Abstract
Objective
To assess patient radiation doses during cerebral angiography and embolization of intracranial aneurysms in a large sample size from a single center.
Materials and Methods
We studied a sample of 439 diagnostic and 149 therapeutic procedures for intracranial aneurysms in 480 patients (331 females, 149 males; median age, 57 years; range, 21-88 years), which were performed in 2012 with a biplane unit. Parameters including fluoroscopic time, dose-area product (DAP), and total angiographic image frames were obtained and analyzed.
Results
Mean fluoroscopic time, total mean DAP, and total image frames were 12.6 minutes, 136.6 ± 44.8 Gy-cm2, and 251 ± 49 frames for diagnostic procedures, 52.9 minutes, 226.0 ± 129.2 Gy-cm2, and 241 frames for therapeutic procedures, and 52.2 minutes, 334.5 ± 184.6 Gy-cm2, and 408 frames for when both procedures were performed during the same session. The third quartiles for diagnostic reference levels (DRLs) were 14.0, 61.1, and 66.1 minutes for fluoroscopy time, 154.2, 272.8, and 393.8 Gy-cm2 for DAP, and 272, 276, and 535 for numbers of image frames in diagnostic, therapeutic, and both procedures in the same session, respectively. The proportions of fluoroscopy in DAP for the procedures were 11.4%, 50.5%, and 36.1%, respectively, for the three groups. The mean DAP for each 3-dimensional rotational angiographic acquisition was 19.2 ± 3.2 Gy-cm2. On average, rotational angiography was used 1.4 ± 0.6 times/session (range, 1-4; n = 580).
Conclusion
Radiation dose in our study as measured by DAP, fluoroscopy time and image frames did not differ significantly from other reported DRL studies for cerebral angiography, and DAP was lower with fewer angiographic image frames for embolization. A national registry of radiation-dose data is a necessary next step to refine the dose reference level.
Interventional radiological procedures have the potential to expose a patient to high doses of radiation, and, in particular, a high entrance surface dose of up to several Gy. There have been many studies investigating patient doses from radiation exposure, especially in cardiology and general interventional radiology (1, 2, 3).
As radiation effects can be categorized as stochastic and deterministic effects, surface skin dose is important because of the potential for well-documented deterministic effects, such as skin erythema, necrosis, and even ulceration, with high doses. Direct measurement is desirable, but not typically practical because thermoluminescent dosimeters (TLD) are cumbersome with limitations in terms of practical use (4, 5). An alternative is an indirect estimate of the radiation dose using a dose-area product (DAP) meter, which is common in modern angiographic systems.
While a single measured DAP does not provide dose optimization guidelines for balancing the medical benefit and risk of injury, a dose reference level (DRL) can give information regarding current practice in a hospital with respect to patient dose. DRLs are exposure levels not expected to be exceeded during normal diagnostic and technical performance of procedures typically set as the 75th percentile. Commonly used parameters for a DRL are DAP, number of exposures, and fluoroscopy time.
Previous studies in other countries have proposed and investigated DRL levels, and reported large variation within hospitals using different DRL guidelines (6, 7). There is at present no published study of DRLs specific to Korea regarding neurointerventional procedures. Thus, a national reference DRL would improve the standard of care.
The goal of the present study was to obtain baseline data for a proposed local guideline for diagnostic and therapeutic DRLs in Korea by retrospective evaluation of patient exposure at a major academic center over a 1-year period. As the radiation doses show marked variation due to pathology and type of procedure, a more standardized examination protocol was focused on and selected for analysis. Thus, this study was limited to diagnostic cerebral angiographies and therapeutic embolizations of intracranial aneurysms.
The study was approved by our Institutional Review Board. A waiver of the need for consent was obtained for this Health Insurance Portability and Accountability Act-compliant retrospective study.
From the angiographic database, in total, 739 diagnostic cerebral angiographies and 196 neurointerventional procedures were identified from the period January to December, 2012. All procedures were performed by a team of two experienced neurointerventional radiologists. A clinical fellow with experience of over 700 previous cases performed the diagnostic angiographic procedures while embolizations were performed in conjunction with a radiologist with over 19 years of experience regarding aneurysm embolizations. Diagnostic procedures performed for follow-up after clipping or coiling of an aneurysm were excluded. In total, 480 patients had intracranial aneurysms (331 females, 149 males). The median age was 57 (21-88) years. There were, in total, 439 diagnostic procedures and 149 therapeutic procedures. In 38 patients, diagnostic and therapeutic interventions were performed during the same session.
The angiographic system used was a biplane angiographic unit (Axiom Artis dBA, Siemens Healthcare, Erlangen, Germany) with a flat panel detector: A and B planes (48 cm) with variable fields of view of 42-32-22-16 cm. The total filtration was 2.5 (mmAl). The angiography unit had three pulsed fluoroscopy modes, of 10, 15, and 30 P/s, of which 15 P/s was used most frequently. The system included a DAP meter. Variables collected were DAP (Gy-cm2), fluoroscopy exposure time, and number of angiographic image acquisitions. As collected radiation doses will show a skewed distribution with extreme values and a long upper tail, a diagnostic reference level at the 75th percentile is appropriate. So with the acquired set of our data, the third quartiles were calculated and proposed as DRLs for our institution. Data were collected separately for AP and lateral views but were added together and compared for analysis. Pearson's r was used for statistical analysis regarding whether there was a significant correlation among DAP, fluoroscopy time, and number of images. In addition, statistical correlations between the radiation doses and patient age were also evaluated.
The results, including the third quartiles, are presented in Table 1 and Figure 1. Mean fluoroscopic time, total mean DAP (± standard deviation), and total angiographic image frames were 12.6 minutes, 136.6 ± 44.8 Gy-cm2, and 251 ± 49 frames for diagnostic procedures, 52.9 minutes, 226.0 ± 129.2 Gy-cm2, and 241 frames for therapeutic procedures, and 52.2 minutes, 334.5 ± 184.6 Gy-cm2, and 408 frames when both procedures were performed during the same session. The third quartiles, which may be set as a DRL, were 14.0, 61.1, and 66.1 minutes for fluoroscopy times, 154.2, 272.8, and 184.6 Gy-cm2 for DAP, and 272, 276, and 535 for number of image frames in diagnostic, therapeutic, and both procedures during the same session, respectively.
The proportion of fluoroscopic DAP in the procedure was 11.4% for diagnostic only, 50.5% for therapeutic only, and 36.1% for both during the same session. The amount of time spent using fluoroscopy was lower for diagnostic applications than therapeutic interventions. The overall mean DAP was 0.376 ± 0.125 Gy-cm2 per image frame, and the mean fluoroscopic DAP was 1.467 ± 0.623 Gy-cm2 per minutes.
The mean DAP for each three-dimensional rotational angiographic acquisition was 19.2 ± 3.2 Gy-cm2. On average, rotational angiography was used 1.4 ± 0.6 times/session (range, 1-4; n = 580). In the statistical analysis, the only significant correlation was for diagnostic angiography, in which fluoroscopic time was correlated positively with patient age (r = 0.544, p < 0.01).
Dose-area product can be used as an indicator of radiation exposure levels, from which medical follow-up for possible radiation injuries due to the skin exposure can be suggested (8). The DAP meter uses a transmission-type air ionization chamber, mounted on the X-ray tube collimator; DAP is a function of the cross section of an X-ray beam and the X-ray exposure at the collimator (9) and the unit is Gy-cm2. Furthermore, using known estimated conversion factors from the literature (10), an estimated effective dose can be obtained. The precise estimation of local skin dose using DAP is difficult because the dose rates and field size changes differ among measured DAPs, but a correlation between high DAP and skin dose has been reported (5). Thus, DAP is more practical as the entire examination is conducted without effects due to beam position, in contrast to TLD. In addition, its use and monitoring are possible during and after procedures.
A recent study by D'Ercole et al. (7) proposed a local DRL regarding diagnostic and cerebral embolizations and compared these levels with other recently published data. Other more comprehensive studies have proposed DRLs for numerous radiological procedures; the data regarding cerebral interventions are summarized in Tables 2 and 3 (6, 7, 11, 12, 13). DRLs cannot be used individually on a per-patient basis and are not a restrictive threshold, but do provide a practical guideline for monitoring and comparing radiation doses among procedures and institutions. With 588 total cases our study is the largest to date regarding cerebral aneurysms for a proposed DRL. This may increase the validity of the third quartile values, which may be set as DRLs. They were 14.0, 61.1, and 66.1 minutes for fluoroscopy times, 154.2, 272.8, and 393.8 Gy-cm2 for DAP, and 272, 276, and 535 for numbers of image frames for diagnostic, therapeutic, and both procedures during the same session, respectively. Regarding angiographic procedures associated with aneurysms, our DRLs are comparable but with fewer angiographic image acquisitions. Regarding embolization procedures, our data showed comparatively lower DAPs, longer fluoroscopy times, and fewer angiographic image frames.
We categorized our patient data into a separate group when diagnostic cerebral angiographies and embolizations were performed during the same session. This is the only published report with such reported separated data; it is possible that previous studies may have included these cases in the embolization group which might have slightly increased the reported DRL regarding embolization procedures. In the embolization group, the DRL for number of angiographic image frames in our study was significantly lower than previously published studies. This may be due to inclusion of cases with simultaneous diagnostic and therapeutic procedures into embolization cases, which may have contributed to the increased DRL in other reports.
An appropriate uniform dose index for use in all types of radiological examinations is difficult because some partial-body irradiation is inevitable (14). There is a lack of standardization of radiation dose measurements; DAP, effective dose, skin dose, and air kerma are all used and dependent on the monitoring equipment available. Although DAP measurements cannot be used to evaluate individual patient risk, they may be used to estimate stochastic and deterministic effects by estimating the effective dose and skin dose. Although some consider effective dose to be inappropriate for use in epidemiological studies of radiation risk and assessment of the likelihood of the occurrence and severity of deterministic effects (15), DAP values, as in proposed DRLs, are valuable in estimating radiation exposure and in comparing local DAP values with those at other facilities. Similarly, periodic comparisons of exposure parameters, such as DAP, fluoroscopy time, and numbers of angiographic frames, with newer equipment and protocols and periodic radiation monitoring may be possible.
Bogaert et al. (8) suggested cumulative DAP as an indicator of maximum skin dose. The probability of a maximum skin dose over 2 Gy-cm2 is about 30% when DAP is > 125 Gy-cm2, and of a dose over 3 Gy-cm2 is 60% when DAP is > 250 Gy-cm2. In our study, the maximum DAP was 792 Gy-cm2, indicating that this type of exposure does occur in practice. Struelens et al. (3) proposed a trigger level of 220-330 Gy-cm2 as a threshold for deterministic skin effects (2 Gy). We agree in that we should be careful about drawing definite conclusions. Aroua et al. (6) reported that the associated DAP can exceed 1300 Gy-cm2 in specific circumstances. In these high-exposure situations, a recommendation for clinical follow-up of potential deterministic effects would be of value. A wide spectrum of radiation dose is more common with cerebral angiographic studies versus other angiographic studies due to the complex nature of the procedures. Thus, it would be difficult to determine a single internationally accepted standard reference dose level.
Regarding the diagnosis and treatment of aneurysms, our results showed that rotational angiography was used 1.4 ± 0.6 times/session (range, 1-4; n = 580), and the average DAP for rotational angiography alone was 19.2 ± 3.2 Gy-cm2. In the diagnosis and therapeutic interventions for cerebral aneurysms, rotational angiography facilitates evaluation of the potential for endovascular treatment, permits finding a good working view, aids in performing accurate measurements, depicts additional aneurysms during an initial evaluation, enables accurate determination of stent positions during stent-assisted coiling, evaluates subarachnoid hemorrhage in the angiographic suite during the therapeutic procedure, determines status after clipping/coiling, and provides baseline data for long-term follow-up (16, 17, 18, 19, 20). Our study showed the DAP in rotational angiography to be similar to that during acquisition of antero-posterior and lateral angiography for a single vessel.
This is the first study to report independent data for diagnostic, therapeutic, and combined situations. Analysis of the dose distribution with regard to fluoroscopy time and image frames may enable a reduction in overall patient radiation doses. For diagnostic cerebral angiography, the fluoroscopy time was shorter than that for therapeutic interventions because fluoroscopy was used mostly for catheter placement, resulting in a lower contribution to total radiation dose. The fluoroscopic radiation dose in therapeutic procedures was considerably higher than that in diagnostic angiography (11.4% vs. 50.5%), likely due to the complexity of aneurysm coil embolizations. To decrease total radiation exposure in cerebral embolization cases, decreasing the fluoroscopic time would be an effective method, because it is responsible for a larger amount of radiation compared with diagnostic cerebral angiography. For diagnostic angiography, decreasing the number of images and the radiation dose parameters per exposure may be more effective. Combining diagnostic and therapeutic procedures in the same session decreased DAP and fluoroscopic time. Thus, if a therapeutic option with an advantageous risk/benefit ratio is considered and appropriate informed patient consent is obtained, use of a combined approach can decrease the radiation dose.
The main limitation of this study was the use of one angiographic unit from a single institution to determine the DRL level. According to Marshall et al. (21), a national DRL would require a minimum of 20 centers with 10 patients at each, for a total minimum of 200 patients. Against this, our study included data from > 500 procedures. Moreover, we believe these results are of value as a preliminary study in terms of providing baseline data and methodology for determining a preliminary DRL specific to Korea. This may facilitate further dose-reduction protocols and radiation-exposure monitoring. If, unexpectedly, increased radiation doses are detected by periodic monitoring, investigation of causative factors may be warranted. In addition there is continuous development of novel radiation reduction technologies such as new image processing technology with combined temporal and spatial noise reduction filters with automatic pixel shift functionality (22). A proposed DRL would be of value in comparing radiation dose. Further studies at a national level with additional institutions and involving other radiological procedures would result in a more comprehensive national DRL. In addition, another limitation is the small number of operators, as radiation exposure distribution regarding neurointerventional procedures are highly operator dependent.
In conclusion, radiation dose as measured by DAP, fluoroscopy time and image frames did not differ significantly from other reported DRL studies regarding cerebral angiography, and DAP was lower with fewer angiographic image frames regarding aneurysm embolization. This work represents one of the largest studies of patient radiation dose during cerebral angiography and embolization of intracranial aneurysm, and permits a baseline data for further monitoring at our institution after future implementation of a dose-reduction protocol, inter-institutional comparison for quality control, and initial proposal of values for a reference level in Korea. A national registry of radiation-dose data is a necessary next step to refine the reference level.
Figures and Tables
Fig. 1
Chart showing mean of fluoroscopy time, total dose-area product (DAP), and number of image frames among three groups with standard deviation.

Table 1
Mean and Third Quartile Data for Fluoroscopic Time, DAP, and Image Frames for Diagnostic, Therapeutic Procedures, and Both Performed during Same Session
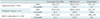
Table 2
Review of Mean and Third Quartile Data of DAP, Fluoroscopic Time, and Number of Image Frames Acquired during Diagnostic Cerebral Angiography for Intracranial Aneurysms

| Reference | No. of Patients | DAP (Gy-cm2) | Fluoroscopy Time (Min) | Number of Frames | |||
|---|---|---|---|---|---|---|---|
| Mean | 3rd Quartile | Mean | 3rd Quartile | Mean | 3rd Quartile | ||
| This study | 439 | 136.6 | 154.2 | 12.6 | 14.0 | 251 | 273 |
| D'Ercole et al. (7) | 100 | 142.1 | 180 | 9.9 | 12.3 | 220 | 317 |
| Aroua et al. (6) | 91 | 121 | 125 | 12.6 | 15 | 679 | 480 |
| Verdun et al. (13) | 91 | 107 | 124 | ||||
| Brambilla et al. (11) | 188 | 158 | 198 | 13.7 | 17.5 | ||
Table 3
Review of Mean and Third Quartile Data of DAP, Fluoroscopic Time, and Number of Image Frames Acquired during Endovascular Coil Embolization of Intracranial Aneurysms

| Reference | No. of Patients | DAP (Gy-cm2) | Fluoroscopy Time (Min) | Number of Frames | |||
|---|---|---|---|---|---|---|---|
| Mean | 3rd Quartile | Mean | 3rd Quartile | Mean | 3rd Quartile | ||
| This study | 111 | 226.0 | 272.8 | 52.9 | 61.1 | 241 | 276 |
| D'Ercole et al. (7) | 72 | 382.2 | 487 | 37.2 | 46.3 | 558 | 717 |
| Aroua et al. (6) | 52 | 335 | 440 | 36.5 | 50 | 760 | 800 |
| Verdun et al. (13) | 58 | 335 | 352 | ||||
| Miller et al. (12) | 356 | 319.9 | 87.1 | 1053 | |||
References
1. Bor D, Cekirge S, Türkay T, Turan O, Gülay M, Onal E, et al. Patient and staff doses in interventional neuroradiology. Radiat Prot Dosimetry. 2005; 117:62–68.
2. McParland BJ. A study of patient radiation doses in interventional radiological procedures. Br J Radiol. 1998; 71:175–185.
3. Struelens L, Vanhavere F, Bosmans H, Van Loon R, Mol H. Skin dose measurements on patients for diagnostic and interventional neuroradiology: a multicentre study. Radiat Prot Dosimetry. 2005; 114:143–146.
4. Mahesh M. Fluoroscopy: patient radiation exposure issues. Radiographics. 2001; 21:1033–1045.
5. Suzuki S, Furui S, Matsumaru Y, Nobuyuki S, Ebara M, Abe T, et al. Patient skin dose during neuroembolization by multiple-point measurement using a radiosensitive indicator. AJNR Am J Neuroradiol. 2008; 29:1076–1081.
6. Aroua A, Rickli H, Stauffer JC, Schnyder P, Trueb PR, Valley JF, et al. How to set up and apply reference levels in fluoroscopy at a national level. Eur Radiol. 2007; 17:1621–1633.
7. D'Ercole L, Thyrion FZ, Bocchiola M, Mantovani L, Klersy C. Proposed local diagnostic reference levels in angiography and interventional neuroradiology and a preliminary analysis according to the complexity of the procedures. Phys Med. 2012; 28:61–70.
8. Bogaert E, Bacher K, Lemmens K, Carlier M, Desmet W, De Wagter X, et al. A large-scale multicentre study of patient skin doses in interventional cardiology: dose-area product action levels and dose reference levels. Br J Radiol. 2009; 82:303–312.
9. Nickoloff EL, Lu ZF, Dutta AK, So JC. Radiation dose descriptors: BERT, COD, DAP, and other strange creatures. Radiographics. 2008; 28:1439–1450.
10. Bor D, Sancak T, Olgar T, Elcim Y, Adanali A, Sanlidilek U, et al. Comparison of effective doses obtained from dose-area product and air kerma measurements in interventional radiology. Br J Radiol. 2004; 77:315–322.
11. Brambilla M, Marano G, Dominietto M, Cotroneo AR, Carriero A. Patient radiation doses and references levels in interventional radiology. Radiol Med. 2004; 107:408–418.
12. Miller DL, Balter S, Cole PE, Lu HT, Schueler BA, Geisinger M, et al. Radiation doses in interventional radiology procedures: the RAD-IR study: part I: overall measures of dose. J Vasc Interv Radiol. 2003; 14:711–727.
13. Verdun FR, Aroua A, Trueb PR, Vock P, Valley JF. Diagnostic and interventional radiology: a strategy to introduce reference dose level taking into account the national practice. Radiat Prot Dosimetry. 2005; 114:188–191.
14. Le Heron JC. Estimation of effective dose to the patient during medical x-ray examinations from measurements of the dose-area product. Phys Med Biol. 1992; 37:2117–2126.
15. Harrison JD, Streffer C. The ICRP protection quantities, equivalent and effective dose: their basis and application. Radiat Prot Dosimetry. 2007; 127:12–18.
16. Anxionnat R, Bracard S, Ducrocq X, Trousset Y, Launay L, Kerrien E, et al. Intracranial aneurysms: clinical value of 3D digital subtraction angiography in the therapeutic decision and endovascular treatment. Radiology. 2001; 218:799–808.
17. Doelken M, Struffert T, Richter G, Engelhorn T, Nimsky C, Ganslandt O, et al. Flat-panel detector volumetric CT for visualization of subarachnoid hemorrhage and ventricles: preliminary results compared to conventional CT. Neuroradiology. 2008; 50:517–523.
18. Kang HS, Han MH, Kwon BJ, Jung SI, Oh CW, Han DH, et al. Postoperative 3D angiography in intracranial aneurysms. AJNR Am J Neuroradiol. 2004; 25:1463–1469.
19. Richter G, Engelhorn T, Struffert T, Doelken M, Ganslandt O, Hornegger J, et al. Flat panel detector angiographic CT for stent-assisted coil embolization of broad-based cerebral aneurysms. AJNR Am J Neuroradiol. 2007; 28:1902–1908.
20. van Rooij WJ, Sprengers ME, de Gast AN, Peluso JP, Sluzewski M. 3D rotational angiography: the new gold standard in the detection of additional intracranial aneurysms. AJNR Am J Neuroradiol. 2008; 29:976–979.
21. Marshall NW, Chapple CL, Kotre CJ. Diagnostic reference levels in interventional radiology. Phys Med Biol. 2000; 45:3833–3846.
22. Dekker LR, van der Voort PH, Simmers TA, Verbeek XA, Bullens RW, Veer MV, et al. New image processing and noise reduction technology allows reduction of radiation exposure in complex electrophysiologic interventions while maintaining optimal image quality: a randomized clinical trial. Heart Rhythm. 2013; 10:1678–1682.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download