epithelial tumor, 3 had sarcoma from different extrathoracic organs, and 2 had lymphoma.
The most common CT finding was pleural thickening in both MPM (96.1%) and MPD (93.8%), and the second most common finding was pleural effusion in both MPM (91.3%) and MPD (87.5%). Circumferential pleural thickening, thickening of interlobar fissure, thickening of diaphragmatic pleura, pericardial involvement, and the presence of a pleural mass and of pleural plaque were more frequent in MPM. Nodular pleural thickening, hilar lymph node enlargement, mediastinal lymph node enlargement, presence of primary lung tumor and hematogenous lung metastasis were more frequent in MPD than MPM (Table 2, Figs. 1, 2, 3).
When we analyzed MPD from extrathoracic malignancy (EMPD) separately and compared them to MPM, circumferential pleural thickening, thickening of interlobar fissure, pericardial involvement and presence of pleural plaque were significant findings indicating MPM than EMPD. MPM had significantly lower occurrence of hematogenous lung metastasis compared with EMPD (Table 3).
In the present study, we evaluated the MDCT features of pathologically confirmed MPM and MPD. The analysis was performed using multiplanar reformatted and thin slice thickness images, and thus, was more detailed than those performed previously (45678910). MDCT with multiplanar reformation might improve the accuracy of tumor detection, because it provides better visualization of tumor extent, especially in regions like the diaphragm, which can be difficult to assess using axial images (16). In the present study, diaphragmatic pleural thickening was observed in 90% of the MPM patients and was valuable for distinguishing between MPM and MPD (OR = 3.364). In previous studies, the prevalence of diaphragmatic pleural thickening has been found to vary from 6.1 to 76% in MPM (78). However, variation in prevalence was probably related to CT image quality and the availability of coronal reformatted images. In the present study, pericardial involvement was observed in 56.3% of MPM patients and was significantly more prevalent in MPM than in MPD (OR = 5.056). When we reviewed the axial images of MPM or MPD patients, pericardial thickening was often confused with mediastinal pleural thickening in some areas, and coronal or sagittal reformatted images were helpful to decide the pericardial involvement.
Although CT remains the primary imaging modality for the evaluation of MPM, the differentiation of MPM and MPD is difficult because their CT features are similar. Metastatic pleural involvement is the most common type of pleural malignancy, and the predominant underlying primary diseases are bronchogenic carcinoma (40%), breast carcinoma (20%), and lymphoma (10%) followed by gastrointestinal and genitourinary malignancies (17). The most frequent manifestation of metastatic pleural involvement is pleural effusion (4). Circumferential pleural thickening, nodular pleural thickening, involvement of interlobar fissures, mediastinal pleural involvement, contraction of pleura, and mediastinal lymph node invasion are considered strongly suggestive of malignant pleural disease (45618), but none are pathognomonic for MPM. According to Metintas et al. (5), pleural rind (circumferential pleural thickening with mediastinal pleural involvement) is the most specific finding (specificity 85%) for distinguishing between MPM and MPD. In agreement with previous studies, circumferential pleural thickening was one of the more significant findings indicating MPM (OR = 3.670) rather than MPD in our study.
Mediastinal and hilar lymph node enlargement was significantly more common in patients with MPD, which may have been related to the larger proportion of lung cancer patients in the MPD group. Hilar lymph nodes drain lungs and visceral pleura, whereas internal mammary, cardiophrenic, extrapleural, and intercostal nodes drain parietal pleura (81920). In a study that addressed the prevalence of lymph node metastasis in MPM, Abdel Rahman et al. (20) concluded that hilar node involvement occurred secondary to parenchymal infiltration and was not due to direct spread from pleura, whereas extrapleural and cardiophrenic nodes were primarily involved in MPM. Feragalli et al. (19) concluded that the presence of extrapleural and cardiophrenic nodes associated with other signs of MPM, especially with involvement of mediastinal pleura and volume loss of affected hemithorax, could play an important diagnostic role in the early diagnosis of MPM. Seely et al. (8) analyzed CT images of 92 MPM patients and reported the prevalence of internal mammary, cardiophrenic, and retropleural lymphadenopathy as 52%, 46%, and 33%, respectively, whereas, mediastinal and hilar lymphadenopathy were 23% and 0%, respectively. The authors concluded that the higher prevalence of these extrapleural nodes in their study than in previous studies were probably due to the use of MDCT.
Pleural plaques are the most common radiological manifestations of asbestos exposure (2122). In the present study, the prevalence of pleural plaque, defined as calcified or noncalcified plaque regardless of size, was 66% in MPM, which is higher than previously reported values, which ranged from 8% to 43% (689). In some studies, pleural plaque was defined as transverse or craniocaudal pleural thickening of < 5 cm in length (78), whereas others only evaluated calcified pleural plaque. Accordingly, the definition of pleural plaque used in the present study was based on review of the thin section and coronal reformatted images. Seely et al. (8) reported the prevalence of calcified plaque and noncalcified plaque of 43% and 35%, respectively, in 92 MPM patients. However, they included 27 patients scanned by single track CT and 65 patients (71%) scanned by MDCT. In the present study, pleural plaque was observed in 14 (21.9%) of the 64 MPD patients, and the primary lesions were lung cancer in 11, ovary cancer in 1, extrathoracic liposarcoma in 1 and invasive ductal carcinoma in 1 patient.
When CT images suggest malignant pleural disease, primary tumor in the lung should be ruled out, because bronchogenic carcinoma is the most common cause of the MPD (17). Therefore, we analyzed CT findings of EMPD separately and compared them with MPM, because both of them hardly show the primary tumor in the lung. Circumferential pleural thickening, fissural pleural thickening, pericardial involvement, presence of pleural plaque, and absence of hematogenous pulmonary metastasis were significant findings indicating MPM, as compared with EMPD. Although some patients may have a single pulmonary metastasis mimicking primary lung tumor, these findings may be helpful in confirming malignant pleural diseases without primary lung tumor.
The present study has some limitations that require consideration. First, it is inherently limited by its retrospective nature. In particular, observers were unaware of histological results but aware of the presence of malignant pleural disease, which may have increased sensitivities for the detections of pleural thickening and adenopathy. Second, imaging data were obtained using various scanners and protocols, and the resulting variations in image qualities could have affected evaluations of thin pleural plaque and asbestosis of mild degree. Third, no pathological proof of lymph node metastasis or lung metastasis was obtained, because although some patients underwent extrapleural pneumonectomy, radiologic-pathologic correlations were not possible for individual lymph nodes or lung nodules.
In summary, circumferential pleural thickening, fissural pleural thickening, diaphragmatic pleural thickening, pericardial involvement, and the presence of a pleural mass and pleural plaque were significantly more frequent CT findings in MPM, whereas nodular pleural thickening, hilar lymph node enlargement, mediastinal lymph node enlargement, and hematogenous lung metastasis were more frequent CT findings in MPD. Circumferential pleural thickening, fissural pleural thickening, pericardial involvement, the presence of pleural plaque and absence of hematogenous pulmonary metastasis were findings indicating MPM rather than EMPD. Awareness of frequent and infrequent CT findings could aid in distinguishing MPM from MPD.
Figures and Tables
Fig. 1
CT findings of MPM and MPD.
A, B. 48-year-old woman with epithelioid type MPM. Circumferential pleural thickening and fissural pleural thickening (arrows) involving right hemithorax. C, D. 82-year-old woman with MPD from renal cell carcinoma. Nodular pleural thickeing involving both hemithorax and hematogenous metastases in both lungs. CT = computed tomography, MPD = metastatic pleural disease, MPM = malignant pleural mesothelioma
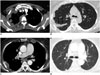
Fig. 2
63-year-old woman with bilateral MPM involving pericardium and both diaphragmatic pleura.
Transverse CT images (A, B) and coronal reformatted image (C) show bilateral pleural effusion, pericardial thickening (arrows) and thickening of both diaphragmatic pleura. MPM = malignant pleural mesothelioma
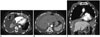
Fig. 3
71-year-old man with MPM.
Transverse CT images show circumferential pleural thickening in right hemithorax. Note calcified pleural plaques in both hemithorax (arrows). CT = computed tomography, MPM = malignant pleural mesothelioma

Table 1
Patient Demographics

Table 2
CT Findings of MPM and MPD

Table 3
CT Findings of MPM and EMPD

References
1. Antman KH. Natural history and epidemiology of malignant mesothelioma. Chest. 1993; 103:4 Suppl. 373S–376S.
2. Nickell LT Jr, Lichtenberger JP 3rd, Khorashadi L, Abbott GF, Carter BW. Multimodality imaging for characterization, classification, and staging of malignant pleural mesothelioma. Radiographics. 2014; 34:1692–1706.
3. Robinson BW, Lake RA. Advances in malignant mesothelioma. N Engl J Med. 2005; 353:1591–1603.
4. Leung AN, Müller NL, Miller RR. CT in differential diagnosis of diffuse pleural disease. AJR Am J Roentgenol. 1990; 154:487–492.
5. Metintas M, Ucgun I, Elbek O, Erginel S, Metintas S, Kolsuz M, et al. Computed tomography features in malignant pleural mesothelioma and other commonly seen pleural diseases. Eur J Radiol. 2002; 41:1–9.
6. Kawashima A, Libshitz HI. Malignant pleural mesothelioma: CT manifestations in 50 cases. AJR Am J Roentgenol. 1990; 155:965–969.
7. Okten F, Köksal D, Onal M, Ozcan A, Sims¸ek C, Ertürk H. Computed tomography findings in 66 patients with malignant pleural mesothelioma due to environmental exposure to asbestos. Clin Imaging. 2006; 30:177–180.
8. Seely JM, Nguyen ET, Churg AM, Müller NL. Malignant pleural mesothelioma: computed tomography and correlation with histology. Eur J Radiol. 2009; 70:485–491.
9. Ng CS, Munden RF, Libshitz HI. Malignant pleural mesothelioma: the spectrum of manifestations on CT in 70 cases. Clin Radiol. 1999; 54:415–421.
10. Tamer Dogan O, Salk I, Tas F, Epozturk K, Gumus C, Akkurt I, et al. Thoracic computed tomography findings in malignant mesothelioma. Iran J Radiol. 2012; 9:209–211.
11. Jung SH, Kim HR, Koh SB, Yong SJ, Chung MJ, Lee CH, et al. A decade of malignant mesothelioma surveillance in Korea. Am J Ind Med. 2012; 55:869–875.
12. Akira M, Yamamoto S, Yokoyama K, Kita N, Morinaga K, Higashihara T, et al. Asbestosis: high-resolution CT-pathologic correlation. Radiology. 1990; 176:389–394.
13. Akira M, Yokoyama K, Yamamoto S, Higashihara T, Morinaga K, Kita N, et al. Early asbestosis: evaluation with high-resolution CT. Radiology. 1991; 178:409–416.
14. Kim Y, Myong JP, Lee JK, Kim JS, Kim YK, Jung SH. CT characteristics of pleural plaques related to occupational or environmental asbestos exposure from South Korean asbestos mines. Korean J Radiol. 2015; 16:1142–1152.
15. Kusaka Y, Hering KG, Parker JE. Coding CT-classification in occupational and environmental respiratory disease (OERD). In : Kusaka Y, Hering KG, Parker JE, editors. International classification of HRCT for occupational and environmental respiratory diseases. Tokyo: Springer;2005. p. 20–21.
16. Wang ZJ, Reddy GP, Gotway MB, Higgins CB, Jablons DM, Ramaswamy M, et al. Malignant pleural mesothelioma: evaluation with CT, MR imaging, and PET. Radiographics. 2004; 24:105–119.
17. Qureshi NR, Gleeson FV. Imaging of pleural disease. Clin Chest Med. 2006; 27:193–213.
18. Sahin AA, Cöplü L, Selçuk ZT, Eryilmaz M, Emri S, Akhan O, et al. Malignant pleural mesothelioma caused by environmental exposure to asbestos or erionite in rural Turkey: CT findings in 84 patients. AJR Am J Roentgenol. 1993; 161:533–537.
19. Feragalli B, Mantini C, Civitareale N, Polverosi R, Tartaro A, Cotroneo AR. Extrapleural and cardiophrenic lymph nodes: prevalence, clinical significance and diagnostic value. Radiol Med. 2014; 119:20–26.
20. Abdel Rahman AR, Gaafar RM, Baki HA, El Hosieny HM, Aboulkasem F, Farahat EG, et al. Prevalence and pattern of lymph node metastasis in malignant pleural mesothelioma. Ann Thorac Surg. 2008; 86:391–395.
21. Aberle DR, Gamsu G, Ray CS. High-resolution CT of benign asbestos-related diseases: clinical and radiographic correlation. AJR Am J Roentgenol. 1988; 151:883–891.
22. Meirelles GS, Kavakama JI, Jasinowodolinski D, Nery LE, Terra-Filho M, Rodrigues RT, et al. Pleural plaques in asbestos-exposed workers: reproducibility of a new high-resolution CT visual semiquantitative measurement method. J Thorac Imaging. 2006; 21:8–13.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download