Abstract
Objective
To test the hypothesis that a safety margin may affect local tumor recurrence (LTR) in subsegmental chemoembolization.
Materials and Methods
In 101 patients with 128 hepatocellular carcinoma (HCC) nodules (1-3 cm in size and ≤ 3 in number), cone-beam CT-assisted subsegmental lipiodol chemoembolization was performed. Immediately thereafter, a non-contrast thin-section CT image was obtained to evaluate the presence or absence of intra-tumoral lipiodol uptake defect and safety margin. The effect of lipiodol uptake defect and safety margin on LTR was evaluated. Univariate and multivariate analyses were performed to indentify determinant factors of LTR.
Results
Of the 128 HCC nodules in 101 patients, 49 (38.3%) nodules in 40 patients showed LTR during follow-up period (median, 34.1 months). Cumulative 1- and 2-year LTR rates of nodules with lipiodol uptake defect (n = 27) and those without defect (n = 101) were 58.1% vs. 10.1% and 72.1% vs. 19.5%, respectively (p < 0.001). Among the 101 nodules without a defect, the 1- and 2-year cumulative LTR rates for nodules with complete safety margin (n = 52) and those with incomplete safety margin (n = 49) were 9.8% vs. 12.8% and 18.9% vs. 19.0% (p = 0.912). In multivariate analyses, ascites (p = 0.035), indistinct tumor margin on cone-beam CT (p = 0.039), heterogeneous lipiodol uptake (p = 0.023), and intra-tumoral lipiodol uptake defect (p < 0.001) were determinant factors of higher LTR.
Recently, cone-beam CT hepatic arteriography has served as a guiding tool to better select feeding arteries to hepatocellular carcinoma (HCC) lesions. It might lead to better therapeutic efficacy with lower local tumor recurrence (LTR) after subsegmental transarterial chemoembolization (TACE). Even after a curative treatment such as surgical resection or radiofrequency ablation (RFA), tumor recurrence is commonly observed (123). To lower LTR, performing anatomical resection in surgery or ablation of parenchymal tissues surrounding the tumor in RFA has been commonly used (456). With surgical resection and RFA, embolization of liver parenchyma surrounding the tumor nodule in TACE may reduce LTR. However, the effect of a 'safety margin' on LTR has been rarely investigated in TACE. Therefore, the aim of this study was to test the hypothesis that achieving a safety margin around the tumor nodule might affect the LTR in cone-beam CT-assisted subsegmental TACE for small nodular HCC.
We performed a single-institution retrospective study to evaluate the relationship between the presence of a safety margin (defined by parenchymal lipiodol uptake completely surrounding the HCC nodule on non-contrast CT performed immediately after TACE) and cumulative LTR. This study was approved by our Institutional Review Board. The need to obtain informed consent was waived due to its retrospective nature.
Since January 1999, we have prospectively registered all TACE procedures performed in our hospital in an electronic database. From March 2009 to August 2011, 514 patients underwent their first session of TACE under cone-beam CT guidance. For this study, we selected patients who satisfied the following inclusion criteria: 1) typical imaging features of HCC based on the American Association for the Study of Liver Disease criteria (7); 2) no prior treatment for HCC; 3) nodular HCCs without vascular invasion or metastasis; 4) tumor size ≤ 3 cm; 5) tumor number ≤ 3; and 6) available 1 mm-thick non-contrast liver CT taken immediately after TACE. Following these inclusion criteria, 128 nodules in 101 patients (77 men, 24 women with mean age of 60.6 ± 9.4 years, range of 40-83 years) were selected for this study. Demographic data of the 101 patients are summarized in Table 1.
To confirm the presence of HCC, pre-TACE imaging with multiphasic contrast-enhanced CT scan (3-phasic or 4-phasic CT) was performed in 54 patients, multiphasic contrast-enhanced MR in 3 patients, both CT and MR in 44 patients.
A 5 Fr sheath was inserted through the right femoral artery to obtain vascular access under local anesthesia. Hepatic arteriography was performed with a 5 Fr catheter (RH catheter; Cook, Bloomington, IN, USA) placed at the common hepatic artery. A cone-beam CT hepatic arteriography was then obtained to identify the tumor-feeding arteries using a uniplanar cone-beam CT scanner (Syngo Dyna CT; Siemens, Forchheim, Germany) (8910). Cone-beam CT acquisition and three-dimensional (3D) image rendering were processed using Syngo InSpace 3D (Siemens Healthcare Sector, Forchheim, Germany). When hepatic artery variations with different origins were encountered during the procedures (11), cone-beam CT hepatic arteriography was performed by placing the catheter at the left and right hepatic arteries separately to obtain the entire hepatic vascular map.
Under the guidance of the obtained 3D imaging, the feeding arteries were selected as close to the tumors as possible beyond the segmental arteries with 2 Fr microcatheter (Progreat; Terumo, Tokyo, Japan). Lipiodol (Lipiodol; Andre Guerbet, Aulnary-sous-Bois, France) and doxorubicin hydrochloride (Adriamycin RDF; Ildong Pharmaceuticals, Seoul, Korea) emulsion made by conventional pumping method was injected until the appearance of the portal venules around the tumor nodules had oily portogram (12). The amount of the lipiodol used ranged from 2 to 6 mL and the dose of doxorubicin used ranged from 10 to 30 mg. Embolization using gelatin sponge particles (Cutanplast; Mascia Brunelli, Milano, Italy) soaked in a mixture of 10 mg of doxorubicin hydrochloride and 10 mL of a nonionic contrast media was performed after the completion of lipiodol emulsion injection.
Immediately after TACE, patients were transferred to a CT room. A 1-mm-thick non-contrast liver CT was taken from each patient to evaluate the distribution of lipiodol in the tumor and the surrounding liver parenchyma. Parameters for multidetector CT used in our institution were: detector configuration at 16 × 0.75 or 64 × 0.625 mm; section thickness at 1 mm.
After TACE, follow-up contrast-enhanced dynamic CT or MR scans were performed at 2- and 3-month intervals to evaluate tumor response and recurrence. If there was no evidence of recurrence after 1 year, follow-up contrast-enhanced dynamic CT or MR was performed at 6-month intervals.
All image analyses were performed by two experienced radiologists by consensus opinion. The pre-TACE multiphase CT or MR studies were assessed for the following features: tumor size, tumor number, morphologic liver cirrhosis, and clinical portal hypertension. The criteria for morphologic liver cirrhosis included liver surface nodularity, edge blunting, and left or caudate lobe hypertrophy (131415). The criteria for clinical portal hypertension were ascites or varices (on endoscopy or multiphasic CT or MR scan) or splenomegaly (≥ 12 cm) combined with thrombocytopenia (< 100000/µL) (161718).
Images obtained during the TACE procedure were assessed for the following features: clarity of tumor margin, tumor-staining homogeneity, tumor vascularity on cone-beam CT images, and pattern of oily portogram (Figs. 1, 2). The pattern of oily portogram was classified into three grades: grade 0 = no obvious branching portal vein visualization; grade 1 = visualization of portal vein adjacent to the tumor; and grade 2 = marked visualization of portal veins in the whole embolized area or extending to the surrounding nonembolized areas (12).
Lipiodol CT images taken immediately after the TACE procedure were assessed for the following features: intensity and homogeneity of lipiodol uptake, presence or absence of a defect in lipiodol uptake, and completeness of the safety margin in the liver parenchyma surrounding the tumor nodule (Figs. 1, 2). The defect in lipiodol uptake was defined as the intra-tumoral area without any lipiodol uptake (Fig. 3). Safety margin was defined as the peri-tumoral parenchymal lipiodol uptake on an immediate non-contrast liver CT. A complete safety margin meant a total encasement of the tumor by lipiodolized parenchyma of any thickness, whereas an incomplete safety margin referred to incomplete encasement of the tumor by lipiodolized parenchyma (Fig. 4). LTR on follow-up CT or MR was determined as the appearance of a viable enhancing area in previously treated nodule.
Because the purpose of this study was to investigate whether establishing a safety margin could reduce local recurrence in subsegmental TACE for small nodular HCCs, it was necessary to exclude cases of incomplete TACE that might manifest as intra-tumoral lipiodol uptake defects on lipiodol CT taken immediately after TACE. Based on the findings on lipiodol CT, treated nodules were divided into two groups with or without a lipiodol uptake defect. The group without lipiodol uptake defect was further divided into two subgroups with or without a complete safety margin, in which cumulative LTR rates were calculated and compared.
To determine significant independent factors predicting LTR, univariate and multivariate analyses were performed for thirteen clinical factors (sex, age, hepatitis B surface antigen, hepatitis C virus antibody, serum aspartate aminotransferase, alanine aminotransferase, albumin, total bilirubin and alpha fetoprotein level, Child-Pugh score, ascites, morphological liver cirrhosis, and clinical portal hypertension), five tumoral factors (tumor size, tumor number, tumor vascularity, tumor stain homogeneity, and tumor margin on cone-beam CT), and five factors related to lipiodol uptake after TACE (oily portogram, lipiodol uptake intensity, lipiodol uptake defect, lipiodol uptake homogeneity, and safety margin).
The initial clinical characteristics were assessed for each patient. The potential risk factors for LTP were analyzed for each nodule using SPSS software for Windows (version 21.0; SPSS Inc., Chicago, IL, USA). On univariate analysis, cumulative LTR during the follow-up period was reported with a 95% confidence interval (CI) using Kaplan-Meier method. As patients who were lost during follow-up or were treated with liver transplantation after TACE were censored, 19 nodules in 15 patients were censored as follows: 13 nodules in 10 patients with follow-up loss, 6 nodules in 5 patients with liver transplantation. A log-rank test was used to compare the curves of each subgroup. Parameters proved to be significant on univariate analysis with two-tail p values less than 0.05 were subsequently tested in multivariate analysis. For multivariate analysis, Cox proportional hazard model was used to assess significant independent factors predicting LTR. For all statistical analyses, two-tail p values less than 0.05 were considered statistically significant.
The median follow-up time was 34.1 months (range, 1-52.7 months). Of the 128 HCC nodules in 101 patients, 49 (38.3%) nodules in 40 patients showed LTR during follow-up period (median, 34.1 months). Of the 128 nodules included in this study, defect in lipiodol uptake was observed in 27 nodules, while no defect was observed in 101 nodules. The 1- and 2-year cumulative LTR rates for nodules with lipiodol uptake defect and those without a defect were 58.1% vs. 10.1% and 72.1% vs. 19.5% (p < 0.001), respectively. Of the 101 nodules without lipiodol uptake defect, 52 had complete safety margins surrounding the tumors, whereas 49 nodules had incomplete safety margins. The 1- and 2-year cumulative LTR rates for nodules with complete safety margin and those with incomplete safety margin were 9.8% vs. 12.8% and 18.9% vs. 19.0% (p = 0.912), respectively. For nodules without defect in lipiodol uptake, the completeness of safety margin in liver parenchyma surrounding the tumor nodules did not affect the LTR (Fig. 5).
In univariate analyses performed for each nodule, variables that significantly contributed to higher LTR rate were: chronic hepatitis C (p = 0.009), ascites (p = 0.011), tumor size (≥ 20 mm) (p = 0.004), indistinct tumor margin on cone-beam CT (p = 0.018), heterogeneous lipiodol uptake in the tumor (p < 0.001), lipiodol uptake defect in the tumor (p < 0.001), and incomplete safety margin in the surrounding liver parenchyma on immediate CT (p = 0.029) (Table 2). In multivariate analyses, ascites (hazard ratio [HR] = 2.17; 95% CI: 1.01, 4.67; p = 0.047), indistinct tumor margin on cone-beam CT (HR = 2.42; 95% CI: 1.13, 5.19; p = 0.039), heterogeneous lipiodol uptake in the tumor (HR = 2.31; 95% CI: 1.09, 4.90; p = 0.029), and lipiodol uptake defect in the tumor (HR = 4.35; 95% CI: 2.33, 8.11; p < 0.001) remained significant contributors to higher LTR rates (Table 3). In subgroup multivariate analysis of 101 nodules without lipiodol uptake defect (Table 4), indistinct tumor margin on cone-beam CT (HR = 9.10; 95% CI: 1.63, 50.8; p = 0.012) and heterogeneous lipiodol uptake in the tumor (HR = 2.86; 95% CI: 1.05, 4.42; p = 0.035) remained significant contributors to higher LTR rates. The incomplete safety margin on immediate CT failed to significantly (HR = 1.35; 95% CI: 0.54, 3.34; p = 0.518) affect the LTR rate.
The concept of a 'safety margin' in curative treatment of HCCs is based on the fact that microsatellite lesions are commonly observed in small HCCs. Sasaki et al. (19) reported that microscopic invasion of HCC cells into the portal vein was observed in 39% of tumors ≤ 5 cm, while microsatellite lesions were detected in 46% of tumors. In tumors smaller than 25 mm, microsatellite lesions were observed in seven (29.2%) of 24 tumors. All but one were located within 5 mm from the main tumor. For tumors between 25 mm and 50 mm, microsatellite lesions were observed in 39 (51.3%) of 76 tumors (19).
The role of the 'safety margin' has been intensely investigated in surgical resection or RFA for HCCs. Many researchers insist that an optimal safety margin is crucial for surgical resection to lower tumor recurrence (42021), whereas others have reported that a surgical safety margin does not influence tumor recurrence (522). Recently, an anatomical resection has been recommended for patients with durable hepatic function because intrahepatic metastasis is believed to arise from portal dissemination (2324). As establishing a safety margin around the ablated tumor is critical in achieving lower LTR in RFA, a 5-mm safety margin on a contrast-enhanced CT has been recommended (262526). However, the effect of a safety margin on LTR has been rarely investigated in TACE.
Very recently, it has been reported that a sufficient circumferential safety margin can significantly reduce LTR after superselective TACE (29.1% in complete safety margin vs. 66.7% in incomplete safety margin) (27). In that report, complete safety margin was achieved in 65.3% of digital subtraction angiography alone group while it was achieved in 87.2% of cone-beam CT assisted group. However, the results of our study are different from those of that previous study. The complete safety margin did not reduce LTR in our study. Complete safety margin was achieved in only 52 (40.6%) of 128 tumor nodules. For incomplete safety margin, the 2-year cumulative LTR rate was only 19.0% once the tumor was completely treated. It is difficult to explain the discrepancies between this study and the previous study (27). However, there are differences in the methodologies used in the two studies. In the previous study, the minimum safety margin for the treatment was defined as a 5-mm wide with persistent lipiodol uptake in normal liver parenchyma surrounding the tumor on CT taken one week later. In this study, a 1-mm thick non-contrast CT image was taken immediately after TACE for detailed evaluation of the embolized area and safety margin. A meticulous comparison between the axial images of the cone-beam CT hepatic arteriography and post-procedural non-contrast CT might have increased the cases of lipiodol uptake defect. In the previous study, newly developed HCC lesions ≤ 6 cm were selected for the study. However, HCC lesions were much bigger than in this study. Larger tumors might have a greater chance of LTR due to microsatellite lesions in the safety margin. However, it is certain that the presence of lipiodol uptake defect on lipiodol CT is the most important risk factor for LTR after TACE because it signifies incomplete treatment for target lesions (27). This study also showed that lipiodol uptake defect was the strongest risk factor for LTR (p < 0.001). Our study further identified ascites (p = 0.035), indistinct tumor margin on the cone-beam CT (p = 0.039) before TACE, and heterogeneous lipiodol uptake (p = 0.023) as independent determinant factors for higher LTR rate.
The presence of ascites is one of complications of portal hypertension due to advanced liver cirrhosis. A shrunken liver due to advanced liver cirrhosis can cause intrahepatic arterial tortuosity, making the superselection of tumor-feeding arteries difficult. Consequently, there is a greater chance of incomplete TACE in patients with ascites. In the subgroup analysis of 101 tumor nodules without lipiodol uptake defect, ascites was not a significant determinant factor for LTR.
The indistinct tumor margin and heterogeneous lipiodol uptake are mostly caused by tumor hypovascularity or hypovascular component in tumor nodules. According to multi-step hepatocarcinogenesis, early-stage HCCs have arterial hypovascularity with decreased but persistent portal venous supply (28). It has been known that TACE is only effective for hypervascular malignant foci supplied by arterial blood (29). Ultraselective TACE definitely can increase the therapeutic efficacy for early-stage HCCs (30). However, for early-stage HCCs with hypovascular component, the local tumor control rate over 15.8 months ± 11.8 (range 2-40 months) of follow-up was only 53.2% even after ultraselective TACE (30).
Subsegmental or ultraselective TACE becomes more feasible due to the development of cone-beam CT which enables us to perform TACE more selectively at far distal feeding arteries by providing detailed information about the embolized tumors (2731). However, more selective TACE makes it difficult to achieve a safety margin around the target lesion. In this study, the complete safety margin was achieved only in 52 (40.6%) of 128 tumor nodules after cone-beam CT-assisted superselective TACE. To achieve a complete safety margin, it is necessary to perform additional selective catheterization of the hepatic arteries that supply the liver parenchyma surrounding the tumor nodule without evidently supplying the tumor itself. This requires additional procedural time and radiation exposure to both operators and patients. In addition, more severe postembolization syndrome and damage to the surrounding normal parenchyma are inevitable. Therefore, the survival advantage of the attempt to achieve a complete safety margin in TACE is questionable. Based on the results of this study, it is unnecessary to achieve a complete safety margin if the tumor nodule is small and completely treated.
Our study has several limitations. First, this was a single-center retrospective study. There might have been unavoidable selection bias. Second, the pathologic grades of HCCs could not be evaluated because the diagnosis of HCC was based on the typical imaging findings without biopsy. Third, the imaging analysis was performed by two radiologists with a consensus opinion. However, inter-observer variability was not assessed. Fourth, the effect of safety margin on long-term survival was not assessed.
In conclusion, the safety margin in completely lipiodolized nodule without a defect would not affect LTR in cone-beam CT-assisted TACE for small nodular HCCs. However, a prospective study with a larger sample size is needed to draw a definite conclusion.
Figures and Tables
Fig. 1
Sustained complete tumor response after TACE in 60-year-old female with hepatocellular carcinoma.
A. Arterial-phase CT scan showing 1.5 cm-sized hypervascular tumor at segment 5 of right hepatic lobe (arrows). B. On cone-beam CT hepatic arteriography, hypervascular tumor nodule showed clear tumor margin (arrows). C. After subsegmental TACE, tumor showed homogenous lipiodol uptake in entire tumor nodule (arrows) on lipiodol CT. D. Three years later, TACE-treated nodule was shrunken in size without evidence of local tumor recurrence (arrowheads). TACE = transarterial chemoembolization
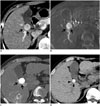
Fig. 2
Local tumor recurrence after TACE in 80-year-old female with hepatocellular carcinoma.
A. Arterial-phase CT scan showing 1.5 cm-sized hypervascular tumor at segment 8 of right hepatic lobe. Posterior aspect of tumor is relatively hypovascular (black arrows). B. On cone-beam CT hepatic arteriography, anterior hypervascular part of tumor showed strong contrast enhancement with clear tumor margin. In contrast, posterior hypovascular part showed subtle contrast enhancement with indistinct tumor margin (black arrows). C. After subsegmental TACE, tumor showed heterogeneous lipiodol uptake (dense lipiodol uptake in hypervascular component and faint lipiodol uptake in hypovascular component) (white arrow) on lipiodol CT. D. Three years later, arterial-phase CT scan showed enhanced recurrent tumor at area of hypovascular component (black arrowheads). Area of hypervascular component was shrunken in size with persistent lipiodol uptake (white arrowhead). TACE = transarterial chemoembolization

Fig. 3
Methods used to determine lipiodol uptake defect in tumor after TACE.
A, B. Non-contrast thin-section CT taken immediately after TACE (B) revealed complete lipiodol retention throughout hypervascular tumor at left hepatic lobe on cone-beam CT hepatic arteriography (A). C, D. Non-contrast thin-section CT after TACE (D) revealed focal lipiodol uptake defect (arrow in D) in hypervascular tumor at segment 4 upon cone-beam CT hepatic arteriography (C). TACE = transarterial chemoembolization
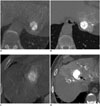
Fig. 4
Methods used to determine safety margin in liver parenchyma surrounding tumor after TACE.
A. On non-contrast thin-section CT taken immediately after TACE, wedge-shaped parenchymal lipiodol deposition completely encased entire surface of tumor, which was interpreted as 'complete safety margin' (black arrows). B. On non-contrast CT thin-section CT taken immediately after TACE, posteromedial aspect of tumor was not completely covered by parenchymal lipiodol deposition (white arrows), which was interpreted as 'incomplete safety margin'. TACE = transarterial chemoembolization
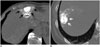
Fig. 5
Cumulative local tumor recurrence curve showing cumulative local tumor recurrence rate depending on presence or absence of intra-tumoral lipiodol uptake defect and safety margin.
In completely lipiodolized nodules (nodules without intra-tumoral lipiodol uptake defect), completeness of safety margin did not affect cumulative local recurrence rate (p = 0.912). SM = safety margin

Table 1
Patient Demographics
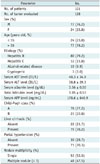
Table 2
Results of Univariate Analysis on Potential Determinant Factors for LTR after TACE

Table 3
Results of Multivariate Analysis on Potential Determinant Factors for LTR after TACE

Table 4
Results of Multivariate Analysis on Potential Determinant Factors for LTR after TACE in Subgroup of 101 Nodules without Lipiodol Uptake Defects

References
1. Portolani N, Coniglio A, Ghidoni S, Giovanelli M, Benetti A, Tiberio GA, et al. Early and late recurrence after liver resection for hepatocellular carcinoma: prognostic and therapeutic implications. Ann Surg. 2006; 243:229–235.
2. Zytoon AA, Ishii H, Murakami K, El-Kholy MR, Furuse J, El-Dorry A, et al. Recurrence-free survival after radiofrequency ablation of hepatocellular carcinoma. A registry report of the impact of risk factors on outcome. Jpn J Clin Oncol. 2007; 37:658–672.
3. Kim YS, Lim HK, Rhim H, Lee MW, Choi D, Lee WJ, et al. Ten-year outcomes of percutaneous radiofrequency ablation as first-line therapy of early hepatocellular carcinoma: analysis of prognostic factors. J Hepatol. 2013; 58:89–97.
4. Shi M, Guo RP, Lin XJ, Zhang YQ, Chen MS, Zhang CQ, et al. Partial hepatectomy with wide versus narrow resection margin for solitary hepatocellular carcinoma: a prospective randomized trial. Ann Surg. 2007; 245:36–43.
5. Tang YH, Wen TF, Chen X. Resection margin in hepatectomy for hepatocellular carcinoma: a systematic review. Hepatogastroenterology. 2012; 59:1393–1397.
6. Kim KW, Lee JM, Klotz E, Kim SJ, Kim SH, Kim JY, et al. Safety margin assessment after radiofrequency ablation of the liver using registration of preprocedure and postprocedure CT images. AJR Am J Roentgenol. 2011; 196:W565–W572.
7. Bruix J, Sherman M. American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology. 2011; 53:1020–1022.
8. Lee IJ, Chung JW, Yin YH, Kim HC, Kim YI, Jae HJ, et al. Cone-beam CT hepatic arteriography in chemoembolization for hepatocellular carcinoma: angiographic image quality and its determining factors. J Vasc Interv Radiol. 2014; 25:1369–1379. quiz 1379-1379.e1
9. Choi WS, Kim HC, Hur S, Choi JW, Lee JH, Yu SJ, et al. Role of C-arm CT in identifying caudate arteries supplying hepatocellular carcinoma. J Vasc Interv Radiol. 2014; 25:1380–1388.
10. Kim HC. Role of C-arm cone-beam CT in chemoembolization for hepatocellular carcinoma. Korean J Radiol. 2015; 16:114–124.
11. Song SY, Chung JW, Yin YH, Jae HJ, Kim HC, Jeon UB, et al. Celiac axis and common hepatic artery variations in 5002 patients: systematic analysis with spiral CT and DSA. Radiology. 2010; 255:278–288.
12. Miyayama S, Matsui O, Yamashiro M, Ryu Y, Kaito K, Ozaki K, et al. Ultraselective transcatheter arterial chemoembolization with a 2-f tip microcatheter for small hepatocellular carcinomas: relationship between local tumor recurrence and visualization of the portal vein with iodized oil. J Vasc Interv Radiol. 2007; 18:365–376.
13. Ito K, Mitchell DG, Siegelman ES. Cirrhosis: MR imaging features. Magn Reson Imaging Clin N Am. 2002; 10:75–92. vi
14. Gupta AA, Kim DC, Krinsky GA, Lee VS. CT and MRI of cirrhosis and its mimics. AJR Am J Roentgenol. 2004; 183:1595–1601.
15. Lafortune M, Matricardi L, Denys A, Favret M, Déry R, Pomier-Layrargues G. Segment 4 (the quadrate lobe): a barometer of cirrhotic liver disease at US. Radiology. 1998; 206:157–160.
16. de Franchis R. Baveno V Faculty. Revising consensus in portal hypertension: report of the Baveno V consensus workshop on methodology of diagnosis and therapy in portal hypertension. J Hepatol. 2010; 53:762–768.
17. de Franchis R, Primignani M. Natural history of portal hypertension in patients with cirrhosis. Clin Liver Dis. 2001; 5:645–663.
18. Annet L, Materne R, Danse E, Jamart J, Horsmans Y, Van Beers BE. Hepatic flow parameters measured with MR imaging and Doppler US: correlations with degree of cirrhosis and portal hypertension. Radiology. 2003; 229:409–414.
19. Sasaki A, Kai S, Iwashita Y, Hirano S, Ohta M, Kitano S. Microsatellite distribution and indication for locoregional therapy in small hepatocellular carcinoma. Cancer. 2005; 103:299–306.
20. Nonami T, Harada A, Kurokawa T, Nakao A, Takagi H. Hepatic resection for hepatocellular carcinoma. Am J Surg. 1997; 173:288–291.
21. Nagasue N, Uchida M, Makino Y, Takemoto Y, Yamanoi A, Hayashi T, et al. Incidence and factors associated with intrahepatic recurrence following resection of hepatocellular carcinoma. Gastroenterology. 1993; 105:488–494.
22. Poon RT, Fan ST, Ng IO, Wong J. Significance of resection margin in hepatectomy for hepatocellular carcinoma: a critical reappraisal. Ann Surg. 2000; 231:544–551.
23. Makuuchi M, Hasegawa H, Yamazaki S. Ultrasonically guided subsegmentectomy. Surg Gynecol Obstet. 1985; 161:346–350.
24. Arii S, Tanaka S, Mitsunori Y, Nakamura N, Kudo A, Noguchi N, et al. Surgical strategies for hepatocellular carcinoma with special reference to anatomical hepatic resection and intraoperative contrast-enhanced ultrasonography. Oncology. 2010; 78:Suppl 1. 125–130.
25. Goldberg SN, Grassi CJ, Cardella JF, Charboneau JW, Dodd GD 3rd, Dupuy DE, et al. Image-guided tumor ablation: standardization of terminology and reporting criteria. J Vasc Interv Radiol. 2009; 20:7 Suppl. S377–S390.
26. Fujioka C, Horiguchi J, Ishifuro M, Kakizawa H, Kiguchi M, Matsuura N, et al. A feasibility study: evaluation of radiofrequency ablation therapy to hepatocellular carcinoma using image registration of preoperative and postoperative CT. Acad Radiol. 2006; 13:986–994.
27. Miyayama S, Yamashiro M, Hashimoto M, Hashimoto N, Ikuno M, Okumura K, et al. Comparison of local control in transcatheter arterial chemoembolization of hepatocellular carcinoma ≤6 cm with or without intraprocedural monitoring of the embolized area using cone-beam computed tomography. Cardiovasc Intervent Radiol. 2014; 37:388–395.
28. Matsui O, Kadoya M, Kameyama T, Yoshikawa J, Takashima T, Nakanuma Y, et al. Benign and malignant nodules in cirrhotic livers: distinction based on blood supply. Radiology. 1991; 178:493–497.
29. Takayasu K, Wakao F, Moriyama N, Muramatsu Y, Sakamoto M, Hirohashi S, et al. Response of early-stage hepatocellular carcinoma and borderline lesions to therapeutic arterial embolization. AJR Am J Roentgenol. 1993; 160:301–306.
30. Miyayama S, Matsui O, Yamashiro M, Ryu Y, Takata H, Takeda T, et al. Iodized oil accumulation in the hypovascular tumor portion of early-stage hepatocellular carcinoma after ultraselective transcatheter arterial chemoembolization. Hepatol Int. 2007; 1:451–459.
31. Wallace MJ, Murthy R, Kamat PP, Moore T, Rao SH, Ensor J, et al. Impact of C-arm CT on hepatic arterial interventions for hepatic malignancies. J Vasc Interv Radiol. 2007; 18:1500–1507.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download