Abstract
This manuscript describes an unusual case of rapid intra-hepatic dissemination of hepatocellular carcinoma with pulmonary metastases occurring 1 month after combined chemoembolization and radiofrequency ablation. Inferior vena cava and portal vein invasion tumor thrombus was also detected, possibly accounting for the mechanism of disease dissemination route of disease.
Rapid intra-hepatic dissemination of hepatocellular carcinoma (HCC) is a rare but known occurrence after radiofrequency ablation (RFA) of solitary HCCs (1-4). The exact incidence is unknown but it is believed to be underreported, and the mechanism of spread remain elusive (1-4). We hereby report a case of rapid intra-hepatic dissemination of HCC with pulmonary metastases that developed within 1 month of combined transarterial chemo-embolization (TACE) and RFA of a solitary HCC.
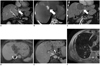
An 83-year-old man with alcoholic liver cirrhosis was treated with microwave ablation of a solitary HCC in segment 4A (2.8 cm in size), and developed tumor recurrence at the ablation site 5 months later (Fig. 1A, white arrow), with the recurrent tumor measuring 3.5 cm in size. Given the early recurrence with microwave ablation, the decision was made to perform combination therapy, using TACE and RFA. TACE was performed using a single vial of 100-300 um of DC Beads (Biocompatibles, Surrey, England, UK) loaded with 50 mg of doxorubicin. During arteriography, variant hepatic arterial anatomy was noted. The right lobe was supplied by a replaced right hepatic artery arising from the superior mesenteric artery, a replaced left hepatic artery arising from the gastrohepatic trunk to supply the segment II and III and the middle hepatic artery arising from the gastroduodenal artery supplied only segment 4 and the tumor. No other hypervascular tumors were detected on angiography in the rest of the liver. Cone-beam CT was performed following infusion of the DC beads from the middle hepatic artery (segmental TACE), showing good tumoral uptake of the DC beads (Fig. 1B, white arrow). While RFA was scheduled for the next day, it was delayed for 2 weeks for the patient to recover from a urinary tract infection following TACE.
Radiofrequency ablation was performed with Cool-tip RF system (Valleylab, Boulder, CO, USA), utilizing a switch-box with three active electrodes. Probe placement was performed under combined US and cone-beam CT guidance with no transgression of vascular structures. Active ablation was performed for a total of 16 minutes per device protocol, achieving tip temperature of 70-82 degrees Celsius in the electrodes. Track ablation was performed on completion of the ablation cycle and the patient tolerated the procedure well. The immediate post ablation contrast enhanced CT confirmed adequate ablation margins, with no evidence of disease in the lung bases and rest of the liver (Fig. 1C).
Four weeks after ablation, the patient complained of mild epigastric discomfort and on examination was found to have hepatomegaly. This was associated with close to 10 times elevation of serum alpha-fetoprotein levels from the pre-TACE/RFA level of 2662 ng/L to 24734 ng/L. Contrast enhanced MRI showed multiple (> 15), new 1-1.5 cm rim enhancing nodules around the ablation site and scattered in the left lobe (segment IV, II and III) (Fig. 1D, E). Additionally, tumor thrombus was also present in the inferior vena cava (Fig. 1D, curved white arrow) and the left portal vein (Fig. 1E, curved white arrow). Several new pulmonary nodules had developed, consistent with pulmonary metastases (Fig. 1F). The patient declined systemic therapy, received symptomatic treatment, and died 3 months later from diffuse metastatic disease.
Rapid intra-hepatic metastasis following RFA is a rare event and can be perplexing for clinicians following apparently technically successful loco-regional therapy. More importantly, it poses significant treatment challenges with poor prognostic implications, as mortality is high in these patients and treatment options become limited (4). In our case, the progression from early stage or Barcelona clinic Liver Cancer (BCLC) stage A to advanced stage or BCLC stage C occurred within 1 month, dictating a change from curative to palliative intent. Additionally, the progression excluded further loco-regional therapy, leaving only systemic therapy.
Our case shared several common features with those described in the literature: 1) most occurring within 6 months of RFA, 2) multiple tumors of similar sizes scattered around the ablation zone and liver lobe(s), 3) tumors located with no relation to the ablation track (compared to track seeding) (1-4). Several differences however are present: extremely rapid progression of disease within 1 month (usual reported timeframe of 2-6 months), development of venous invasion (left portal vein and inferior vena cava thrombus), and pulmonary metastases in a very short time span.
The pathophysiology of tumor dissemination is currently poorly understood. Our case supports the proposed mechanism of venous dissemination, as evidence of venous invasion is present in the territory of tumor recurrence. For instance, the presence of left portal vein tumor thrombus could account for tumors within the left lobe and sparing the right lobe; and the presence of inferior vena cava tumor thrombus would account for the pulmonary metastases. Segmental TACE 2 weeks prior to RFA also did not seem to have a "protective" effect on tumor dissemination even within the treated segment. It also suggests that the appearance of new tumors was likely related to RFA, rather than progression of pre-existing sub-radiologic tumor foci, as pre-existing tumor foci would have been controlled by the segmental TACE, at least in segment IV. Additionally, the variant arterial anatomy with origin of the middle hepatic artery (segment IV) arising from the gastro-duodenal artery and origin of the replaced left hepatic artery (segment II/III) from a gastrohepatic trunk suggest that the arterial route of dissemination is less likely.
From a technique viewpoint, rapid intra-hepatic metastases have only been reported with ablation using RFA, and can occur with both straight tip (e.g., Cool-tip) and expendable tinges (e.g., Leveen [Boston Scientific, Natick, MA, USA]) RFA electrodes (4). Modification of ablation protocol such as using a lower starting wattage to avoid rapid increase in intra-tumoral pressure and subsequent tumor "explosion" and dissemination of tumoral cells has been suggested (4). To this end, other ablative techniques such as ethanol injection that has no such reported cases has also been suggested, but ethanol ablation has limitations both from the technical and tumor control perspective (4). In the future, non-RF related technologies such as microwave ablation and irreversible electroporation could address whether this is a solely RFA related occurrence and serve as viable alternative options.
Figures and Tables
Fig. 1
Multimodality images (A) before TACE, (B) during TACE, (C-E) 4 weeks after TACE.
A. Contrast enhanced coronal T1 weighted MR image of solitary segment IV tumor (white arrow) showing areas of heterogeneous central enhancement. No other tumors were present in liver. B. Axial cone-beam CT image after segmental TACE with DC beads infused via middle hepatic artery showing dense tumoral uptake of beads (white arrow) with background staining and beads deposition in segment IV parenchyma. C. Contrast enhanced CT of liver reconstructed in coronal plane, obtained immediately post-RFA, showing ablation zone (white arrow) to be encompassing tumor (compare with A) with at least 1 cm margin. No other tumors in rest of liver and lung bases were seen. D, E. Contrast enhanced T1 weighted axial images MR images, obtained 4 weeks after RFA. D. Multiple small rim enhancing nodules of similar sizes were present in left lobe and around ablation zone (*), with sparing of right lobe. Small tumor thrombus in inferior vena cava was present (curved white arrow). E. Tumor thrombus in left portal vein (curve arrow) was present. Similar findings of multiple small nodules of similar sizes scattered in left lobe with sparing of right lobe were also noted. F. Ax T2 image of right lung obtained 4 weeks after RFA showed several new pulmonary nodules, consistent with metastases. Left lung base nodules of similar sizes were also present (not shown). These nodules were not present on CT performed immediate post-RFA. TACE = transarterial chemo-embolization, RFA = radiofrequency ablation
References
1. Nicoli N, Casaril A, Abu Hilal M, Mangiante G, Marchiori L, Ciola M, et al. A case of rapid intrahepatic dissemination of hepatocellular carcinoma after radiofrequency thermal ablation. Am J Surg. 2004; 188:165–167.
2. Ruzzenente A, Manzoni GD, Molfetta M, Pachera S, Genco B, Donataccio M, et al. Rapid progression of hepatocellular carcinoma after Radiofrequency Ablation. World J Gastroenterol. 2004; 10:1137–1140.
3. Mori Y, Tamai H, Shingaki N, Moribata K, Shiraki T, Deguchi H, et al. Diffuse intrahepatic recurrence after percutaneous radiofrequency ablation for solitary and small hepatocellular carcinoma. Hepatol Int. 2009; 3:509–515.
4. Kotoh K, Enjoji M, Arimura E, Morizono S, Kohjima M, Sakai H, et al. Scattered and rapid intrahepatic recurrences after radio frequency ablation for hepatocellular carcinoma. World J Gastroenterol. 2005; 11:6828–6832.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download