Abstract
Objective
To investigate the feasibility of a rat model on hindlimb ischemia induced by embolization from the administration of polyvinyl alcohol (PVA) particles or N-butyl cyanoacrylate (NBCA).
Materials and Methods
Unilateral hindlimb ischemia was induced by embolization with NBCA (n = 4), PVA (n = 4) or surgical excision (n = 4) in a total of 12 Sprague-Dawley rats. On days 0, 7 and 14, the time-of-flight magnetic resonance angiography (TOF-MRA) and enhanced MRI were obtained as scheduled by using a 3T-MR scanner. The clinical ischemic index, volume change and degree of muscle necrosis observed on the enhanced MRI in the ischemic hindlimb were being compared among three groups using the analysis of variance. Vascular patency on TOF-MRA was evaluated and correlated with angiographic findings when using an inter-rater agreement test.
Results
There was a technical success rate of 100% for both the embolization and surgery groups. The clinical ischemic index did not significantly differ. On day 7, the ratios of the muscular infarctions were 0.436, 0.173 and 0 at thigh levels and 0.503, 0.337 and 0 at calf levels for the NBCA, PVA and surgery groups, respectively. In addition, the embolization group presented increased volume and then decreased volume on days 7 and 14, respectively. The surgery group presented a gradual volume decrease. Good correlation was shown between the TOF-MRA and angiographic findings (kappa value of 0.795).
Peripheral arterial occlusive disease has increased in prevalence and has become a major cause of mortality and morbidity rates (1-3). Numerous studies have been performed to understand both the mechanisms of the underlying angiogenesis and arteriogenesis, and how to enhance these mechanisms in ischemic limbs during recent decades (4-6), and intensive additional efforts are expected, especially given the introduction of stem cells and gene therapies (7-9).
To translate the results from animal studies into humans, a proper experimental model is essential. The most widely used hindlimb ischemia models involve surgical ligation or excision, which is simple, low cost and practical for small animals. However, the surgical method generates local tissues inflammation that can confound data and the analysis of cytokines mediating angiogenesis (10, 11). Recently, Zhuang et al. (11) reported a successful minimally invasive technique using hydrogel coil embolization. This minimally invasive method has great advantages in preserving tissue integrity and minimizing inflammation, but is potentially limited by susceptibility artifacts from the coil during magnetic resonance imaging (MRI).
MRI is an excellent in vivo imaging modality that can obtain information from deep organs with good tissues contrast and relatively high spatial resolutions in a noninvasive manner (12-16). Although the laser Doppler perfusion imaging (LDPI) is the standard noninvasive method for evaluating hindlimb perfusion in small experimental models, the MRI can also be used to evaluate tissues perfusion with MR perfusion (12-14) as well as morphologic changes which include necrosis and atrophy with conventional MR (15), metabolites with MR spectroscopy (15) and vascular status with MR angiography (15, 16).
N-butyl cyanoacrylate (NBCA) and polyvinyl alcohol (PVA) particles are readily available embolic materials that are clinically used and do not cause susceptibility artifacts, unlike coils. Thus, these materials are potentially more useful in experimental studies of hindlimb ischemia. Therefore, the aim of this study was to investigate the feasibility of a hindlimb ischemia model induced via embolization with NBCA and PVA particles.
The study was approved by the institutional animal care and use committee.
Twelve male Sprague-Dawley (SD) rats weighing 350 g to 400 g with 10-12 weeks of age, were used for all experiments. For the procedures, the rats were anesthetized with an intraperitoneal injection in a mixture of zolazepam (5 mg/kg, Zoletil®; Virbac, Carros Cedex, France) and xylazine (3 mg/kg, Rompun®; Bayer-Schering Pharma, Berlin, Germany).
The rats were divided into three study groups based on the method of inducing hindlimb ischemia: embolization with NBCA (NBCA group, n = 4), embolization with PVA particles (PVA group, n = 4) and surgical method (surgery group, n = 4). Ischemia was induced in the left limb, and the right limb served as an internal control for each rat. Two rats from each group were sacrificed on days 7 and 14, respectively. Before their sacrifice, angiography was performed. For NBCA and PVA groups, the time-of-flight magnetic resonance angiography (TOF-MRA) and enhanced MR were performed on days 0 and 7 for all rats, and on day 14 for the remaining half. For surgery group, the MRI was performed on days 7 and 14 for each two rats which were scheduled for sacrifice, because of the movement barriers between Specific Pathogen Free space and MRI room of conventional space.
The embolization procedure was performed by an interventional radiologist. After anesthesia, with the assistance of a binocular loupe, a 2-cm longitudinal left paramedian incision was made in the neck. The left common carotid artery was exposed, and the distal common carotid artery was ligated using 4-0 Mersilk (Ethicon, Norderstedt, Germany), and a double-loop sling was formed around the proximal common carotid artery to control bleeding around the catheter. An 18-gauge intravenous catheter (BD angiocath Plus, Becton Dickinson Medical, Singapore) was inserted into the left common carotid artery, and a 1.7-Fr microcatheter (SL-10, Boston Scientific, Natick, MA, USA) and a guidewire (Transend, Boston Scientific) was inserted through the cannula to the aorta under fluoroscopic guidance (AXIOM Artis dTA/VB30; Siemens, Erlangen, Germany). An angiogram was produced with the use of 0.5 mL iodixanol (Visipaque 370, GE healthcare, Princeton, NJ, USA).
The target for embolization was the left external iliac artery and more distal arteries, including the femoral artery, saphenous artery and popliteal artery. Embolic materials were administered via microcatheters placed between the left internal iliac artery and superficial caudal epigastric artery. In the NBCA and PVA groups, embolization was performed with NBCA (Histoacryl; B. Braun Melsungen AG, Melsungen, Germany) mixed with iodized oil (Lipiodol; Andre Guerbet, Aulnay-Sous-Bois, France), in a ratio of 1 : 3 and 150-250 µm PVA particles (Contour, Boston Scientific Natick, MA, USA), respectively. Before injection of the NBCA mixture, the microcatheter was flushed with a 5% dextrose solution to prevent premature polymerization of the mixture in contact with residual blood or saline. After embolization, angiography was performed with catheter placed at the lower aorta level. The technical success was defined as the nonvisualization of the target vessels on postembolic arteriography, without compromise in the ipsilateral internal iliac artery and contralateral limb vessels.
Surgical procedure was performed by the other radiologists of 2-year experiences in hindlimb modeling. A 2-cm longitudinal incision was made in the left inguinal region. The left external iliac artery was dissected from the nerve and vein, and ligated twice distally at a point just proximal to the superficial caudal epigastric artery and proximally at a point 1-cm from the distal ligation site with 4-0 silk. The external iliac artery was transected between the two ligatures, and the incision was closed with Vicryl (Ethicon, Norderstedt, Germany) sutures.
All rats were observed at 1 week after induction of hindlimb ischemia for a clinical assessment of ischemia based on a previously published ischemic index (17). The animals were scored according to the following ischemic grading scale: 0, normal; 1, pale; 2, pressure sore; and 3, necrosis.
To evaluate the status of the arterial occlusion in a noninvasive manner, the TOF-MRA was performed. A 3 tesla clinical MR scanner (MAGNETOM Trio, Siemens Healthcare Solutions, Erlangen, Germany) was used with a tailored coil for small animals. The anesthetized rats were fixed in a supine position by an in-house-created holder. The MRA scan was performed with a two-dimensional TOF sequence technique (TR = 21 ms, TE = 5.1 ms, FA = 55°, slice thickness = 1.5 mm, slice overlap = 0.5 mm, FOV = 142 × 85 mm, matrix = 256 × 92, NEX = 4, and scanning time = 9 minutes 5 seconds). To suppress venous signals, a 20-mm thick slice at the caudal side of the measured slice was saturated.
Each common iliac, internal iliac, external iliac, femoral, saphenous, and popliteal arteries of the affected hindlimb was visually assessed in TOF-MRAs and conventional angiograms. A total of 120 segments were evaluated, which are of 6 arterial segments in 20 datasets obtained from 8 rats of NBCA and PVA groups on day 0, and 6 rats of three groups on days 7 and 14, respectively.
For dynamic contrast enhancement, a three-dimensional radial gradient-echo technique with a k-space-weighted image contrast sequence was used. For T1 mapping, three sets of T1-weighted images with different flip angles were obtained. A dynamic acquisition on a series of 30 axial images was started with a baseline scan (TR = 5.1 ms, TE = 2.0 ms, flip angle = 11°, FOV = 100 × 100 mm, slice thickness = 2.5 mm, interslice gap = 0, NEX = 2, matrix 128 × 128). During this dynamic acquisition, a bolus of gadoterate meglumine (Dotarem®, Guerbet, Paris, France) was injected through the tail vein. A total of 60 image sets were collected in approximately 5 minutes 30 seconds.
To compare the infarcted areas, the regions of interest (ROIs) were drawn along the non-enhancing areas suggesting infarction and whole muscle of the left thigh and calf on the representative single axial image of the enhanced MR collected on day 7, which showed the largest infarction. The ratio of the infarction was presented as the number of the infarcted area divided by whole muscle area (ratio of the infarction = infarcted area / whole muscle area). To measure volume change, the ROIs were drawn along the ischemic left calf and normal right calf on the representative single axial scan showing the largest outer diameter. The ratios were calculated and compared among the three groups. The volume change was calculated as the division of the sectional area in left affected limb by the right normal limb.
The rats were sacrificed, and the calf muscles from the left and right sides were harvested. After paraffin embedment, 4-µm thick sections were cut from each sample with muscle fibers oriented in the transverse direction and stained with hematoxylin and eosin. The tissue responses to different methods of hindlimb ischemia were assessed.
The results are expressed as the mean ± standard error of the mean. Statistical significance was determined by using the Kruskal-Wallis test for comparison among the three groups (significance level, p < 0.05) using GraphPad InStat (GraphPad Software, Lajolla, CA, USA). To evaluate the correlation between the clinical index and the infarcted area, Spearman's rho test was used (significance level, p < 0.05). To evaluate the concordances between the angiograms and TOF-MRA results, the inter-rater agreement test was used. Kappa values < 0.20 were considered to reflect slight agreements; 0.21 to 0.40, fair; 0.41 to 0.60, moderate; 0.61 to 0.80, substantial; and > 0.80, almost perfect agreement. MedCalc version 12.3.0.0 (MedCalc Software, Mariakerke, Belgium) was used for the correlation and concordance evaluation.
The technical success rate was 100% for both the embolization group and the surgery group. One rat suffered a hematoma at the left carotid artery ligation site but survived.
This result is summarized in Table 1.
The clinical ischemic index was 2 ± 0.7, 1.75 ± 0.75, and 1.75 ± 0.75 for the NBCA, PVA and surgery groups, respectively. There was no significant difference among (p = 0.983) the groups (Fig. 1).
The infarct ratio was 0.436 ± 0.052 and 0.173 ± 0.081 at thigh level and 0.503 ± 0.106 and 0.337 ± 0.073 at calf level for the NBCA and PVA groups, respectively. In the surgery group, no infarcted muscle was noted in the thigh or calf on the enhanced MR. The ratio of the muscle infarction was significantly different among three groups at both of the thigh (p = 0.0082) and calf (p = 0.0182). On post-hoc test, the significant difference was only shown in the comparison between NBCA and surgery group in both thigh (p < 0.01) and calf (p < 0.05) levels (Fig. 2). There were no correlations between the clinical ischemic index and the ratio of the infarcted area in the calf (ρ = -0.197, p = 0.9569), and that in the thigh (ρ = 0.138, p = 0.7036).
The NBCA and PVA groups presented volume increases on day 7 (1.060 ± 0.008 and 1.079 ± 0.050, respectively) and decreases on day 14 (0.848 ± 0.075 and 0.816 ± 0.015, respectively). However, a gradual decrease was observed in the surgery group (0.821 ± 0.049 to 0.739 ± 0.135) (Fig. 3).
Time-of-flight magnetic resonance angiography indicated a signal void, indicating the absence of antegrade flow, distal to the left external iliac or femoral artery (Fig. 4). The number of visible and invisible segments in the left ischemic limb on both the TOF-MRAs and angiograms were 44 and 64 out of 120 total segments, respectively. Eight segments were only visible on angiograms, and only four on TOF-MRAs. There was good correlation, with a kappa value of 0.795 (95% confidence interval: 0.685 to 0.904).
The hematoxylin and eosin stainings revealed muscle infarctions and marked infiltrations of inflammatory leukocytes around the infarcted areas in the embolization group. In the NBCA group, the small arteries were abnormally dilated, the infarcted areas and normal tissues were more clearly divided. However, in the PVA group, there was a wider transition zone indicating ischemic changes than in the NBCA group and no definite disorganization in the small arteries. In the surgery group, there were no infarcted muscles, but small-sized muscle fibers were scattered through the calf muscles, which are indicatives of ischemic change (Fig. 5).
In this study, we demonstrated that embolization with NBCA or small-sized PVA particles can induce muscle infarction and ischemia in SD rats, and surgical excision at the external iliac artery level can induce ischemia in SD rats. Considering that SD rats have a arteriolar shunt connecting the internal iliac artery to the popliteal artery, approximately 140 µm in size (18), the NCBA and PVA particles used in this study is likely to occlude the peripheral small arteries, including the preexisting collaterals of the popliteal artery side. Therefore, the muscles around the occluded arteries were infarcted. In contrast, the surgical ligation and excision was applied to the external iliac artery. Therefore, downstream arteries could be supplied by collateral flows from the internal iliac artery even with very low blood pressures.
Acute muscular infarction is accompanied by tissues edema manifested as volume increase in the affected muscle. Consequently, presence or absence of the muscular infarction according to the hindlimb ischemia methods might result in the different features of volume change on day 7. In other words, volume increase was noted in NBCA and PVA groups presenting muscle infarction, whereas not in the surgery group without infarction. Decreases in muscle volume on day 14 in the NBCA and PVA groups could be explained by resolution of the edema after the acute phase and shrinkage of muscle fibers.
The NBCA group presented a larger infarcted area than the PVA group. This result might be associated with the properties of the embolic materials. Recently, Takasawa et al. (19) reported that the minimal arteriole size could contain an NBCA-iodized oil mixture in the lumen of 2.2 µm to 3.4 µm, depending on the mixture ratio. Therefore, the NBCA-iodized oil mixture flowed into much more distal arterioles than the PVA particle, which was 150-250 µm in size, thus preventing collateral flows and subsequently creating a larger infarction area.
With different ischemic and infarcted conditions from these different experimental methods for hindlimb ischemia using SD rats, the precise and dedicated animal modeling can be expected. Embolization methods would be better for acute arterial embolic occlusion or thrombosis, whereas surgical method might be suitable for severe stenosis or discrete occlusion accompanied by collateral flow. Embolizations with NBCA inducing definite muscle infarction could also be applicable for studying muscular infarctions and its related change with escape from confounding effects by venous occlusions or incision wounds.
Different embolic property also may result in different feature on pathological examinations. After embolization with NBCA, the muscle infarction showed narrow transition zones with relatively clear cut divisions between the infarction and normal tissue, possibly due to very small arteriolar occlusion up to 3.4 µm in diameter. In contrast, the PVA particle used in this experiment was 150-250 µm in size, much larger than the NBCA occlusion, which might induce the arterial occlusion more proximally than the NBCA. Therefore, ischemic zone is thought to be generated showing small muscle calibers around the infarctions.
Various methods have been used in published hindlimb ischemia experiments, with different levels of vascular occlusion, ranging from a proximal ligation of common iliac artery to a distal ligation just distal to the saphenopopliteal bifurcation of the lower limb, and different surgical procedures, ranging from a single ligation to a complete excision. These variations lead to inconstant ischemic conditions. For example, the segmental excision of the external iliac artery induced only ischemia without infarctions in the calf muscles of the SD rats in this study, whereas ligation (20) or transection (17) of the common iliac artery, the femoral artery and all their branches have been shown to induce necrosis and fibrosis in rats of the same species. Variable ischemic conditions impede the comparisons of hindlimb ischemia results. Therefore, given the increase in experiments when using rats, the reference to the degrees of ischemia according to the level of occlusion in the hindlimb, a standard method should be further investigated and developed.
The lack of correlation between the clinical ischemia index and fraction of muscular infarction is interesting. The surgery group presented foot necrosis without any muscular infarctions, whereas some rats in the embolization groups manifested only mild edema of the foot even with a large level of infarction. This discrepancy underscores the need for deep tissue evaluation. The LDPI, the most widely used technique for the assessment of hindlimb ischemia in experimental studies, is a technique that measures blood perfusion in superficial tissues based on the Doppler shift by moving red blood cells to the illuminating coherent light. Unfortunately, the technique has disadvantages, including the fact that the perfusion of the relatively deepseated muscle is not being evaluated. In this context, the MR perfusion is a promising noninvasive tool. The feasibility for use of this technique in hindlimb ischemia models of rats has already been shown (12-14), although it was not addressed in this study.
The TOF-MRA was able to demonstrate the flow discontinuation at the arterial ligation sites or embolized segments, and also demonstrated a good correlation with the findings of conventional angiograms, thus, suggesting that we can evaluate whether the occlusion was successful in a noninvasive manner. In this study, twelve out of 120 segments were discordant between the MRAs and conventional angiograms. Eight segments were visible in the angiogram but not visible with TOF-MRA. These arterial segments presented antegrade fillings on the angiogram but very thin and weak flows that might be insufficient to produce an MR signal. The reason for the other four discordant segments is unknown at this stage. One possible explanation is vasospasm induced by a guided wire during angiography.
A limitation of our study is the small number of cases. In each group, two of the four rats were sacrificed at day 7, and the number of remaining rats at day 14 was too small to be analyzed statistically. A second limitation is the inflammatory cytokines that can affect angiogenesis was not being evaluated. In a previous experiment using HydroCoils, the level of cytokines was shown to be different between interventional embolization and surgical groups (11). The NBCA and PVA particles have different chemical and mechanical profiles which may result in the release of different cytokines. Another shortcoming is the lack of LDPI. A correlation between the ratio of the infarcted sizes as a deep-tissue response and the LDPI result as a superficial perfusion parameter was not being addressed. An understanding of this correlation is required and should be explored in a future study. Infarction on pathologic exam was not analyzed quantitatively. When preparing the pathological slice with blind cutting, the thigh and calf muscle did not guarantee the representative section, unlike the enhanced MR in which the representative one was selected easily, thus, quantitative evaluations could not be performed. A possible solution would be the summation of the serial sections which requires more cost and time.
In conclusion, our study demonstrated that the examined hindlimb ischemia model using embolization with NBCA and PVA particles in rats is a feasible model for future research use, and the muscle necrosis was evident as compared with the surgical model.
Figures and Tables
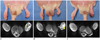 | Fig. 1Typical cases of three groups with gross picture (upper column) and enhanced MR of calf on day 14 (lower column).
A. Rats in NBCA group presented edema of distal toe and larger area of muscular infarction (*) than PVA group (B, lower column) with surrounding enhancements. B. Rat in PVA group presented distal toe necrosis and mild muscle atrophy with tiny necrotic foci (arrowhead) surrounded by large area of enhancement. C. Rat in surgery group presented foot necrosis and marked atrophic change of calf but no definite muscular necrosis. NBCA = N-butyl cyanoacrylate, PVA = polyvinyl alcohol
|
 | Fig. 2Ratio of infarcted muscle in thigh (A) and calf level (B). Both NBCA and PVA induced muscle infarction at both thigh and calf level. Infarct ratio was higher in NBCA group than in PVA group, but difference did not show statistical difference. Excision of external iliac artery did not result in muscular infarction. NBCA = N-butyl cyanoacrylate, PVA = polyvinyl alcohol |
 | Fig. 3Volume changes of calf in ischemic limb is compared with contralateral normal limb. NBCA and PVA groups presented volume increase on day 7, and then decrease on day 14, but surgery group presented gradual decrease. NBCA = N-butyl cyanoacrylate, PVA = polyvinyl alcohol |
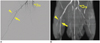 | Fig. 4Example of angiogram (A) and TOF-MRA (B) on day 14 of PVA group.
Left common iliac artery (curved arrows), left internal iliac artery (empty arrows), right popliteal artery (straight arrows), and right saphenous artery (arrowheads) are well correlated on both angiograms and TOF-MRAs. Note that small vessels are in left hindlimb, which are subcutaneous superficial arteries. TOF-MRA = time-of-flight magnetic resonance angiography, PVA = polyvinyl alcohol
|
 | Fig. 5Representative photomicrographs of hematoxylin and eosin staining of calf muscles.
A. In rat of NBCA group, short transition zone (arrowheads) was noted between infarcted (I) and normal tissues (N) (4 ×). B. In rat of PVA group, many leukocytes (empty errows) infiltrated around infarcted tissues (I), with smaller diameter of muscle cells suggesting ischemic changes (4 ×). C. In rat of surgery group, no infarcted tissues were noted, but multifocal small diameter of muscle fibers (arrow) was visible (10 ×). NBCA = N-butyl cyanoacrylate, PVA = polyvinyl alcohol
|
References
1. Ziegler-Graham K, MacKenzie EJ, Ephraim PL, Travison TG, Brookmeyer R. Estimating the prevalence of limb loss in the United States: 2005 to 2050. Arch Phys Med Rehabil. 2008; 89:422–429.
2. Beckman JA, Jaff MR, Creager MA. The United States preventive services task force recommendation statement on screening for peripheral arterial disease: more harm than benefit? Circulation. 2006; 114:861–866.
3. Hirsch AT, Haskal ZJ, Hertzer NR, Bakal CW, Creager MA, Halperin JL, et al. ACC/AHA 2005 Practice Guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease): endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter-Society Consensus; and Vascular Disease Foundation. Circulation. 2006; 113:e463–e654.
4. Asahara T, Kawamoto A. Endothelial progenitor cells for postnatal vasculogenesis. Am J Physiol Cell Physiol. 2004; 287:C572–C579.
5. Helisch A, Schaper W. Arteriogenesis: the development and growth of collateral arteries. Microcirculation. 2003; 10:83–97.
6. Heil M, Eitenmüller I, Schmitz-Rixen T, Schaper W. Arteriogenesis versus angiogenesis: similarities and differences. J Cell Mol Med. 2006; 10:45–55.
7. Menasché P. Cell therapy for peripheral arterial disease. Curr Opin Mol Ther. 2010; 12:538–545.
8. Mughal NA, Russell DA, Ponnambalam S, Homer-Vanniasinkam S. Gene therapy in the treatment of peripheral arterial disease. Br J Surg. 2012; 99:6–15.
9. Simons M, Ware JA. Therapeutic angiogenesis in cardiovascular disease. Nat Rev Drug Discov. 2003; 2:863–871.
10. Lingen MW. Role of leukocytes and endothelial cells in the development of angiogenesis in inflammation and wound healing. Arch Pathol Lab Med. 2001; 125:67–71.
11. Zhuang ZW, Shi J, Rhodes JM, Tsapakos MJ, Simons M. Challenging the surgical rodent hindlimb ischemia model with the miniinterventional technique. J Vasc Interv Radiol. 2011; 22:1437–1446.
12. Hsu LY, Wragg A, Anderson SA, Balaban RS, Boehm M, Arai AE. Automatic assessment of dynamic contrast-enhanced MRI in an ischemic rat hindlimb model: an exploratory study of transplanted multipotent progenitor cells. NMR Biomed. 2008; 21:111–119.
13. Sbarbati A, Marzola P, Nicolato E, Farace P, Asperio RM, Lunati E, et al. Dynamic MRI reveals that the magnitude of the ischemia-related enhancement in skeletal muscle is age-dependent. Magn Reson Med. 2003; 49:386–390.
14. Stekelenburg A, Strijkers GJ, Parusel H, Bader DL, Nicolay K, Oomens CW. Role of ischemia and deformation in the onset of compression-induced deep tissue injury: MRI-based studies in a rat model. J Appl Physiol (1985). 2007; 102:2002–2011.
15. Delli Pizzi S, Madonna R, Caulo M, Romani GL, De Caterina R, Tartaro A. MR angiography, MR imaging and proton MR spectroscopy in-vivo assessment of skeletal muscle ischemia in diabetic rats. PLoS One. 2012; 7:e44752.
16. Jaspers K, Versluis B, Leiner T, Dijkstra P, Oostendorp M, van Golde JM, et al. MR angiography of collateral arteries in a hind limb ischemia model: comparison between blood pool agent Gadomer and small contrast agent Gd-DTPA. PLoS One. 2011; 6:e16159.
17. Paek R, Chang DS, Brevetti LS, Rollins MD, Brady S, Ursell PC, et al. Correlation of a simple direct measurement of muscle pO(2) to a clinical ischemia index and histology in a rat model of chronic severe hindlimb ischemia. J Vasc Surg. 2002; 36:172–179.
18. Herzog S, Sager H, Khmelevski E, Deylig A, Ito WD. Collateral arteries grow from preexisting anastomoses in the rat hindlimb. Am J Physiol Heart Circ Physiol. 2002; 283:H2012–H2020.
19. Takasawa C, Seiji K, Matsunaga K, Matsuhashi T, Ohta M, Shida S, et al. Properties of N-butyl cyanoacrylate-iodized oil mixtures for arterial embolization: in vitro and in vivo experiments. J Vasc Interv Radiol. 2012; 23:1215–1221.
20. Tang GL, Chang DS, Sarkar R, Wang R, Messina LM. The effect of gradual or acute arterial occlusion on skeletal muscle blood flow, arteriogenesis, and inflammation in rat hindlimb ischemia. J Vasc Surg. 2005; 41:312–320.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download